K.C.S.E Biology Q & A - MODEL 2017PP2QN02
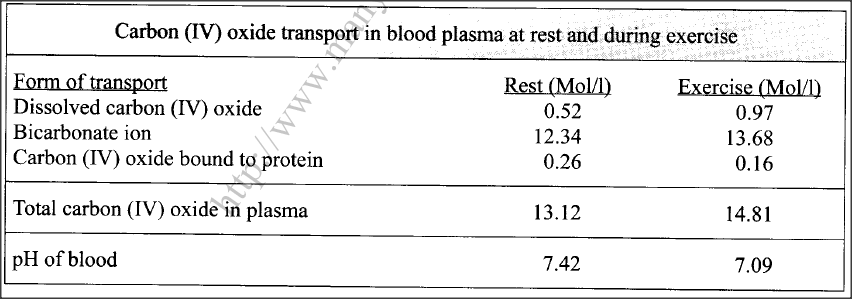
The table below shows variations in the form carbon (IV) oxide is transported in the blood at rest and during physical exercise.
(a) Explain why more carbon (IV) oxide ¡s transported in the form of bicarbonate ion.
(b)Account for the high total plasma content of carbon (IV) oxide during exercises. (c) State how one’s involvement in the exercises affects blood pH. (d) Name the protein responsible for the transport of carbon (IV) oxide in the blood.
ANSWERS
(a) Presence of carbonic anhydrase enzyme; which speeds up the conversion of carbon (IV) oxide to weak carbonic acid; which dissociates into hydrogen carbonate ion/(HCO3) (that diffuses out of the red blood cells into the blood plasma);
(b)The body needs high amount of energy; (for the exercise/muscle activity) hence high respiration rate (more oxygen intake); releasing more carbon (IV) oxide (in the blood plasma); (c)The high rate of respiration (during physical exercises coupled with normal cellular metabolism) results in the production of more carbon(IV) oxide/faster accumulation of lactic acid; lowering the blood plasma pH/making it more acidic (compared to when one is at rest); (d)Haemoglobin
0 Comments
Leave a Reply. |
Archives
December 2024
Categories
All
TOPICSFORM 1
Form 2
Form 3
Form 4
|
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |







 RSS Feed
RSS Feed

