A mass of 2.5g of acid HX was dissolved in water and the resulting solution was diluted to a total of 250cm3, 15cm3 of the final solution was required to neutralize 25.0cm3 of 0.1M aqueous potassium hydroxide. Calculate the relative molecular mass of the acid (3mks)
0 Comments
a) How do their rate of diffusion compare? (2mks)b) Determine the relative molecular mass of X given that the relative molecular mass of Y is 64 (1mk)a) Sugar crystals. (1mk)
b) Hydrated copper (II) sulphate solution (1mk)
c) What type of reaction has taken place above (1mk)
A fixed mass of an ideal gas occupies 200cm3 at a pressure of 740 mmHga) State Charles’s law (1mk)
b) Calculate the volume of the gas at 77-mmHg pressure (2mks)Alkaline earth metals are generally less reactive than alkali metals, explain.Alkaline earth metals loses two electrons while alkali metal lose one electron.
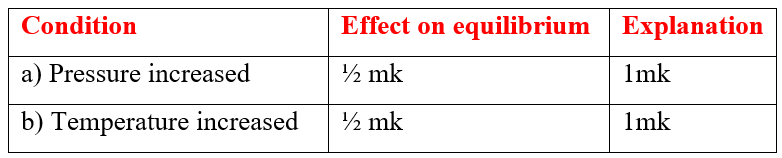
Ammonia gas is prepared by Haber process according to the equation belowN2(g) + 3H2(g) 2NH3(g) + HeatComplete the table below by stating the effect of equilibrium when the following conditions are applied. Give explanation in each caseSolutiona) Explain why sulphur is soluble in ethanol but hot in water (1mk)
b) Explain how a pure sample of sodium chloride can be obtained from a mixture of the two (1mk)
Explain why potassium is kept under paraffin while phosphorous is kept under waterPotassium does not react with paraffin but react with water while phosphorous react with paraffin but does not react with water
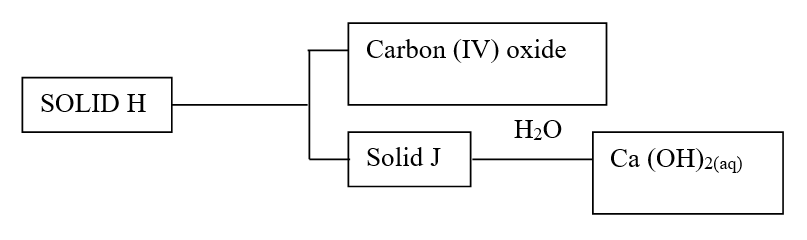
a) Identify the solids
|
| Molten M | Conduct electric current and is not decomposed |
| Molten N | Conduct electric current and a gas is formed at one of the electrodes |
Suggest the type of bonding present in;
a) Substance M (1mk)
Metallic bond
b) Substance N (1mk)
Ionic bond
Chemistry Topics
All
Acetylene Gas
Acid Bases And Indicators
Acids Bases And Salts
Air And Combustion
Alkaline
Ammonia
Calcium Carbonate
Carbon And Its Compounds
Charles's Law
Chemical Families
Chemical Reactions
Chemistry Practical Assignments
Chlorine And Its Compounds
Cleansing Agent
DashBoard
Definition Of Chemistry Terms
Differences In Chemistry
Diffusion
Down's Cell
Electric Current On Substance
Electrochemistry I And II
Empirical Formula
Energy Changes In Chemical And Physical Processes
Ethanol
Ethene Gas
Form 1 Level
Form 2
Form 3
Form 4
Gas Laws
General Chemistry
Haber Process
Hard Water
Hydrochloric Acid
Introduction To Chemistry
Kcse-1995
Kcse-1996
Kcse-1997
Kcse-1998
Kcse 1999
Kcse-2000
Kcse-2001
Kcse-2002
Kcse-2003
Kcse-2004
Kcse2005
Kcse-2005
Kcse-2006
Kcse2007
Kcse-2007
Kcse-2008
Kcse-2009
Kcse-2010
Kcse-2011
Kcse-2012
Kcse-2013
Kcse-2014
KCSE 2015
KCSE 2016
KCSE 2017
KCSE 2018
Kcse 2019
Metals
Molecular Formula
Nitrogen And Its Compounds
Notes And Tutorials
Nuclear Reactions
Organic Chemistry I
Organic Chemistry II
Oxidation
Oxygen
Paper 1
Paper 2
Paraffin
Periodic Table
Phenolphthalein Indicator
Phosphorous
Potassium
Propane
Properties-and-trends-across-the-period
Radioactivity
Reaction Rates And Reversible Reactions
Salts
Simple Classification Of Substances
Sodium
Structure Of The Atom And The Periodic Table
Structures And Bonding
Sulphur And Its Compounds
Sulphuric Acid
The Mole
Water And Hydrogen
Zinc
Archives
December 2024
January 2024
December 2023
November 2023
October 2021
November 2020
October 2020
September 2020
July 2020
May 2020
August 2019
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |


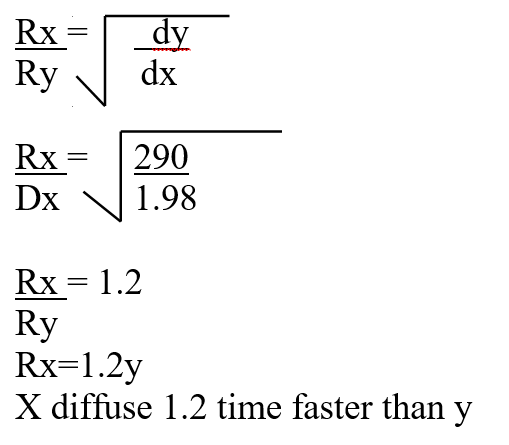

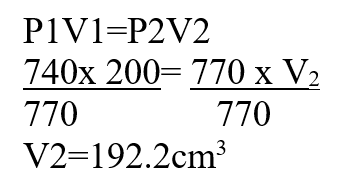


 RSS Feed
RSS Feed

