|
a) Study the information in the table below and answer the questions that follow.
(The letters do not represent the actual symbols of the elements).
c) Neutralization is one of the methods of preparing salts. i) What is meant by neutralization? ii) Describe how you would prepare crystals of sodium nitrate starting with 200cm3 of 2M sodium hydroxide. iii) Write an equation for the reaction that takes place when a solid sample of sodium nitrate is heated.
0 Comments
a) State one cause of temporary hardness in water.
b) How does distillation remove hardness from water?
Expected Response
(a) Presence of Ca (HCO3) or mg (HCO3)2 (b) Water vaporizes and distils off leaving behind ions that cause hardness
The table below gives the solubility’s of substances T and U at 10°C and 40°C.
When an aqueous mixture containing 55g of T and 12g of U at 80°c was cooled to 10°c, crystals formed.
a) Identify the crystals formed b) Determined the mass of the crystals formed c) Name the method used to obtain the crystals
Aqueous hydrogen chloride reacts with potassium manganate (VII) to produce chlorine gas, while a solution of hydrogen chloride in methylbenzene has no effect
on potassium manganate (VII). Explain this observation
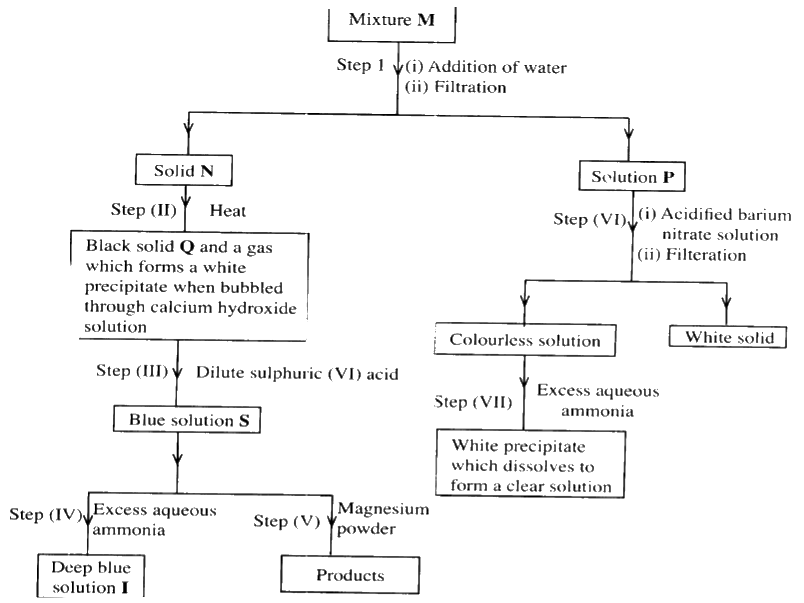
The flow chart below shows a sequence of reactions involving a mixture of two salts, mixture M. Study it and answer the questions that follow.
a) Write the formula of the following;
i) anion in solid Q ii) the two salts present in mixture M. b) Write an ionic equation for the reaction in step (VI) c) State and explain the observations made in step (V). d) i) Starting with Lead (II) oxide, describe how a pure solid sample of lead sulphate can be prepared in the laboratory. ii) How can one determine whether the lead sulphate prepared is pure?
ANSWERS
(c)The solution changes from blue to colourless and a brown solid is formed.
The magnesium which is above copper in the reactivity series displaces the copper ions from the solution. Apparatus become warm. The reaction is exothermic. (d)(i) Add nitric (V) acid to lead oxide, filter add a soluble sulphate/ sulphuric acid to the filtrate . Filter and wash residue with distilled water to remove traces of the filtrate, then dry residue between i. filter papers /oven. (ii) Determine the melting point, if it is pure the melting point will be constant.
Sample solutions of salt were labeled as I,II, III and IV. The actual solutions, not in that order are lead nitrate, zinc sulphate potassium chloride and calcium chloride.
(a) A student was supplied with a colourless liquid suspected to be water
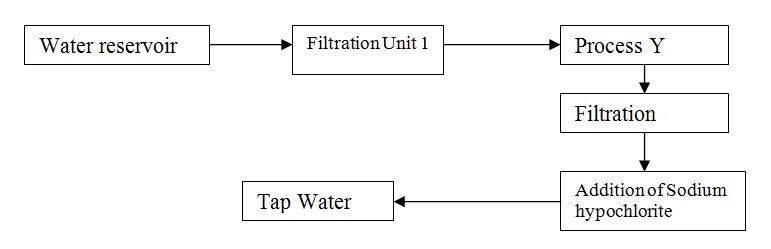
The flow chart below shows the various stages of water treatment. Study it and answer the questions that follow
II Addition of sodium hypochlorite (c) It was confirmed that magnesium sulphate was present in the tap water
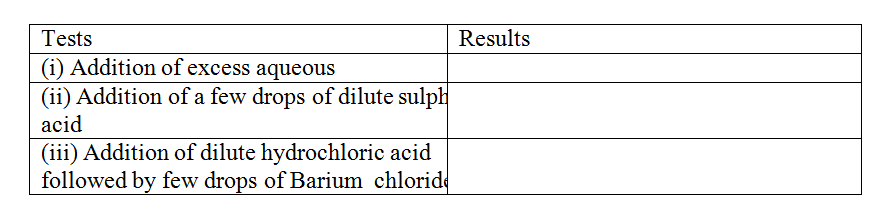
The table below shows the tests carried out on separate samples of water drawn from a well and the results obtained
(a) Identify the cation and the anion present in the water
Cation Anion (b) Write an ionic equation for the reaction which takes place in test (iii)
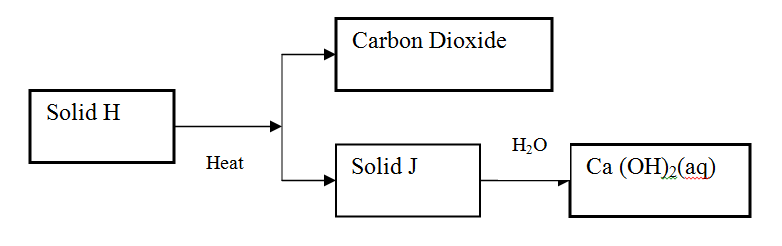
Use the scheme below to answer the questions that follow
(a) Identify the solid
H J (b) State one commercial use of solid J
Study the solubility curves below and answer the questions that follows
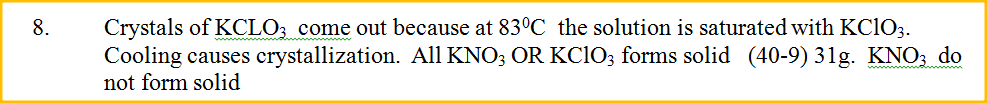
What happens when a solution containing 40gm of potassium chlorate and 40gm of potassium nitrate in 100gm of water at 900C is cooled to 400C? Explain
Starting with copper metal, describe how a solid samples of copper (II) carbonate can be prepared.
(a) Name two cations that are present in hard water.
(b) Explain how the ion exchange resin softens hard water.
Study the information in the table below and answer the questions that follow: A mixture containing 35g of CuS04 and 78g of Pb(N03)2 in l00g of water at 60°C was cooled to 40°C.
(a) Which salt crystallised out? Give a reason
(b) Calculate the mass of the salt that crystallised out. The table below shows the PH values of solutions I, II, III and IV.
(a) Which solution is likely to be that of calcium hydroxide?
(b) Select the solution in which a sample of aluminium oxide is likely to dissolve. Give reason for your answer
A sample of river water is suspected to contain zinc and sulphate ions.
Describe how the presence of zinc ions and sulphate ions can be established.
ANSWERS
When solid magnesium carbonate was added to a solution of hydrogen chloride in methylbenzene, there was no apparent reaction. On addition of water to the resulting mixture, there was vigorous effervescence. Explain these observations
State and explain one disadvantages of using hard water in boilers
Expected Response
On boiling hard water deposits CaCO3 which forma fur/ scales in the boiler. This reduces conductivity
Soap dissolves in water according to the equation below;
NaSt(aq) —> Na+(aq) + St- where St- is the stearate ion. (a) Write the formula of the scum formed when soap is used in hard water. (b) Write the ionic equation for the reaction that occurs when sodium carbonate is used to remove hardness in water.
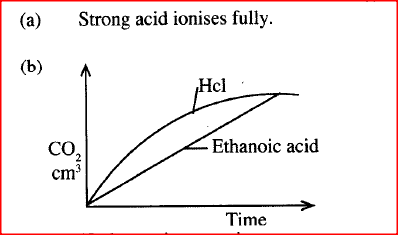
(a) What is meant by a strong acid?
(b) In an experiment 40cm3 of 0.5M hydrochloric acid was reacted with excess sodium carbonate and the volume of carbon (IV) oxide produced recorded with time. In another experiment, the same volume and concentration of ethanoic acid was also reacted with excess sodium carbonate and the volume of carbon (IV) oxide produced recorded with time. On the grid below, sketch and label the curves if the volumes of carbon (IV) oxide were plotted against time. (a) The table below gives the solubilities of hydrated copper (II) sulphate in mol dm3 at different temperatures (i) On the grid provided, plot a graph of solubility of copper (II) sulphate (vertical axis) against temperature. (ii) From the graph, determine the mass of copper (II) sulphate deposited when solution is cooled from 700C to 400C. (Molar mass of hydrated copper (II) sulphate = 250g) (b) In an experiment to determine the solubility of sodium chloride, 5.0cm3 of a saturate solution of sodium chloride weighing 5.35g were placed volumetric and diluted to a total volume of 250cm3. 25.0cm3 of the dilute solution chloride completely reacted with 24cm3 of 0.1M silver nitrate solution. AgNO3(aq) + NaCI(aq) → AgCl(s) + NaNO3(aq) Calculate: (i) Moles of silver nitrate in 24cm3 of solution (ii) Moles of sodium chloride in 25.0cm3 of sodium (iii) Moles of sodium chloride in 250cm3 of solution (iv) Mass of sodium chloride in 5.0cm3 of saturated sodium chloride Solution (Na = 23.0, CI = 35.5) (v) Mass of water in 5.0cm3 of saturated solution of sodium chloride (vi) The solubility of sodium chloride in g/100 water
A sample of river water was divided into three portions. The table below shows the test carried out on the portions and the observations made.
Complete the table by filling in the inferences.
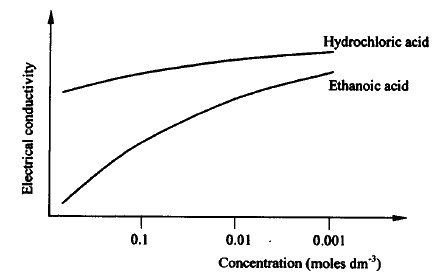
The curves below shows how the electronic conductivity of hydrochloric and ethanoic acids vary with concentration.
Explain why the electrical conductivity of 0.01M hydrochloric acids is higher than that of 0.01M ethanoic acid.
ANSWERS
A sample of fertilizer is suspected to be calcium ammonium nitrate. Describe chemical tests for each of the following ions in the sample:
a) Calcium ions; b) Ammonium ions.
ANSWERS
(a)Add a few drops of NaOH to an aqueous solution of the fertilizer. Forms white precipitate insoluble in excess. .
Add a few drops of aqueous sulphuric (VI) acid to another portion of aqueous solution of fertilizer. Forms a white precipitate insoluble in excess. (b)Heat the sample fertilizer in a test tube, and test gas evolved with damp red litmus paper, turn blue. Or add NaOH to the sample fertilizer and heat the mixture; test gas evolved using damp red litmus paper, turn blue.
The table below shows the solubility of a salt at various temperatures
What would happen if a sample of a saturated solution of the salt at 400C is heated to 800C. Explain
Starting with red roses, describe how;
(i) a solution containing the red pigment may be prepared (ii) the solution can be shown tobe an indicator.
ANSWERS
(i)Crush the roses with a suitable solvent Filter to obtain pigment
(ii)Add pigment to an acid . It turns read |
Chemistry Topics
All
Archives
December 2024
|
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |









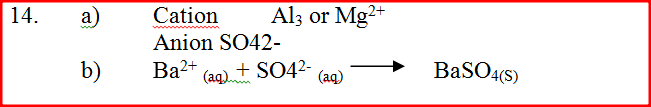




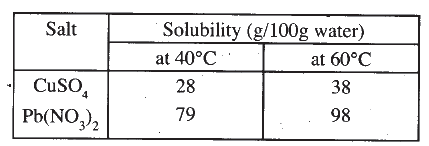

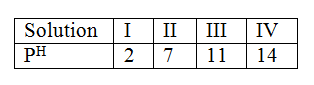









 RSS Feed
RSS Feed

