|
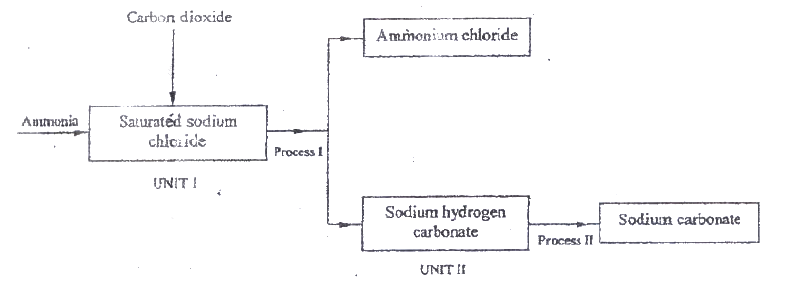
a) the schematic diagram shows part of the Solvay process used for the manufacture of sodium carbonate.
i) Explain how the sodium chloride required for this process is obtained from sea water.
ii) Two main reactions take place in UNIT I . The first one is the formation of ammonium hydrogen carbonate. I. Write an equation for this reaction II. Write an equation for the second reaction iii) State how the following are carried out: I Process I II) Process II iv)In an experiment to determine the percentage purity of the sample of sodium carbonate produced in the Solvay process, 2.15 g of the sample reacted completely with 40.0cm3 of 0.5 M sulphuric acid. I calculate the number of moles of sodium carbonate that reacted. II Determine the percentage of sodium carbonate in the sample. (Na= 23.0, C= 12.0, O = 16.0) b) Name two industrial uses of sodium carbonate
0 Comments
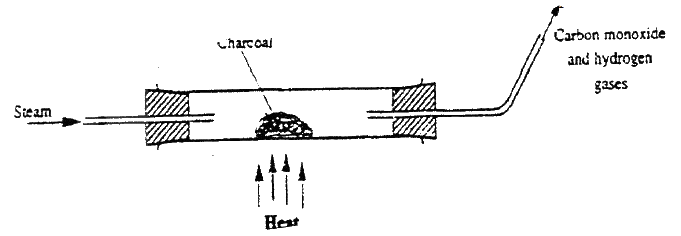
The diagram below represents a charcoal burner. Study it and answer the questions that follow
Write equations for the reactions taking place at I and I and II
Expected Response
ANSWERS
(a) Neutralization
(b)(i)Calcium hydrogen carbonate (ii)Drying agent Extraction of sodium metal
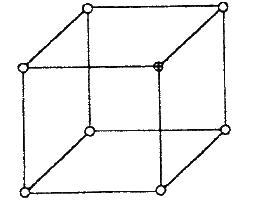
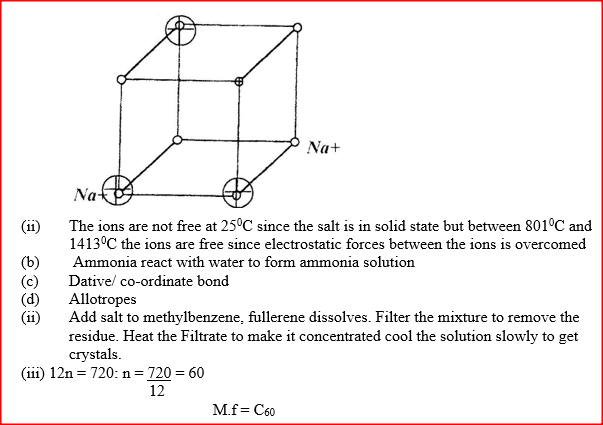
(a) The diagram below represents part of the structure of a sodium chloride crystal.
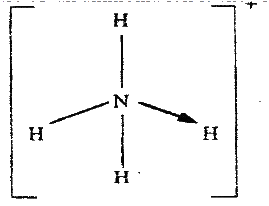
The position of one of the sodium ions in the crystal is shown as (i) On the diagram, mark the position of the other three sodium ions (ii) The melting and boiling points of sodium chloride are 801°C and 1413°C respectively. Explain why sodium chloride does not conduct electricity at 25°C, but does so at temperatures between 801° C and 1413°C (b) Give a reason why ammonia gas is highly soluble in water (c) The structure of an ammonia ion is shown below:
Name the type of bond represented in the diagram by N → H
(d) Carbon exists in different crystalline forms. Some of these forms were recently discovered in soot and are called fullerenes (i) What name is given to different crystalline forms of the same element? (ii) Fullerenes dissolve in methylbenzene while the other forms of carbon do not. Given that soot is a mixture of fullerenes and other solid forms of carbon, describe how crystals of fullerenes can be obtained from soot. (iii) The relative molecular mass of one of the fullerenes is 720. What is the molecular formula of this fullerene? (C=12.0)
ANSWERS
ANSWERS
(a) Carbon (IV) oxide
(b) Blue flame, carbon (II) oxide is burning
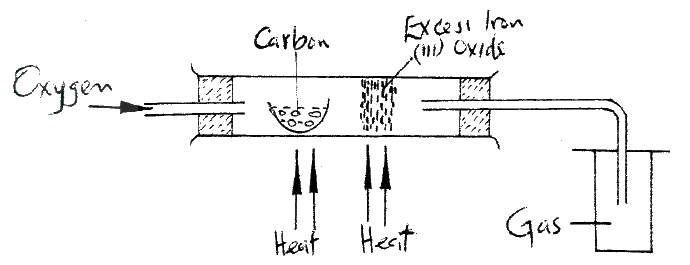
The diagram below represents part of a set – up used to prepare and collect gas T.
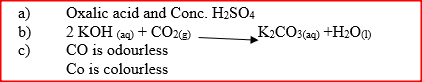
a) Name two reagents that are reacted to produce both carbon (IV) oxide and carbon(II) oxide.
b) Write the equation for the reaction which takes place in the wash bottles. c) Give a reason why carbon(II) oxide is not easily detected.
When extinguishing a fire caused by burning kerosene, carbon dioxide is used in preference to water .Explain
Expected Response
- Kerosene floats on water therefore it continues to burn
- Carbon dioxide blanket covers the flame OR cuts off the supply of oxygen
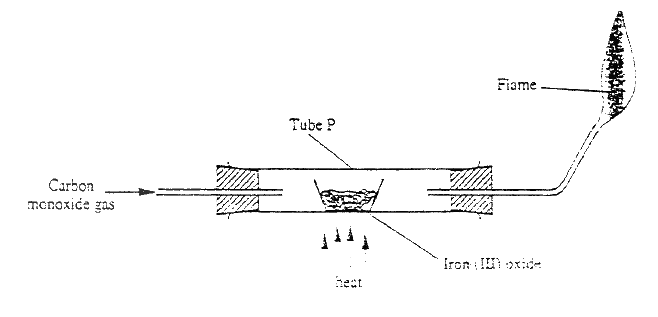
Dry carbon monoxide gas reacts with heated lead (II) oxide as shown in the equation below
PbO(s) + CO(g) →Pb (s) + CO2 (g) (a) Name the process undergone by the lead (II) oxide (b) Give a reason for your answer in (a) above (c) Name another gas that can be used to perform the same function as carbon monoxide gas in the above reaction.
ANSWERS
In terms of structure and bonding, explain why graphite is used as a lubricant.
ANSWERS
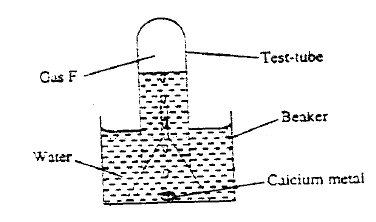
a) The set-up below was used to collect gas F, produced by the reaction between water and calcium metal.
(i) Name gas F
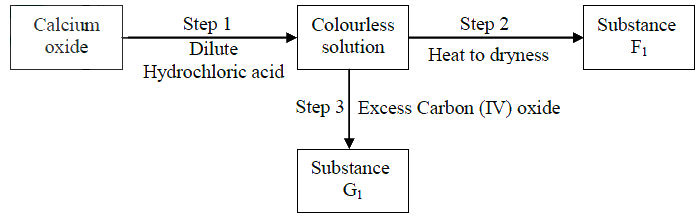
(ii) At the end of the experiment, the solution in the beaker was found to be a weak base. Explain why the solution is a weak base. (iii) Give one laboratory use of the solution formed in a beaker. (b) The scheme below shows some reactions starting with calcium oxide. Study it and answer the questions that follow.
(i) Name the reagents used in steps 2 and 4
Step 2 ………. Step 4 ………. (ii) write an equation for the reaction in step 3. (iii) Describe how a solid sample of anhydrous calcium sulphate is obtained in Step 5
Both diamond and graphite have giant atomic structures. Explain why diamond is hard while graphite is soft.
Answer
When carbon dioxide gas was passed through aqueous calcium hydroxide a white suspension was formed
(a) Write an equation for the reaction that took place (b) State and explain the changes that would occur when carbon dioxide gas is bubbled through the white suspension Give a reason why calcium hydroxide solution is used to detect the presence of carbon dioxide gas while sodium hydroxide in NOT?
Expected Response
Ca (OH)2(aq) forms white precipitate (1/2) with CO2 Can be observed NaOH(1/2)(aq) does not form a precipitate. (1mk) |
Chemistry Topics
All
Archives
December 2024
|
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |







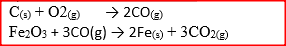


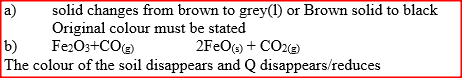
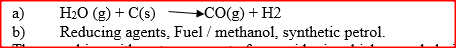
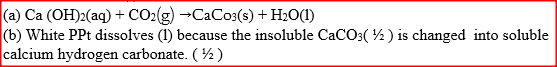

 RSS Feed
RSS Feed

