|
What is the oxidation number of chlorine in CO4
Expected Response
0 Comments
In an experiment, a test-tube full of chlorine water was inverted in chlorine water as shown in the diagram below and the set up left in sunlight for one day.
After one day, a gas was found to have collected in the test-tube
a) Identify the gas. b) What will happen to the PH of the solution in the beaker after one day? Give an explanation.
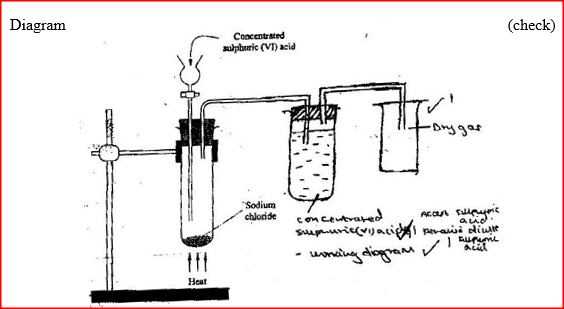
The diagram below shows the set up used in an experiment to prepare chlorine gas and react it with aluminium foil. Study it and answer the question that follow
(a) In the experiment, concentrated hydrochloric acid and potassium manganate (VII) were used to prepare chlorine gas. State two precautions that should be taken in carrying out this experiment.
(b) Write the formula of another compound that could be used instead of potassium manganate (VII) (c) Explain why it is necessary to allow the acid to drip slowly onto potassium manganate (VII) before the aluminium foil is heated. (d) State the property of the product formed in the combustion tube that makes it possible for it to be collected in the receiver (e) When 1.08g of aluminum foil were heated in a stream of chlorine gas, the mass of the product formed was 3.47 g Calculate the: (i) Maximum mass of the product formed if chlorine was in excess; (Al= 27; Cl = 35.5) (ii) Percentage yield of the product formed (f) Phosphorous trichloride is a liquid at room temperature. What modification should be made to set up if it is to be used to prepare phosphorous trichloride?
State and explain the observations made when excess ammonia gas reacts with chlorine gas
ANSWERS
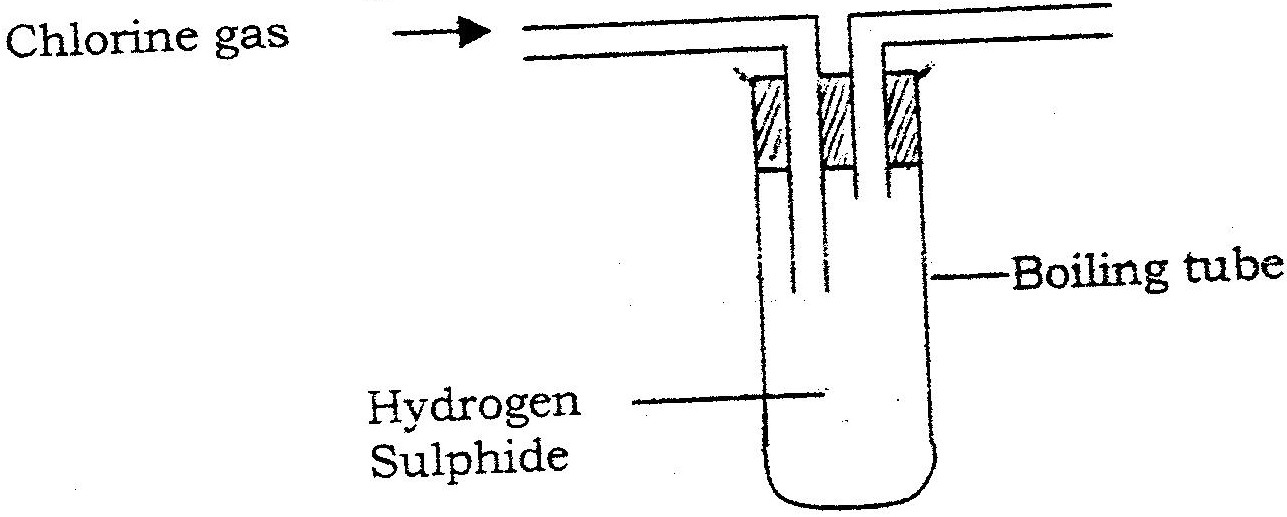
In an experiment, chlorine gas was passed into moist hydrogen sulphide contained in a boiling tube as shown in the diagram
a) What observation was made in the boiling tube?
b) Write an equation for the above reaction. c) What precaution should be taken in carrying out this experiment? Give a reason.
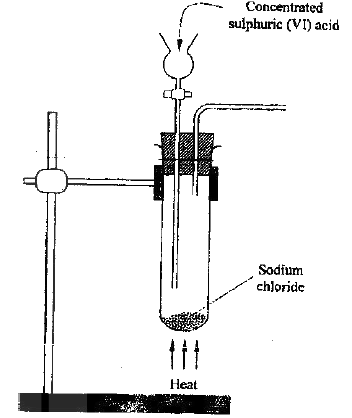
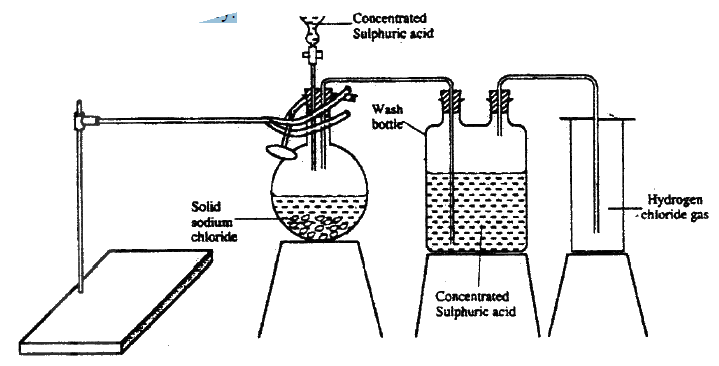
The diagram below represents the set-up that was used to prepare and collect hydrogen chloride gas in the laboratory.
a) State the purpose of concentrated sulphuric acid in the wash bottle.
b) Write an equation for the reaction between dry hydrogen chloride gas and heated iron c) Hydrogen chloride gas is dissolved in water to make hydrochloric acid. State one use of hydrochloric acid.
Name the process which takes place when:
a) Solid carbon (IV) oxide (dry ice) changes directly into gas b) A red litmus paper turns white when dropped into chlorine water. c) Propane gas molecules are converted into a giant molecule
ANSWERS
a)sublimation
b)Bleaching. c)Polymerization
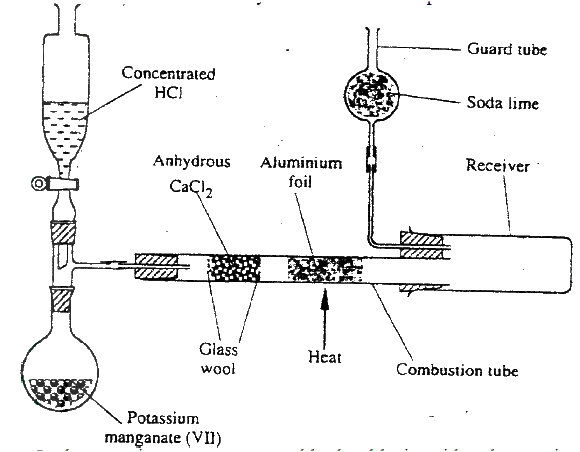
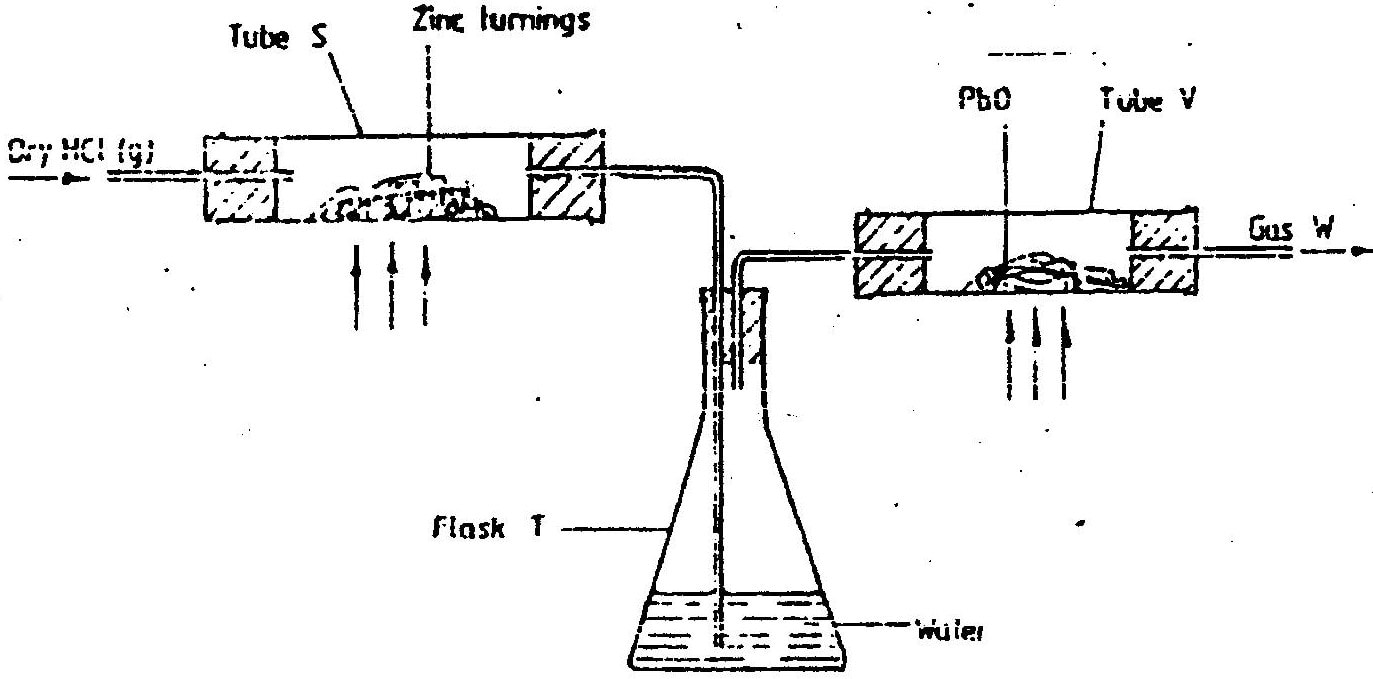
(a) In an experiment, dry hydrogen chloride gas was passed through heated zinc turnings as shown in the diagram below. The gas produced was then passed through heated lead (II) oxide.
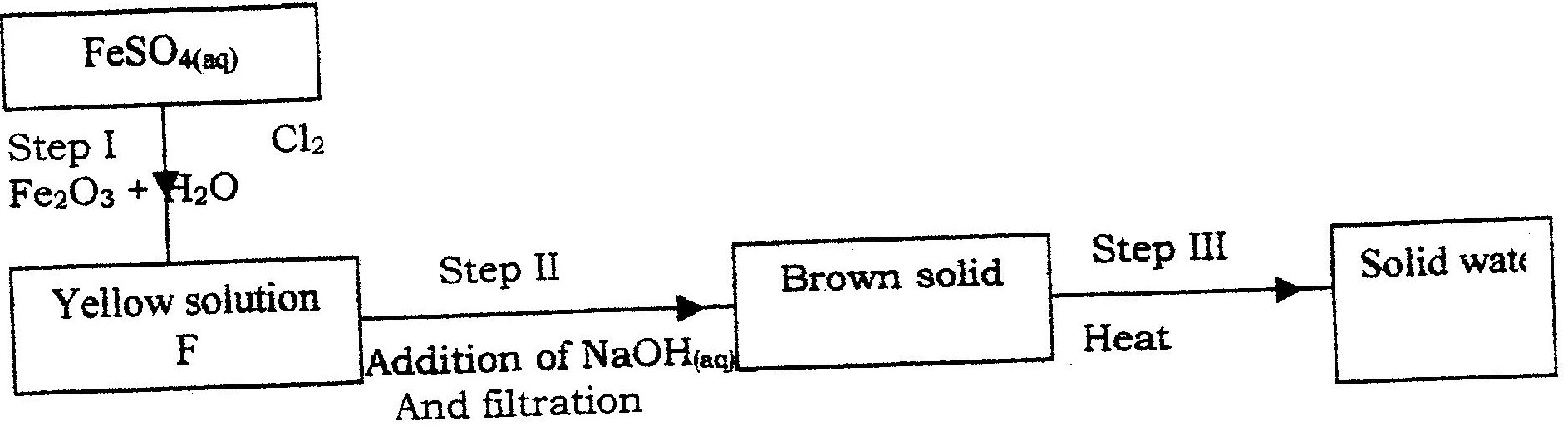
The reaction between how concentrated sodium hydroxide and chlorine produces sodium chlorate (V), sodium chloride and water
(a) Write the equation for the reaction (b) Give one use of sodium chlorate (V)
Calcium oxide can be used to dry ammonia gas.
a) Explain why calcium oxide is not used to dry hydrogen chloride gas b) Name one drying agent for hydrogen chloride gas
a) The table below shows properties of chlorine, bromine and iodine.
Complete the table by giving the missing information in (i),(ii) and (iii)
b) Chlorine gas is prepared by reacting concentrated hydrochloric acids with manganese (iv) oxide. i) Write the equation for reaction between concentrated hydrochloric acid and manganese (iv) oxide. ii) What is the role of manganese (Iv) oxide in this reaction c) i) Iron (II) chloride reacts with chlorine gas to form substance E. Identify substance E ii) During the reaction in c(i) above,6.30g of iron chloride were converted to 8.06 of substance E. Calculate the volume of chlorine used.(Cl = 35.5, Molar gas volume at room temperature = 24000cm3, Fe =56 ) d) Draw and name the structure of the compound formed when excess chlorine gas is reacted with ethane gas. e) Give the industrial use of chlorine Chlorine and iodine are elements in the same group in the periodic table. Chlorine gas is yellow white aqueous, iodine; I2(aq) is brown. a) What observation would be made if chlorine gas is bubbled through aqueous sodium iodide? Explain using and ionic equation. (2mks) b) Under certain conditions chlorine and iodine react to give iodine chloride, ICl3(s)- What type of bonding would you expect to exist in iodine trichloride? Explain (1mk)
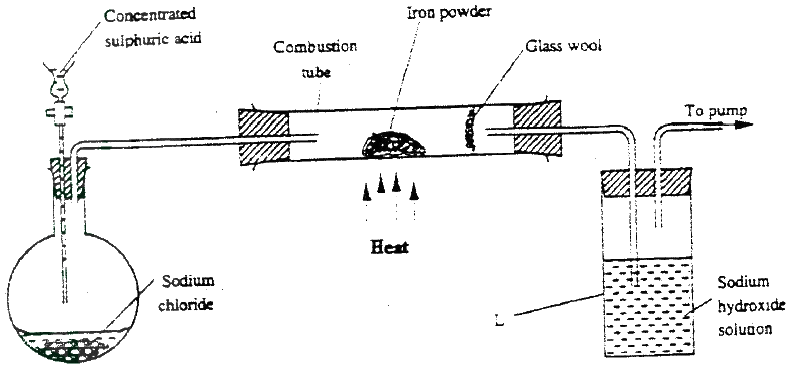
The set – up below was used to prepare hydrogen chloride a gas and react it with iron powder. Study it and answer the questions that follow.
At the end of the reaction, the iron powder turned into a light green solid.
a) Identity the light green solid. b) At the beginning of the experiment, the Ph of the solution in container L was about 14.. At the end, the pH was found to be 2. Explain
answers
The following two tests were carried out on chlorine water contained in two test tubes
(a) A piece of blue flower was dropped into the first – tube. Explain why the flower was bleached (b) The second test- tube was corked and exposed to sunlight after a few days, it was found to contain a gas that rekindled a glowing splint. Write an equation for the reaction which produced the gas a) In an experiment hydrogen chloride gas was prepared and reacted with aluminium turnings to form a solid q and gas R as shown in the diagram (i) Name: Liquid P : Solid Q (1mk) : Gas R (1mk) (ii) Name another substance that could serve the same purpose as the concentrated sulphuric acid. (1mk) (iii) Explain the following observation. When blue litmus paper was dipped into the water in the beaker at the end of the experiment it turned red. Explain why solid Q collects farther away from the heated aluminium (2mks) (b) (i) Write an equation for the reaction that takes place between ammonia gas and hydrogen gas (1mk) (ii) Calculate the mass of the product that would be formed when 2000cm3 of hydrogen chloride gas reacts completely with excess ammonia gas (H=1, O; N= 14.0, C1 = 35.5, one mole of gas occupied 24 litres at room temperature and pressure.) (3mks) |
Chemistry Topics
All
Archives
December 2024
|
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |













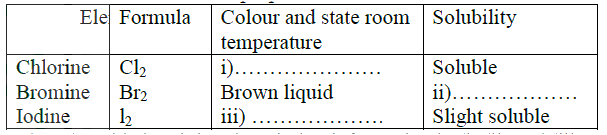








 RSS Feed
RSS Feed

