|
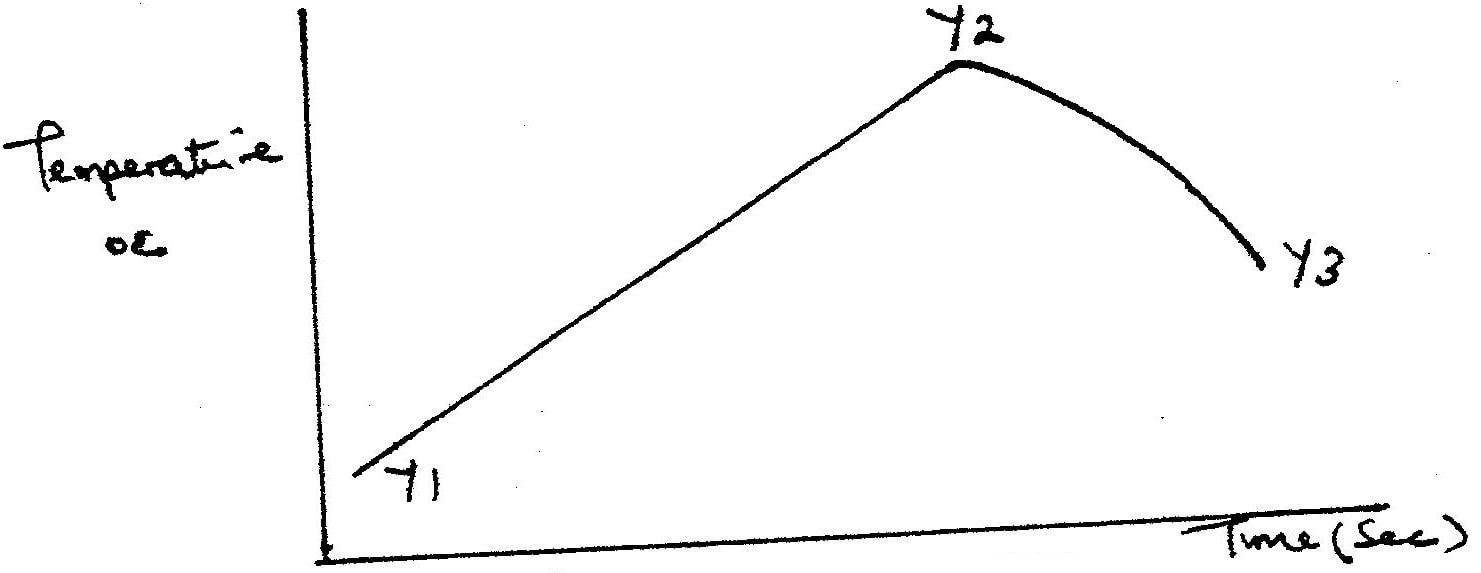
In order to determine the molar of neutralization of sodium hydroxide, 100cm3 of 1M sodium hydroxide and 100cm3 of 1 M hydrochloric acid both at the same initial temperature were mixed and stirred continuously with a thermometer. The thermometer of the resulting solution was recorded after every 30 seconds until the highest temperature of the solution was attained. Thereafter the temperature of the solution was recorded for a further two minutes
(a)
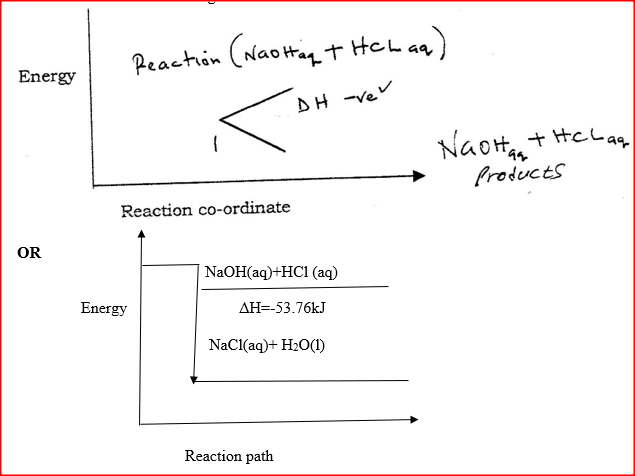
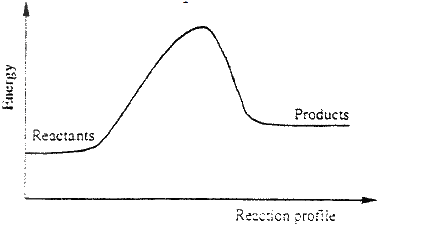
I. What is the significance of point Y2? II. Explain why there is a temperature change between points; Y1 and Y2 Y3 and Y4 (iv) In the initial temperature for both solutions was 24.50C and the highest temperature attained by the mixture was 30.90C Calculate the: I. heat change for the reaction (specific heat capacity of the solution = 4.2Jg -1K-1 and the density of the solution = 1.0g/cm3 II. Molar heat of neutralization of sodium hydroxide (v) Explain how the value of the molar heat of neutralization obtained in this experiment would compare with the one that would be obtained if the experiment was repeated using 100cm3 of 1 Methanoic acid instead of hydrochloric acid. (b) On the grid provided below, draw an energy level diagram for the reaction between hydrochloric acid and sodium hydroxide
0 Comments
ANSWER
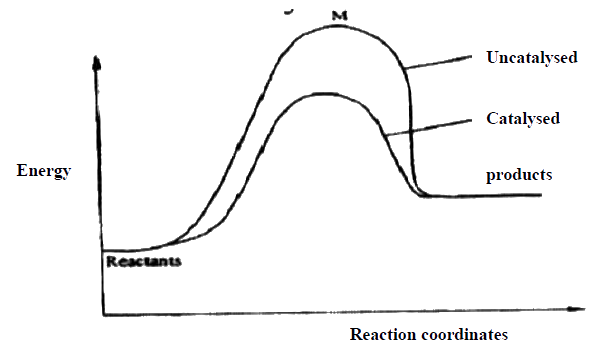
(a) Energy of the activated energy Therefore more molecules will take part in effective collision. Or Energy of the activated/intermediate complex of the unanalyzed reaction
(b) Catalyst lowers the activation energy therefore more molecules will take effective collision
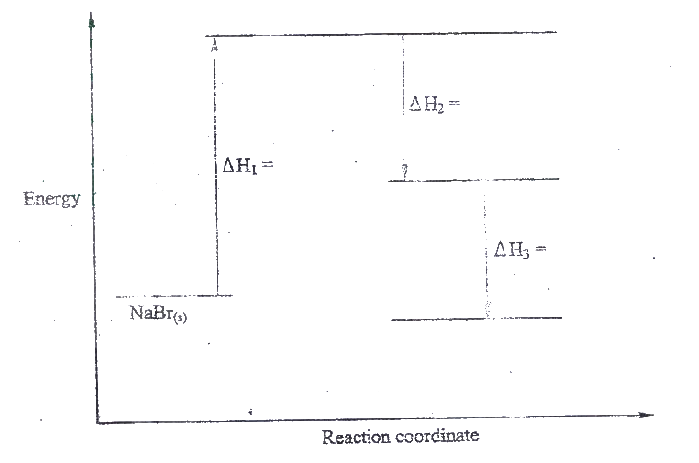
a) what is meant by molar heat of solution?
b) the lattice energy of sodium bromide and hydration energies of sodium and bromide ions are: 733,406 and 335 kJmol -1 respectively. i) Complete the energy cycle diagram below by inserting the values of ΔH1, ΔH2, and ΔH3
ii) Determine the molar heat of solution of solid sodium bromide.
(a) Define the standard enthalpy of formation of a substance
(b) Use the thermochemical equations below to answer the questions that follow.
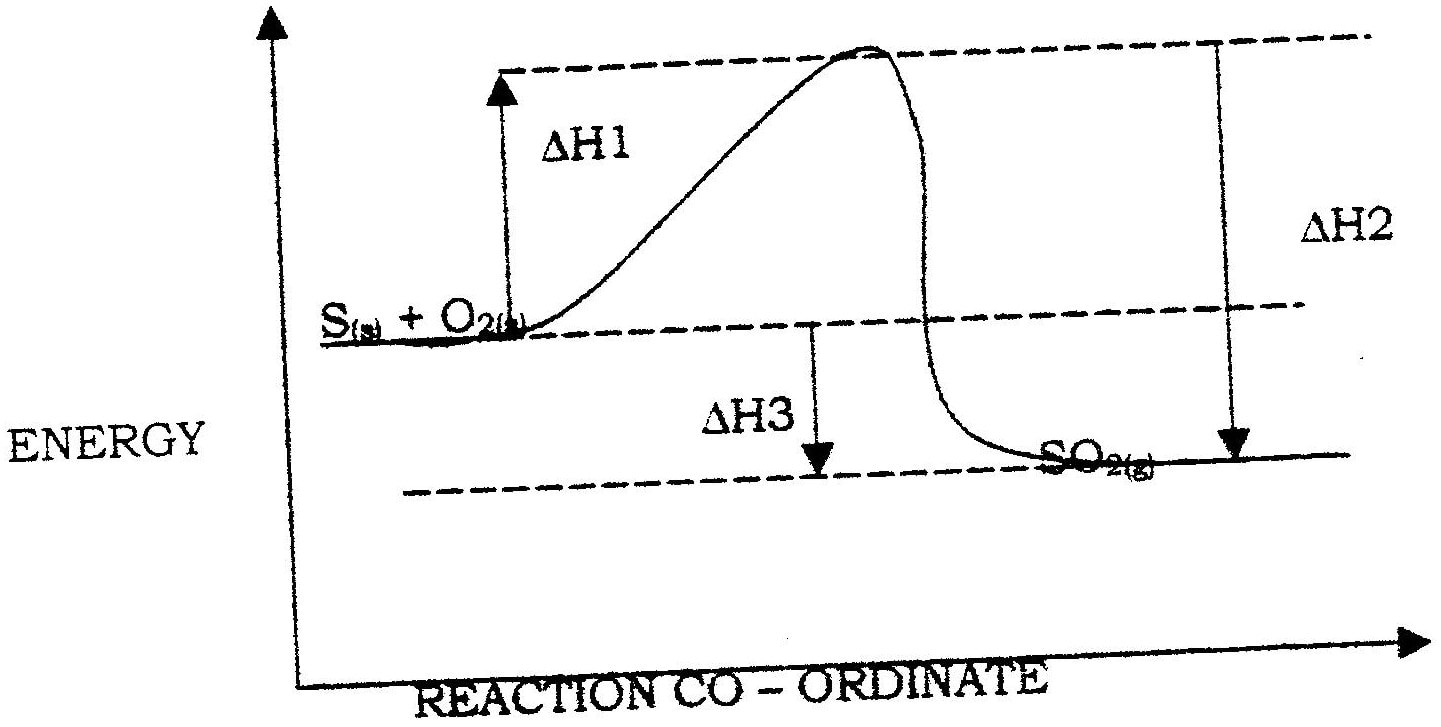
(i) Name two types of heat changes represented by ΔH3
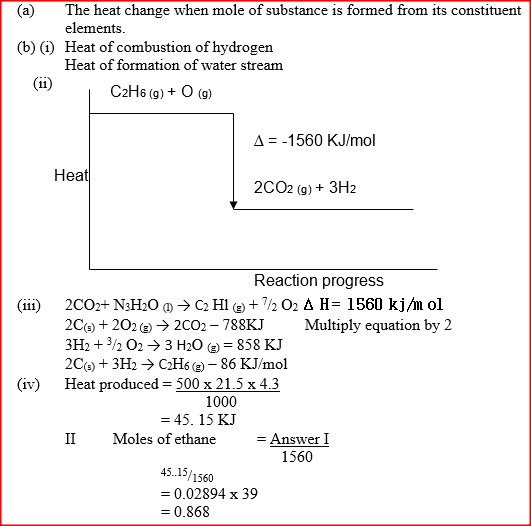
(ii) Draw an energy level diagram for the reaction represented by equation 1 (iii) Calculate the standard enthalpy of formation of ethane (iv) When a sample of ethane was burnt, the heat produced raised the temperature of 500g of water by 21. 5 K, (specific heat capacity of water = 4.2Jg-1K). Calculate the: I. Heat change for the reaction II. Mass of ethane was burnt. ( relative formula mass of ethane= 30)
(a) State two factors that should be considered when choosing fuel for cooking
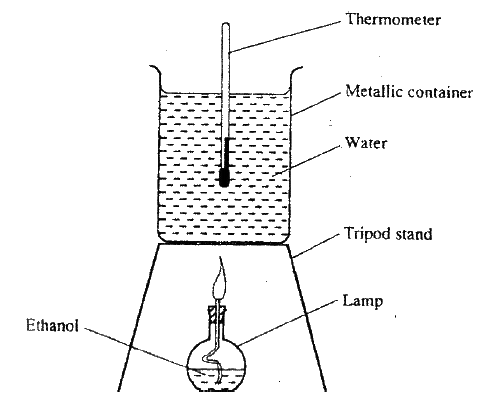
(b) The diagram below represents a set – up that was used to determine the molar heat of combustion of ethanol
During the experiment, the data given below was recorded
Calculate the:
(i) Heat evolved during the experiment (density of water = 1g/cm3 Specific heat capacity of water = 4.2 Jg-1K-1 (ii) Molar heat of combustion of ethanol (C = 12.0, O = 16.0, H=1.0) (c) Write the equation for the complete combustion of ethanol (d) The value of the molar heat of combustion of ethanol obtained in (b) (ii) above is lower than the theoretical value. State two sources of error in the experiment. When 0.6g of element J were completely burnt in oxygen and all the heat evolved was used to heat 500cm3 of water, the temperature of the water rose from 230C to 320C. Calculate the relative atomic mass of element J given that the specific heat capacity of water = 4.2JK-1g-1, density of water = 1.0g/cm3 and molar heat is combustion of J is 380Kjmol-1
ANSWERS
Methane reacts with oxygen according to the equation given below.
CH4 (g) + 2O2 (g) →CO2 (g) + 2H2O (l), ΔH = 890 KJ MOL-1 Calculate the volume of methane which would produce 111.25 kj when completely burnt.(Molar volume of a gas = 24 litres.)
expected Response
a) The energy change that takes place when one mole of the compound is formed from its constituents elements in their state
b) 3x-286 = 2x-394-(277) 858 + 788+ 277 = 11369kjmol
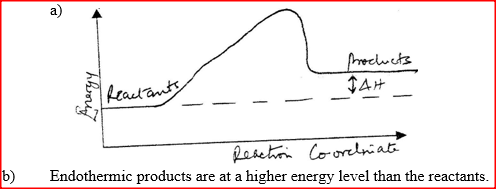
(a) Distinguish between exothermic and endothermic reaction
(b) Changes of state are either exothermic or endothermic Name a change of state that is: (i) Endothermic (ii) Exothermic (c) When pure water is heated at 1 atmospheric pressure at sea level, the temperature of the water does not rise beyond 100°C. Even with continued heated. Explain this observation.
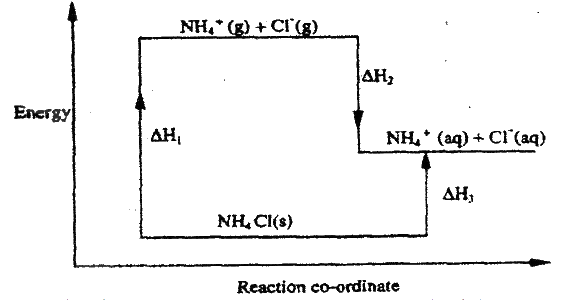
(d) Study the energy cycle diagram below and answer the questions that follow
(i) What does ΔH1 represent? (ii) Show the relationship between ΔH1, ΔH2 and ΔH3 (e) Butane and propane are constituents of a cooking gas. Which produces more energy per mole on combustion? Explain In an experiment to determine the heat of combustion of methanol, CH2OH a student used a set up like the one shown in the diagram below. a) Write an equation for the combustion of methanol b) Calculate: (i) The number of moles of methanol used in this experiment (C = 12; O = 16; H = 1) (ii) The heat of combustion per mole of methanol. (1mk) (iii) The heat of combustion per mole of methanol (2mks) (c) Explain why the value of the molar heat of combustion for methanol obtained in this experiment is different from the theoretical value. (d) On the axis below draw an energy level diagram for the combustion of methanol. Explain why the enthalpy of neutralization of ethanoic acid with sodium hydroxide is different from that of hydrochloric acid with sodium hydroxide. (2mks)
Expected Response
Enthalpy of neutralization between CH3CaOH(aq) and NaOH(aq) is lower than thatbetween HCl(aq) and NaOH because CH3CaOH(aq) is a weak acid which does not dissociate fully in water thus some of heat produced is used for dissociation fully dissociated and partially dissociated. (2mks) |
Chemistry Topics
All
Archives
December 2024
|
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |


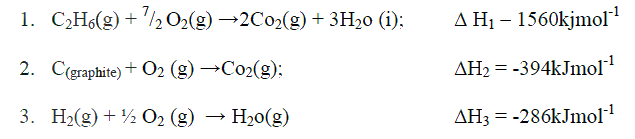
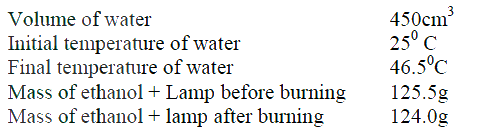




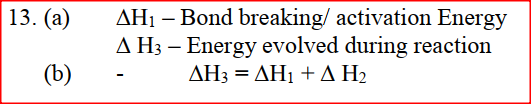


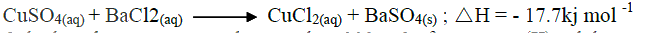
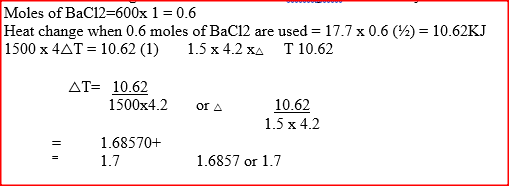
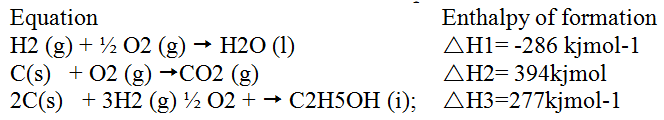











 RSS Feed
RSS Feed

