|
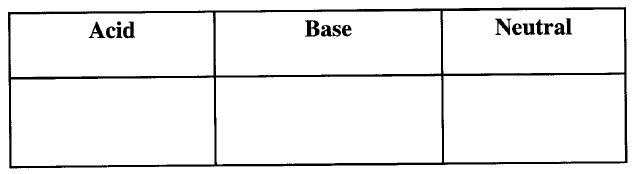
(a)Complete the following table. (2 marks)
(b) Explain why a solution of ammonia in methylbenzene has no effects on red litmus paper while in aqueous ammonia red litmus paper turns blue. (1 mark)
0 Comments
Given the following substances: wood ash, lemon juice and sodium chloride.
(a) Name one commercial indicator that can be used to show whether wood ash, lemon juice and sodium chloride are acidic, basic or neutral. b) Classify the substances in 15(a) above as acids, bases or neutral
ANSWERS
(a) Universal indicator / litmus paper
(b) Acid, base, neutral.
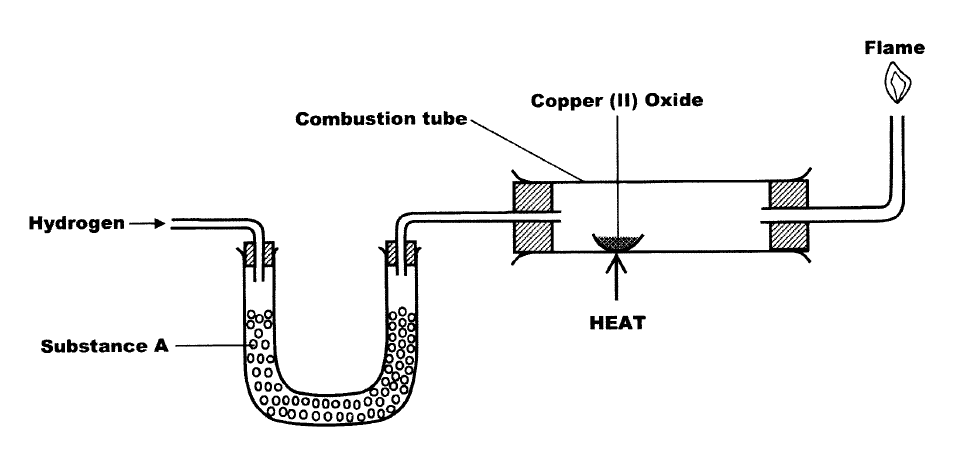
The set up below was used to investigate the reaction between dry hydrogen gas and copper (II) oxide
a) Name substance A
b) state the observation made in the combustion tube c) Explain the observation stated in (b) above.
ANSWERS
(a) Substance A - Calcium Oxide- fused calcium chloride
- Accept any other answer (b) Black Copper (II) Oxide (Solid) changes to brown Colourless liquid formed on the cooler part of the combustion tube. (c) Copper (II) Oxide is reduced to Copper metal.
During laboratory preparation of oxygen, manganese(IV) oxide is added to reagent H.
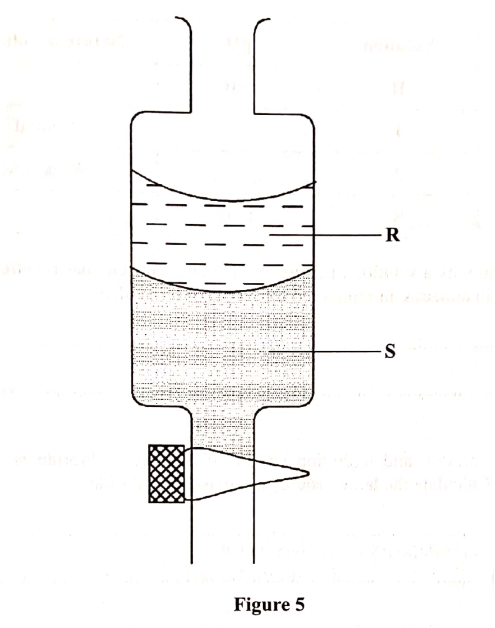
Figure 5 shows an apparatus used to separate a mixture of water and hexene.
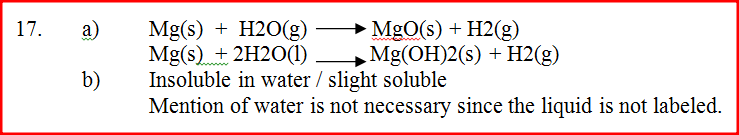
A farmer intended to plant cabbages in his farm. He first tested the pH of the soil and found it to be 3.0. If cabbages do well in alkaline soils, explain the advice that would be given to the farmer in order to realise a high yield. (2 marks)
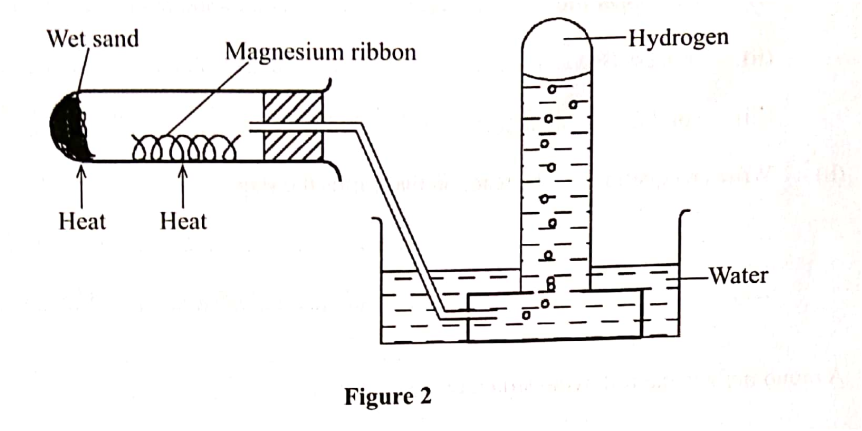
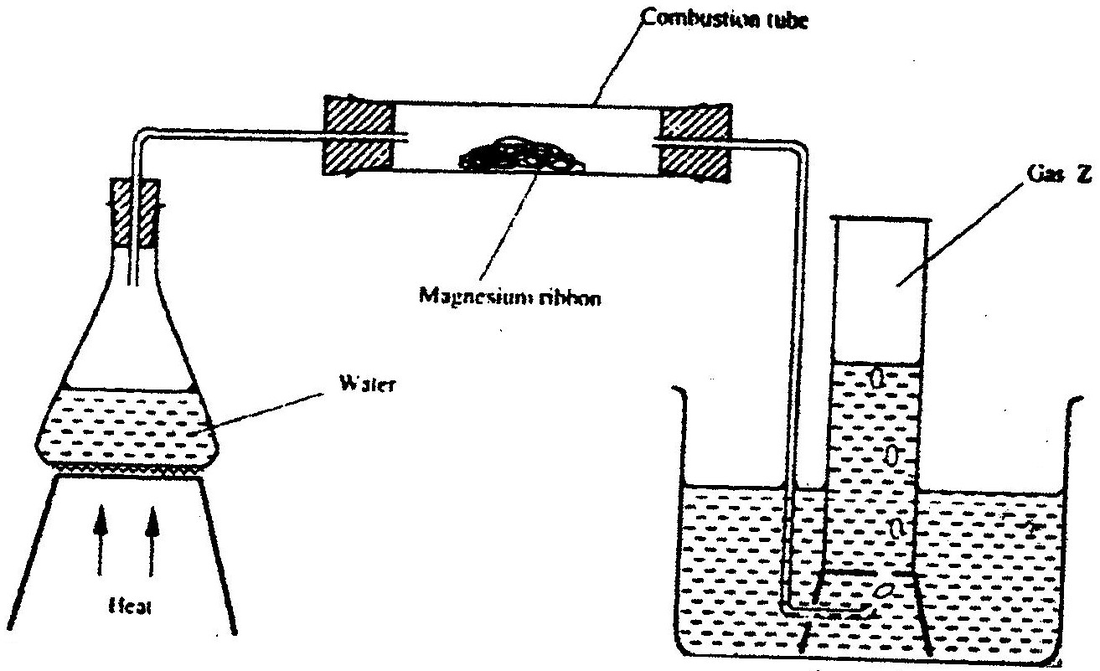
Hydrogen gas can be prepared by passing steam over heated magnesium ribbon as shown in Figure 2.
(a) Write an equation for the reaction that produces hydrogen gas. (1 mark)
(b) Explain why the delivery tube must be removed from beneath the water before heating is stopped. (1 mark) (c) Explain why sodium metal is not suitable for this experiment. (1 mark)
a) Name a suitable solvent for extracting an indicator form flowers:
b) Give a reason why the solvent named in (a) above is used
ANSWERS
(a) Acetone / ethanol / propanone / propanol.
(b) The solvent dissolves the organic compound indicator present in the flowers / it is an organic solvent.
The set up below was used to separate a mixture of methanol and propanol. Study it and answer the question that follow.
a) State the function of X
b) Which liquid will collect first in the beaker? give a reason.
ANSWERS
Draw a set up that can be used to separate a mixture of sand and iodine
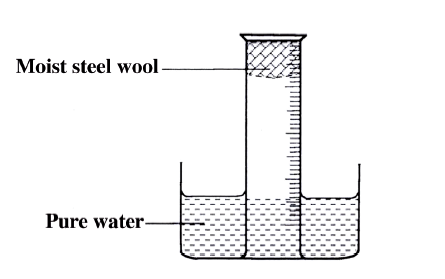
A measuring cylinder fitted with moist steel wool was inverted in a trough of water as shown in the diagram below
(a)State and explain the observations made on the;
i) Moist steel wool after four days. ii) Water level in the measuring cylinder after four days. (b) What would be the effect of using steel wool moistened with salty water?
ANSWERS
(a) (i) It turned brown /blue/violet/green.
(ii) The water level rose up the gas jar/occupy space left by reacted 02 (b) The brown colour would be more since the salt accelerates rusting/rust faster.
Explain how the hotness of a Bunsen burner flame can be increased
ANSWER
The Ph of a sample of soil was found to be 5.0.An agricultural office recommended the addition of calcium oxide in the soil. State two functions of the calcium oxide in the soil.
Expected Response
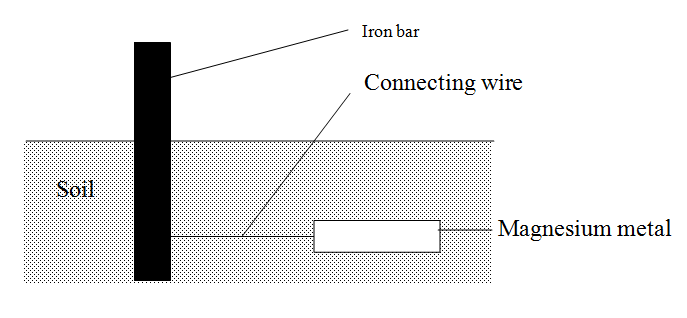
The diagram below shows an iron bar, which supports a bridge. The Iron bar is connected to a piece of magnesium metal
Explain why it is necessary to connect the piece of magnesium metal to the iron bar.
Expected Response
Magnesium is above iron in the activity series. It supplies electrons to the iron bar Hence prevent it from rusting
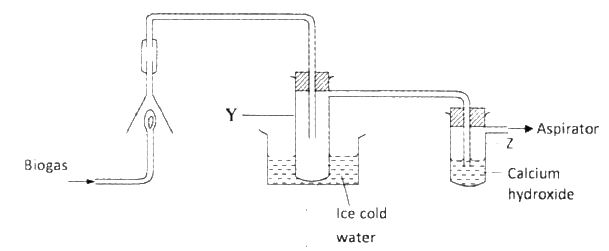
The set-up below was used to investigate the products of burning biogas (methane).
Study it and answer the questions that follow.
a) What product will be formed in test- tube Y?
b) State and explain the observations which would be made in Z?
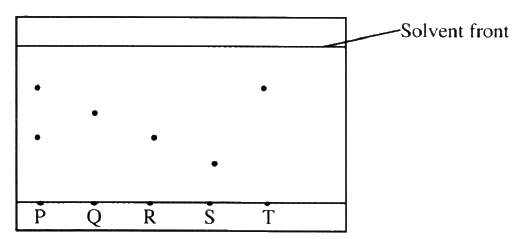
The chromatogram below was obtained from a contaminated food sample P.
Contaminants Q, R, S and T are suspected to be in P. Use it to answer the following questions.
a) Identify the contaminants in mixture P.
b) Which is the most soluble contaminant in P.?
Describe an experiment procedure that can be used to extract oil from nut seeds
ANSWERS
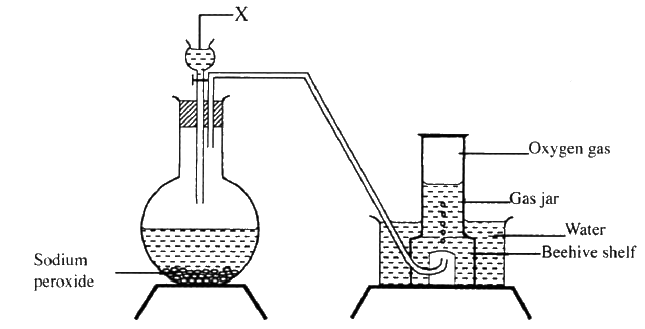
The set up below can be used to prepare oxygen gas. Study it and answer the questions that follow.
(a) Identify X
(b) What property of oxygen makes it possible for it to be collected as shown in the above set up? (c) State two uses of oxygen 10gm of sodium hydrogen carbonate were dissolved in 20cm3 of water in a boiling tube. Lemon juice was then added drop wise with shaking until there was no further observable change.
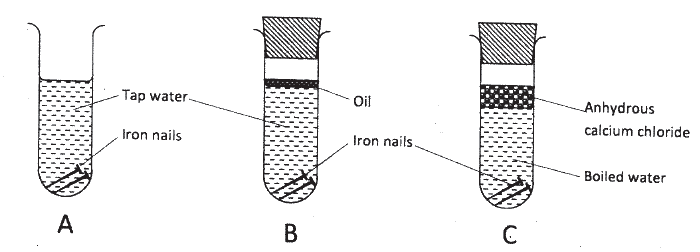
The following set up of three test-tubes was used to investigate rusting of iron.
Study it and answer the questions that follow.
(a) Give a reason why rusting did not occur in test-tube C.
(b) Aluminium is used to protect iron sheets from rusting. Explain two ways in which aluminium protects iron from rusting.
ANSWERS
(a) No air due to boiling.
(b) Aluminium being very reactive forms a layer of Al2O3 on the metal making it impervious to moisture. Aluminium being more reactive than iron protects the iron through sacrificial protection cathodic protection.
Name another gas, which is used together with oxygen in welding
Expected Response
Acet5ylene (ethyne) or Hydrogen
Study the set- up below and answer the questions that follow
(a) Write an equation for the reaction, which take place in the combustion tube
(b) What property of gas Z allows it to be collected as shown in the diagram
State any two differences between luminous and non – luminous flames
Explain the change in mass that occurs when the following substances are separately heated in open crucibles
(a) Copper metal (b) Copper (II) nitrate
Expected Response
a) Mass increases because oxygen combine with copper metal
b) Mass decreases it decomposes into gases that escape.
A sample of water in a beaker was found to boil at 101.5°C at 1 atmospheric pressure.
Assuming that the thermometer was not faulty, explain this observation.
ANSWERS
|
Chemistry Topics
All
Archives
December 2024
|
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |












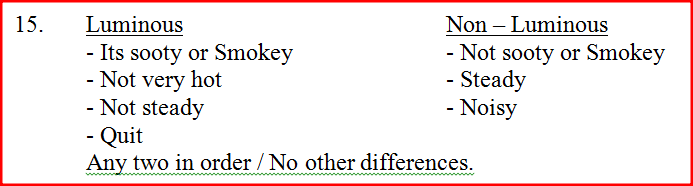

 RSS Feed
RSS Feed

