|
Copper(II) ions react with excess aqueous ammonia to form a complex ion.
(a) (i) Write an equation for the reaction that forms the complex ion. (ii) Name the complex ion. (b) Explain why CH4 is not acidic while HCl is acidic yet both compounds contain hydrogen.
0 Comments
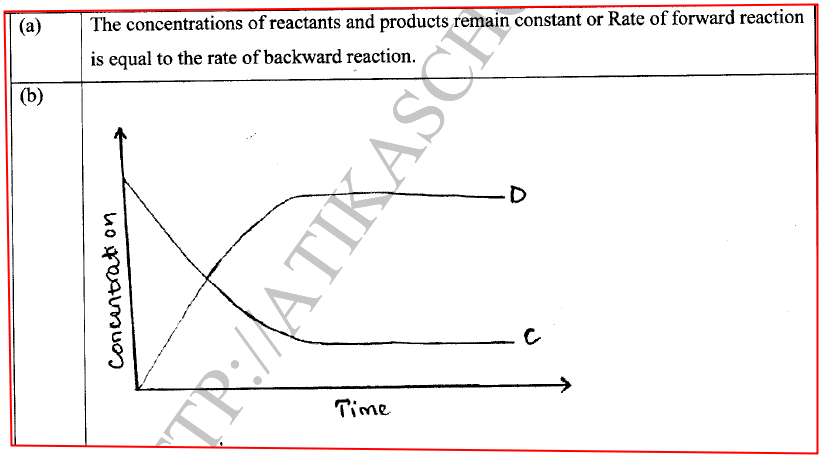
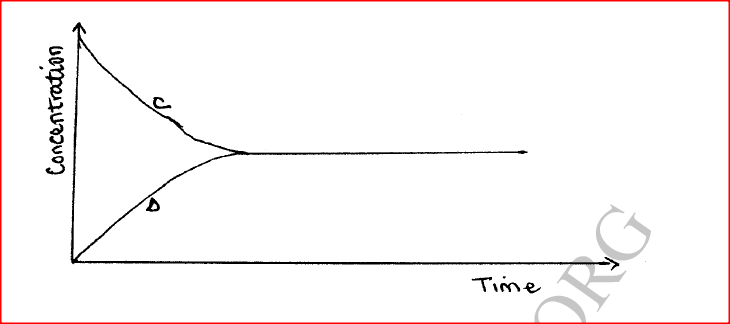
(a) State one characteristic of a reaction where equilibrium has been attained.
A sample of water is suspected to contain sulphate ions. Describe an experiment that can be carried out to determine the presence of sulphate ions.
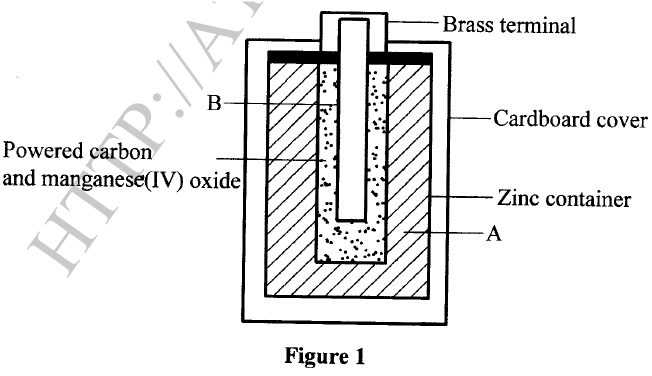
The diagram in Figure 1 shows a section of a dry cell. Study it and answer the questions that follow.
(a) Name the part labelled B.
(b) The part labelled A is a paste. Give a reason why it is not used in dry form. (c) What is the purpose of the zinc container?
ANSWERS
(a)Carbon electrode (Anode) /Graphite electrode.
(b)To allow movement of ions / to have it as an electrolyte. When dry, the ions are immobile. (c)It is the cathode / negative electrode.
Calculate the values of X and Y in the following nuclear equation.
ANSWERS
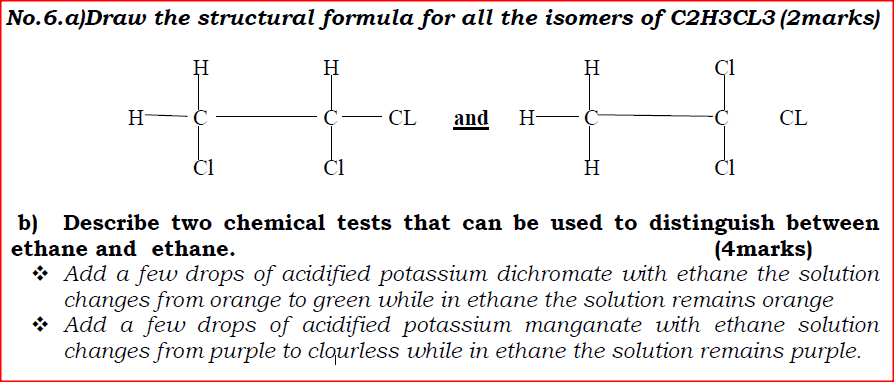
a)Draw the structural formula for all the isomers of C2H3CL3
b) Describe two chemical tests that can be used to distinguish between ethane and ethane.
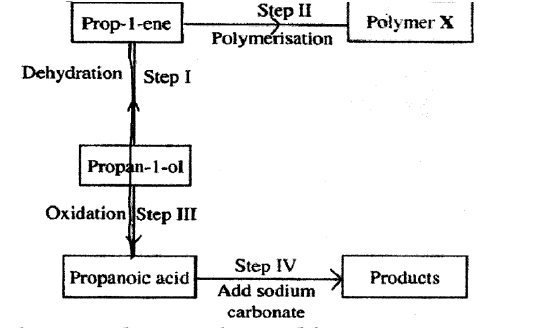
c)The following scheme represents various reactions starting with propan-1-ol. Use it to answer the questions that follow.
i) Name one substance that can be used in step I.
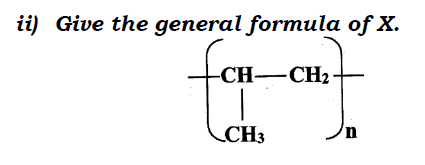
ii) Give the general formula of X.
iii)Write the equation for the reaction in step IV.
iv) Calculate the mass of propan-1-ol which when burnt completely in air at room temperature and pressure would produce 18dm3 of gas. (C = 12.0; O = 16.0; H = 1.0; Molar gas volume = 24dm3)
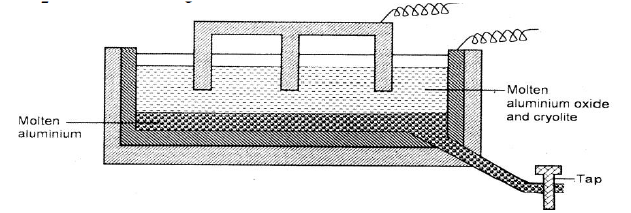
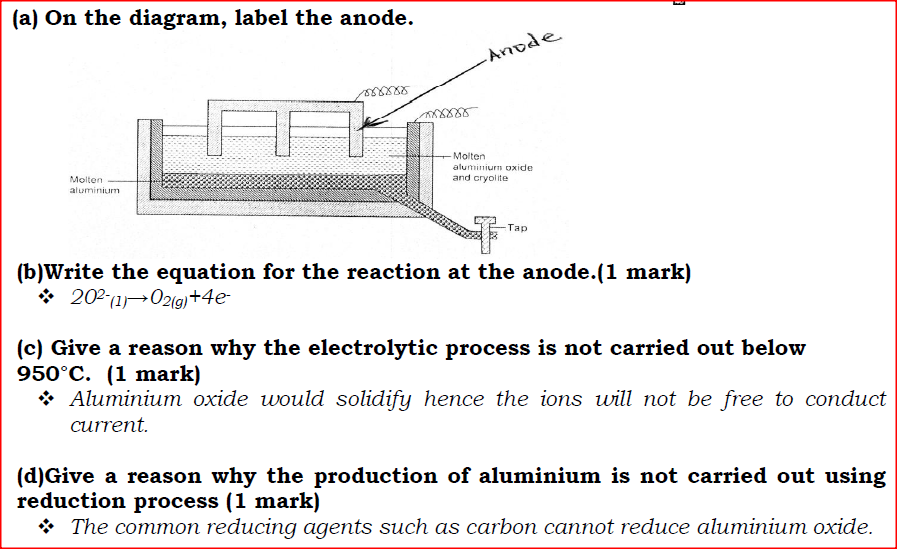
The diagram below represents a set up of an electrolytic cell that can be used in the production of aluminium.
(a) On the diagram, label the anode.
(b)Write the equation for the reaction at the anode. (c) Give a reason why the electrolytic process is not carried out below 950°C. (d)Give a reason why the production of aluminium is not carried out using reduction process (e)Give two reasons why only the aluminium ions are discharged (f)State two properties of duralumin that makes it suitable for use in aircraft industry. (g)Name two environmental effects caused by extraction of aluminium.
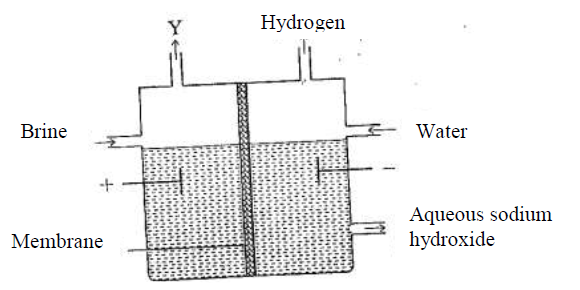
The set up below can be used to produce sodium hydroxide by electolysing brine.
(i) Identify gas Y.
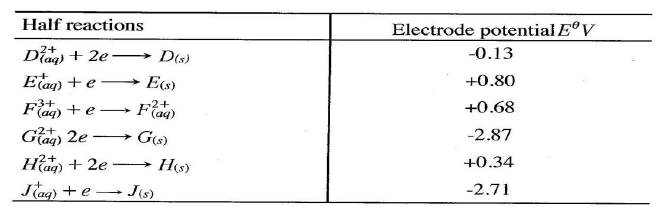
(ii)Describe how aqueous sodium hydroxide is formed in setup above. (iii)One of the uses of sodium hydroxide is in manufacture of soaps. State one other use of sodium hydroxide. (b) Study the information given in the table below and answer the questions that follow.
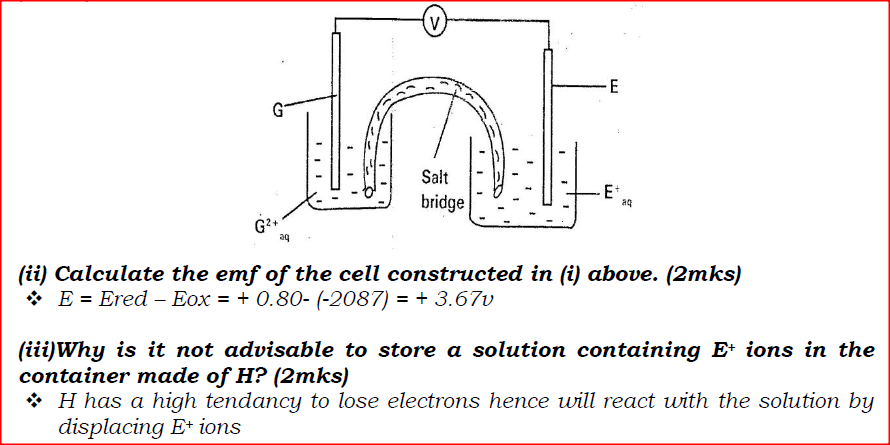
(i) Construct an electrochemical cell that will produce the highest
(ii) Calculate the emf of the cell constructed in (i) above. (iii)Why is it not advisable to store a solution containing E+ ions in the container made of H?
(a) Methanol is manufactured from carbon (IV) oxide and hydrogen gas according to the equation:
The reaction is carried out in the presence of a chromium catalyst at 700K and 30kPa. Under these conditions, equilibrium is reached when 2% of the carbon (IV) oxide is converted to methanol
(i)How does the rate of the forward reaction compare with that of the reverse reaction when 2% of the carbon (IV) oxide is converted to methanol? (ii)Explain how each of the following would affect the yield of methanol: I Reduction in pressure II Using a more efficient catalyst (iii) If the reaction is carried out at 500K and 30kPa, the percentage of carbon (IV) oxide converted to methanol is higher than 2% I what is the sign of ΔH for the reaction? Give a reason II Explain why in practice the reaction is carried out at 700K but NOT at 500K (b)Hydrogen peroxide decomposes according to the following equation:
2H2O2(aq) →2H2O(l) + O2 (g)
In an experiment, the rate of decomposition of hydrogen peroxide was found to be 6.0 x 10-8 mol dm-3 S-1. (i)Calculate the number of moles per dm3 of hydrogen peroxide that had decomposed within the first 2 minutes (ii) In another experiment, the rate of decomposition was found to be 1.8 x 10 - 7 mol dm -3S-1. The difference in the two rates could have been caused by addition of a catalyst. State, giving reasons, one other factor that may have caused the difference in two rates of decomposition
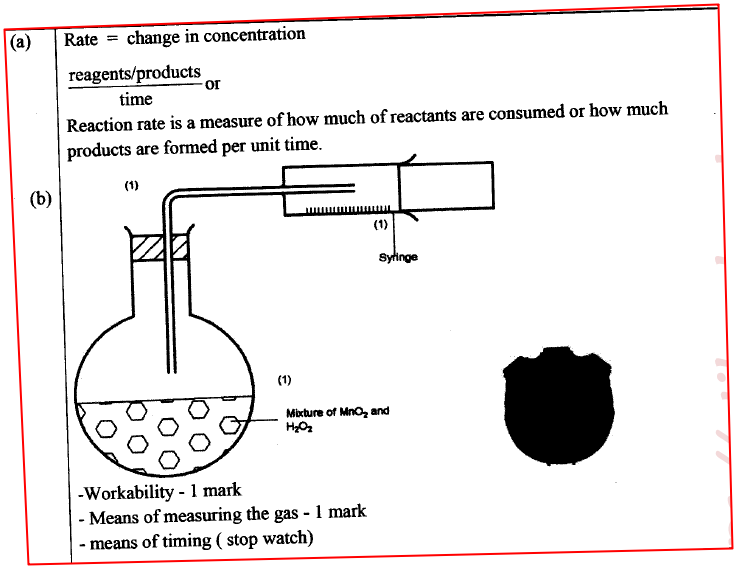
(a) What is meant by rate of reaction. (1 mark)
(b) In the space provided, sketch the diagram of a set-up that can be used to determine the rate of reaction between manganese(IV) oxide and hydrogen peroxide. (3 marks)
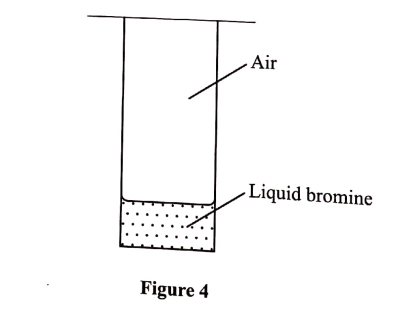
(e) A student placed a small amount of liquid bromine at the bottom of a sealed gas jar of air as shown in Figure 4.
(i) Describe what will be observed: (1 mark)
I. after two minutes . II. after 30 minutes
(ii) Use the Kinetic theory to explain the observations: (2 marks)
I. after 2 minutes . II. after 30 minutes
(d) Some plants have seeds that contain vegetable oil.
(a) What is meant by standard electrode potential of a an element? (1 mark)
(b) use the standard electrode potentials given below to answer the questions that follow.
(i) State whether acidified MnO4- can oxidise M2+. Give a reason. (2 marks) (ii) Select two half-cells which when combined will give the highest e.m.f. (1 mark) (iii) Write the cell representation for the cell formed in b (ii). (I mark) (iv) Calculate the E0 value for the cell formed in b (iii). (2 marks) (c) A mass of 1.24g of a divalent metal was deposited when a current of 6A was passed through a solution of the metal sulphate for 12 minutes. Determine the relative atomic mass of the metal.
(1 Faraday = 96,500 C mol-1 (3 marks)
(d) State two applications of electrolysis. (I mark)
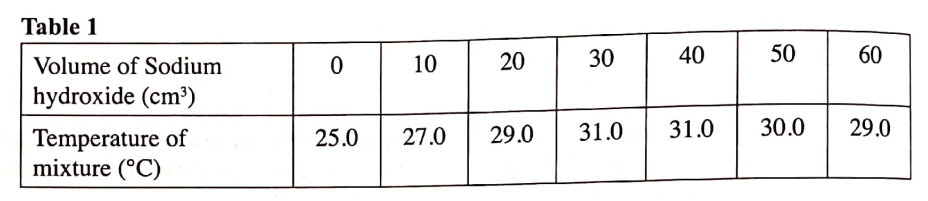
(a) What ¡s meant by molar heat of neutralisation? (1 mark)
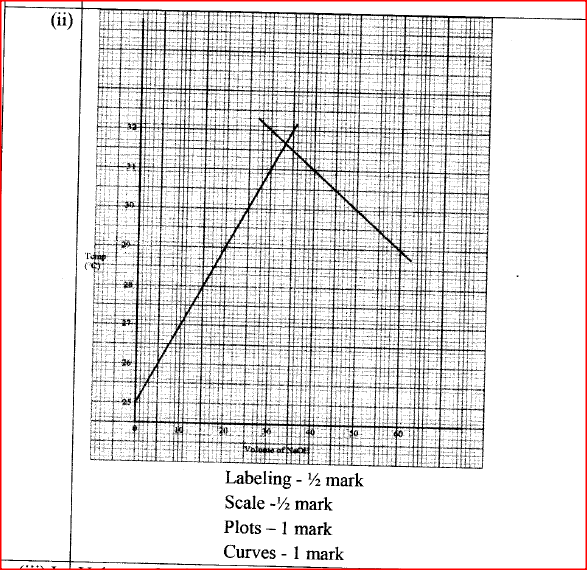
(b) In an experiment to determine the molar heat of neutralisation, 50 cm3 of 1M hydrochloric acid was neutralised by adding 10 cm3 portions of dilute sodium hydroxide. During the experiment, the data in Table 1 was obtained.
(iii) Determine from the graph the: I. volume of sodium hydroxide which completely neutralises 50 cm3 of 1M hydrochloric acid. (1 mark) II. change in temperature,△T, when complete neutralisation occurred. (1 mark) (iv) Calculate: I. the heat change, △H when complete neutralisation occurred. (Specific heat capacity = 4.2 Jg-1K-1, density of solution 1.0 gcm-3) (2 marks) II. molar heat of neutralisation of hydrochloric acid with sodium hydroxide. (1 mark) (v) How would the value of molar heat differ if 50 cm3 of 1M ethanoic acid was used instead of 1M hydrochloric acid? Give a reason. (2 marks)
Describe how a solid sample of potassium sulphate can be prepared starting with 200cm3of 2M potassium hydroxide.
In an experiment on rates of reaction, potassium carbonate was reacted with dilute sulphuric (VI) acid.
(a) What would be the effect of an increase in the concentration of the acid on the rate of the reaction? (b) Explain why the rate of reaction is found to increase with temperature.
ANSWERS
(a)The rate of reaction increases. This is because when the concentration is high: the number of collisions between particles is also high hence reacts faster,
(b)Increase in temperature results in increase in the kinetic energy of the particles. This makes particles move faster and collide frequently leading to faster rate of reaction.
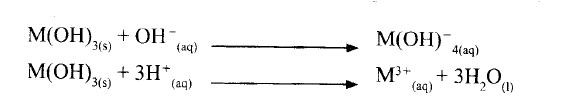
A compound whose general formula is M(OH)3 reacts as shown by the equation below.
(a) What name is given to compounds which behave like M(OH) 3 in the two reactions.
(b) Name two elements whose hydroxides behave like that of M.
ANSWERS
(a) Amphoteric
(b)Lead, Zinc and Aluminium
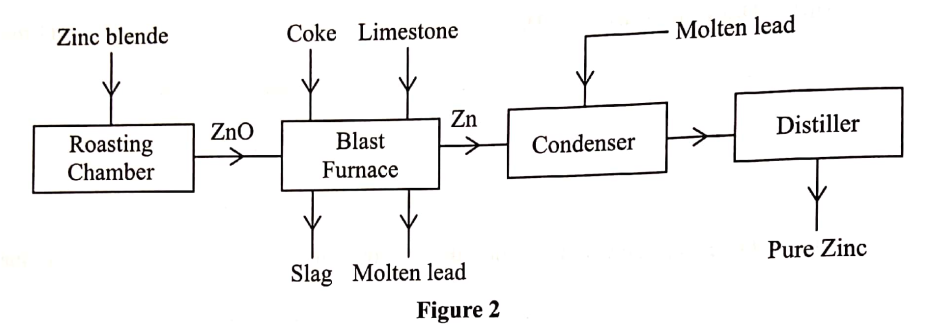
(a) Zinc occurs mainly as zinc blende. Name one other ore from which Zinc can be extracted. (1 mark)
(b) The flow chart in Figure 2 shows the various stages in the extraction of zinc metal. Study it and answer the questions that follow.
(c) Explain the observations made when zinc metal is added to hot sodium hydroxide. (2 marks)
(a) Explain the following observations:
(b) A sample of water is suspected to contain aluminium ions (AI3+) . Describe a laboratory experiment that can be carried out to show that AI3+ ions are present in the water sample. (3 marks) (c) In an experiment to determine the number of moles of water of crystallisation of a hydrated compound, Na2S04. X H2O, 5g of the compound were heated strongly to a constant mass.
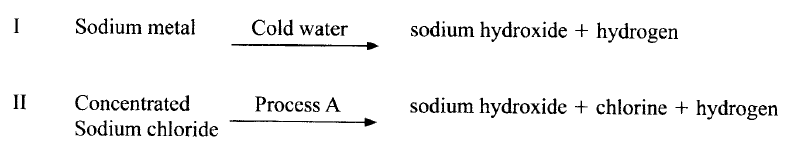
Sodium hydroxide can be prepared by the following methods; I and II
(a) Name one precaution that needs to be taken in method I.
(b) Give the name of process A. (c) Give one use of sodium hydroxide.
ANSWERS
(a) Small piece of sodium metal (pea size) with a lot of water
Perform the experiment wearing goggles. (b) Electrolysis (c) Manufacture of paper (soften), soaps and detergents Fractional distillation of liquid air Extraction of aluminium metal Manufacture of bleaching agents eg NaOCl paper, textiles, oil refinery Making herbicides on weed killers Textile industry to soften
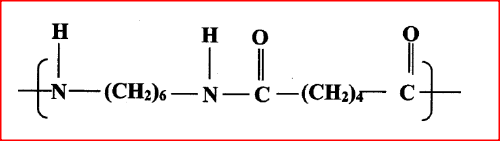
(a)Draw the structure of compound N formed in the following reaction.
(b) Give one use of compound N.
a)Complete the nuclear equation below:
(c)Give one harmful effect of radioisotopes.
Aluminium is both malleable and ductile.
(a)What is meant by? (i) Malleable: (ii)Ductile (b)State One use of aluminium based on: (i)malleability (ii)ductility
ANSWERS
(a) (i) Can be hammered into sheets.
(ii)Can be drawn into wires. (b)(i) Making of sufurias/ motor vehicle parts/ aeroplane parts,window / door flames, cups, plates, packaging materials, pans, making sheets/ roof. (ii)electricity cables/ wires.
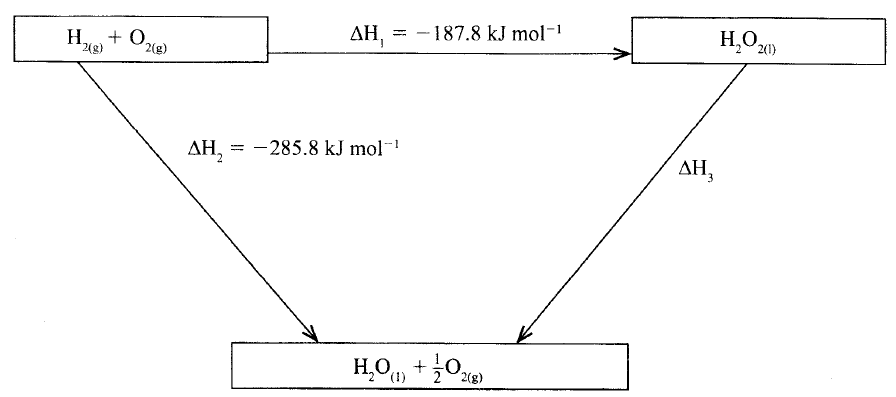
The figure below shows an energy cycle.
(a)Give the name of the enthalpy change ΔH1.
(b)Determine the value of ΔH3.
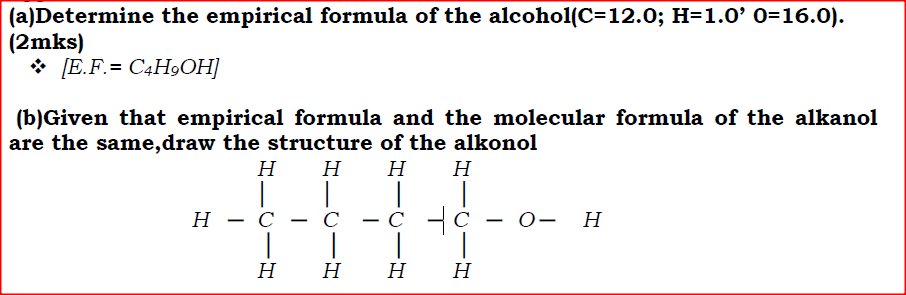
An alknal has the following composition by mass: hydrogen 13.5%, oxygen 21.6% and carbon 64.9%
(a)Determine the empirical formula of the alcohol(C=12.0; H=1.0’ 0=16.0). (b)Given that empirical formula and the molecular formula of the alkanol are the same,draw the structure of the alkonol
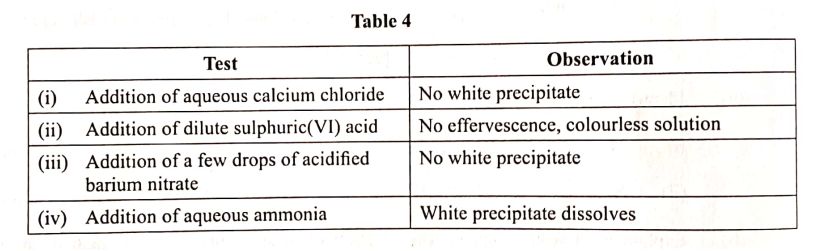
Chemical tests were carried out on separate samples of water drawn from the same source. The observations made were recorded as shown in Table 4.
State the inferences made in reactions:
are isotopes of element X. They occur naturally in the ratio of 9:1 respectively.
Calculate the relative atomic mass of element X. (2 marks) |
Chemistry Topics
All
Archives
December 2024
|
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |



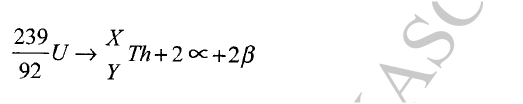




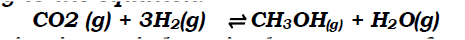



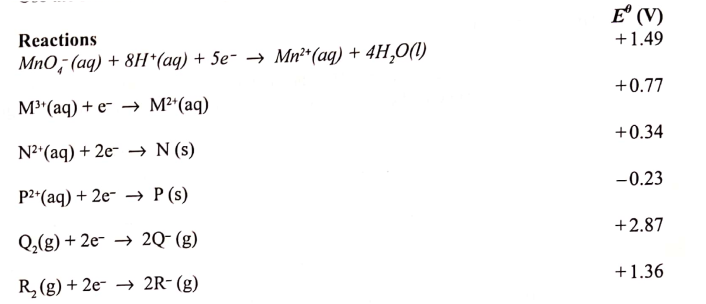










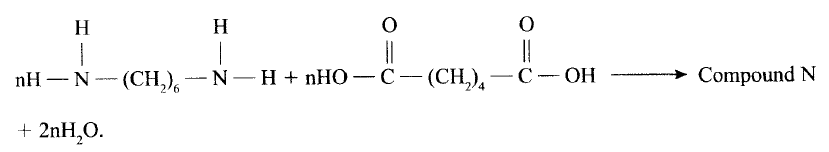

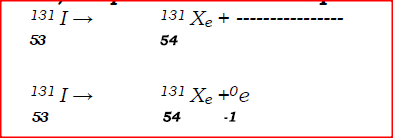
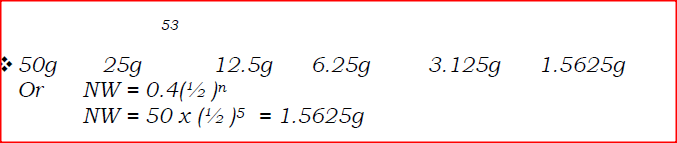
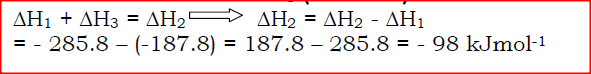

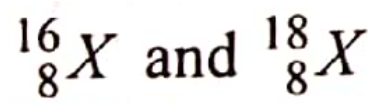


 RSS Feed
RSS Feed

