|
expected response
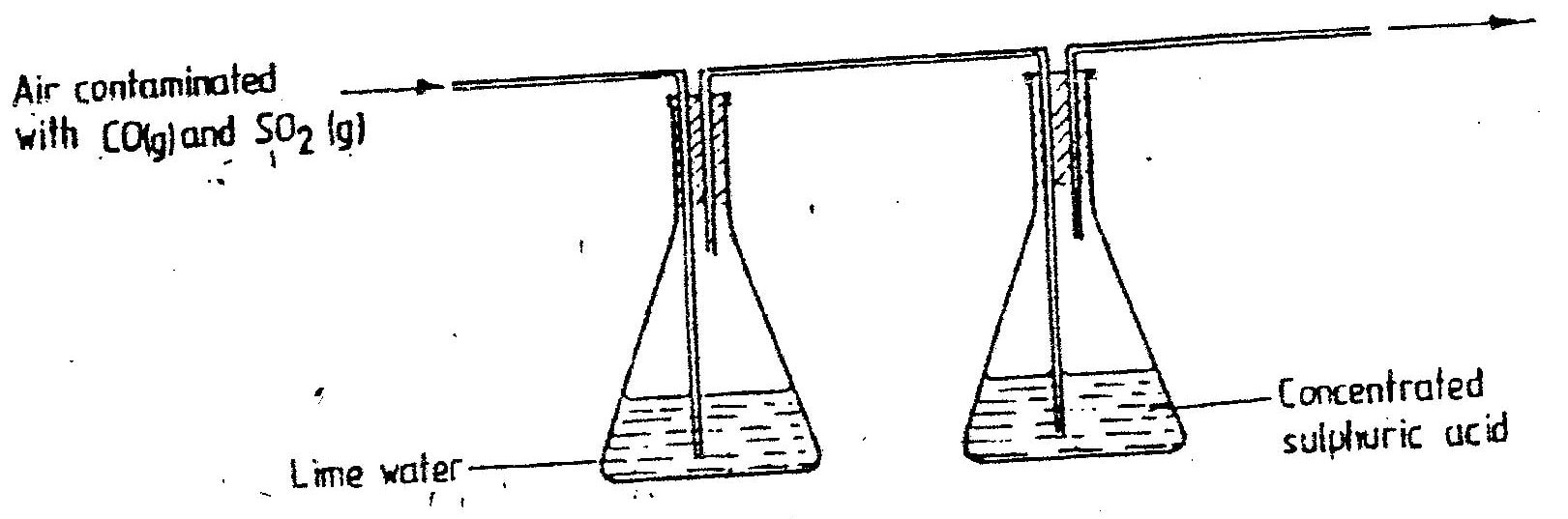
Sulphur dioxide, it reacts with lime-water being an acid gas
0 Comments
expected response
6.
a) G b) E Explain how a sample of CH3CH2CH2OH, could be distinguished from a sample of CH3COOH by means of a chemical reaction (2mks)
expected response
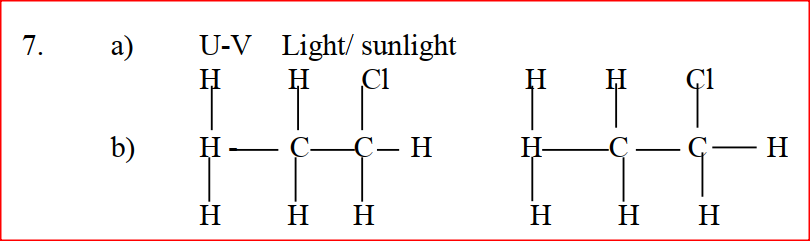
Add solid hydrogen carbonate; CH3COOH produces effervescence; while CH3CH2CH2OH does not (Accept any other carbonate that behaves) The reaction of propane with chlorine gas gave a compound of formula C3H7Cl. a) What condition is necessary for the above reaction to take place? (1mk) b) Draw two structural formulae of the compound C3H7Cl (2mks)
expected response
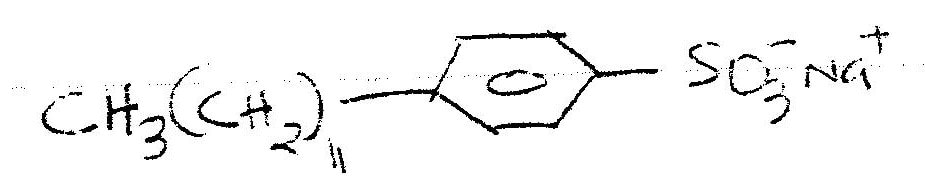
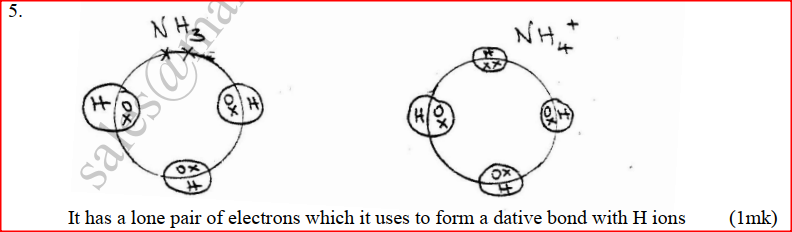
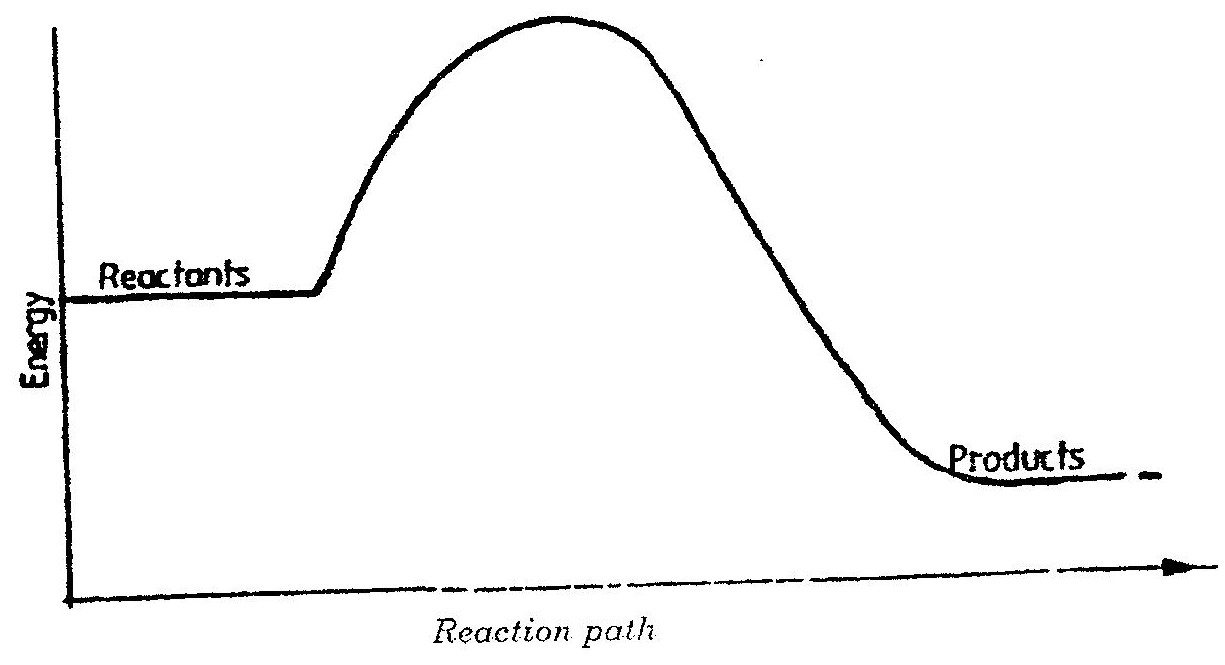
The ionic end lowers the surface tensions of water , facilitating mixing while the nonionic end (non-polar end) mixes with grease, dislodging it from the fabric. a) Using dots(.) and crosses (x) to represent electrons draw diagram to represent the bonding in: (i) NH3 (ii) NH4+ (1mk) b) State why an ammonia molecule (NH3) can combine with H+ to form NH4+ (Atomic numbers: N=7 and H=1) (1mk) Ammonia can be converted to nitrogen monoxide as shown in the equation below (a) Explain how an increase in temperature would affect the yield of nitrogen monoxide (2mks) (b) On the energy level diagram above sketch, the energy level diagram that would be obtained if the reaction is carried out in the presence of platinum catalyst. (1 mk)
expected response
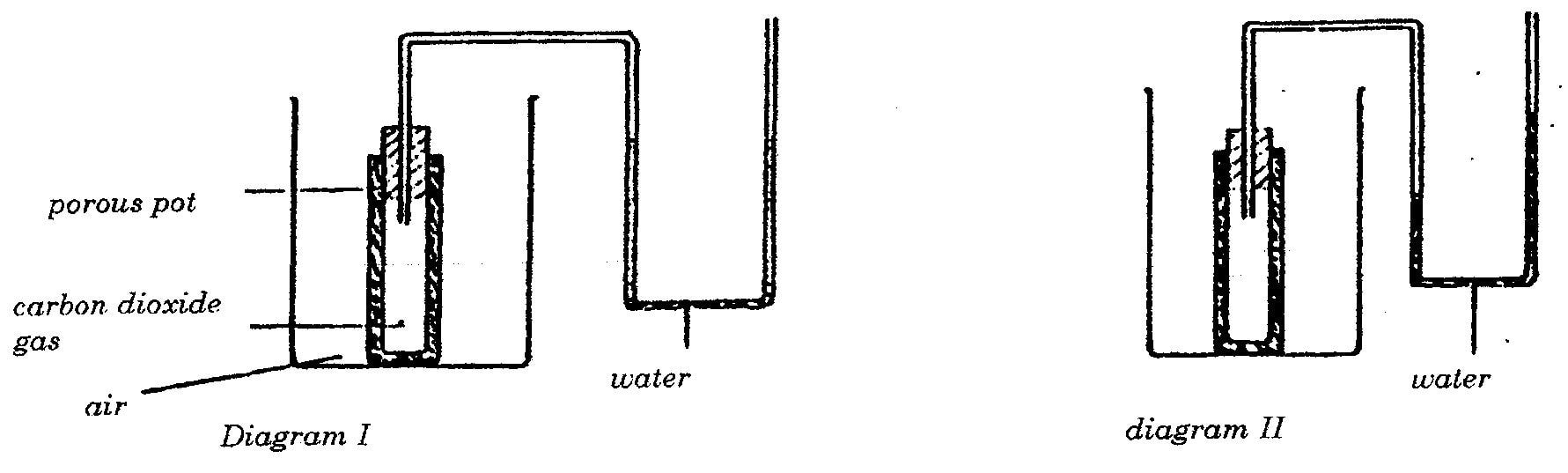
Air is less dense than carbon dioxide and so it enters the porous pot faster than carbon dioxide out of it. This sets up a higher pressure; in the pot and the level rises as shown:
Zinc metal and hydrochloric acid reacts according to the following equation Zn(s) + 2HCI (aq)→ ZnCI2 (aq) + H2 (g) 1.96 g of zinc were reacted with 100cm3of 0.2M hydrochloric acid (a) Determine the reagent that was in excess (b) Calculate the total volume of hydrogen gas was liberated S.T.P (Zn= 65.4 Molar gas volume = 22.4 litres at S.T.P) A fixed mass of a gas has a volume of 250cm3 at a temperature of 270C and 750mm Hg pressure. Calculate the volume the gas would occupy at 420C (2mks) |
Chemistry Topics
All
Archives
December 2024
|
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |




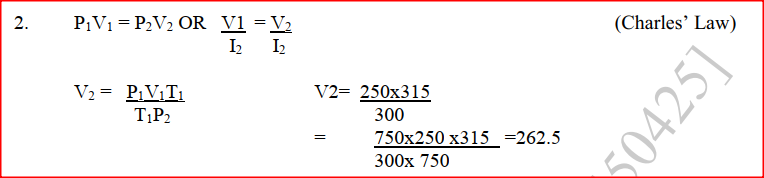

 RSS Feed
RSS Feed

