|
State the observation that would be made when a piece of sodium metal is placed in samples of:
Pentane: __________ Pentanol: __________
0 Comments
The table below shows the solubility of a salt at various temperatures
What would happen if a sample of a saturated solution of the salt at 400C is heated to 800C. Explain
Write the formula of sulphide of an element C, whose atomic number is 5. (C is not the actual symbol of the element)
What type of bond is formed when lithium and Fluorine react? Explain
(Atomic numbers: Li = 3 and F = 9)
Sulphur dioxide and nitrogen dioxide react as shown in the equation below
SO2(g) + NO2(g) → SO3(g) + NO(g)
(i) Using the oxidation numbers of either sulphur or nitrogen, show that this is a redox reaction
(ii) Identify the reducing agent
4.76g of liquid ammonia and 4.76g of liquid nitrogen were each allowed warming up and hanging into gas at warm temperature and pressure. Relative atomic masses: H=1.0 and N=14.0. Using the data given above, explain which gas occupied the greater volume.
State and explain the observation that would be made when a few drops of concentrated sulphuric acid are added to a small sample of hydrated copper (II) sulphate
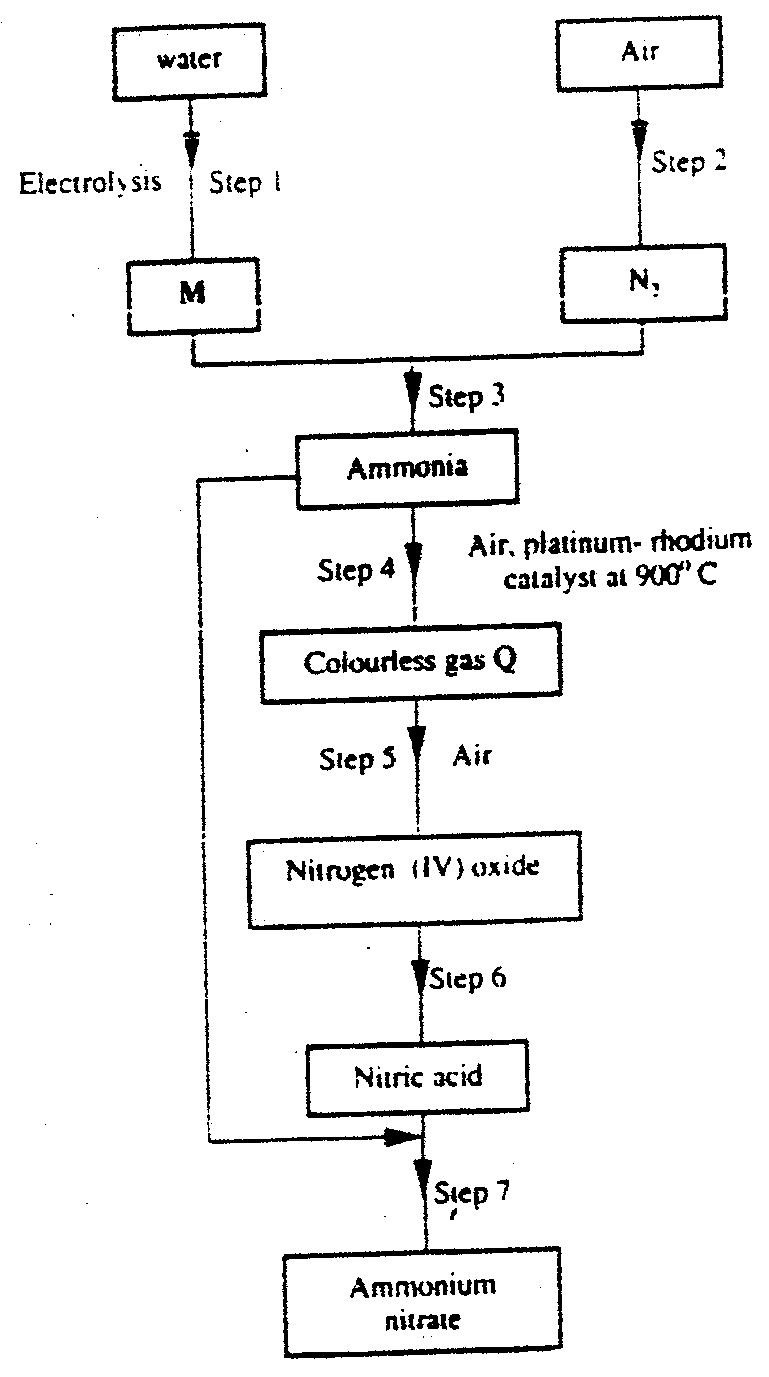
Study the flow chart below and answer the questions that follow
(a) Name:
Aluminium metal is a good conductor and is used for overhead cables. State any other two properties that make aluminium suitable for this use.
|
Chemistry Topics
All
Archives
December 2024
|
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |










 RSS Feed
RSS Feed

