|
A hydrocarbon P was found to decolourise bromine water. On complete combustion of 2 moles of P, 6 moles of carbon dioxide and 6 moles of water were formed
0 Comments
Study the solubility curves below and answer the questions that follows
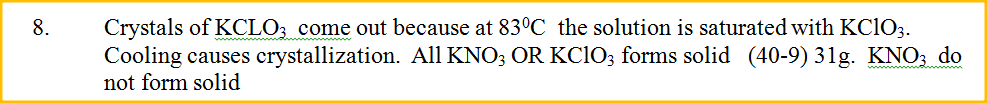
What happens when a solution containing 40gm of potassium chlorate and 40gm of potassium nitrate in 100gm of water at 900C is cooled to 400C? Explain
Give two reasons why helium is used in weather balloons
Expected Response
Starting with copper metal, describe how a solid samples of copper (II) carbonate can be prepared.
The equation below represents a redox reaction
a) Write down the equation for the reduction process
b) Which substance is oxidized
The information below relates to elements S,T,U and X. ( the letters do not represents the actual symbols of the elements.
Both T and X are in group II of the periodic Table) (b) Arrange the elements in order of their increasing reactivity
When a current of 1.5 amperes was passed though a cell containing m3+ ions of a metal M for 15 minutes, the mass of the cathode increased by 0.26g.
(1 Faraday = 96500 coulombs) (a) Calculate the quantity of electricity used (b) Determine the relative atomic mass of metal M
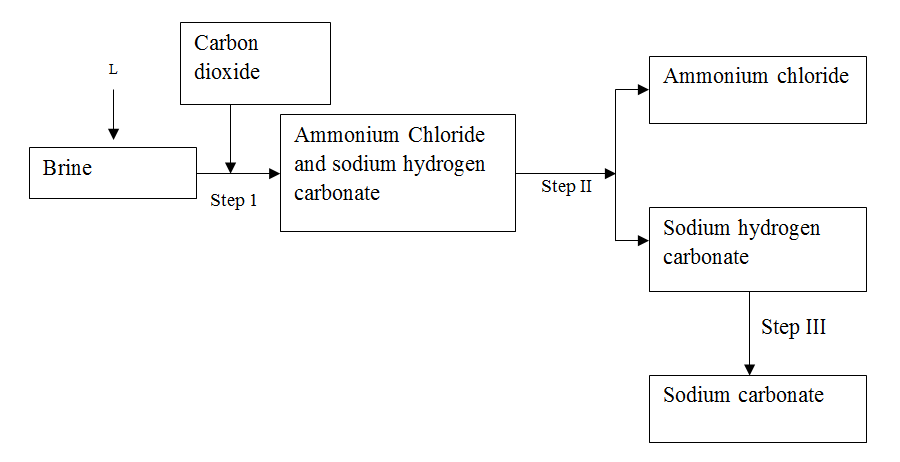
The simplified flow chart shows some of the steps in the manufacture of sodium carbonate by the Solvay process
(a) Identify substance L
(b) Name the process – taking place in step II (c) Write an equation for the reaction, which takes place in step III
Explain the change in mass that occurs when the following substances are separately heated in open crucibles
(a) Copper metal (b) Copper (II) nitrate
Expected Response
a) Mass increases because oxygen combine with copper metal
b) Mass decreases it decomposes into gases that escape. |
Chemistry Topics
All
Archives
December 2024
|
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |




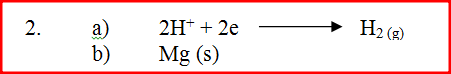




 RSS Feed
RSS Feed

