|
ANSWER
0 Comments
In an experiment, a test-tube full of chlorine water was inverted in chlorine water as shown in the diagram below and the set up left in sunlight for one day.
After one day, a gas was found to have collected in the test-tube
a) Identify the gas. b) What will happen to the PH of the solution in the beaker after one day? Give an explanation.
The table below gives atomic numbers of elements represented b the letters A, B, C and D.
Use the information to answer the questions that follow.
a) Name the type of bonding that exists in the compound formed when A and D react b) Select the letter which represents the best oxidizing agent. Give a reason for your answer.
ANSWERS
(a)Ionic/ electrovalent
(b)Has 7 electrons in its outermost energy level and hence easily gains an electron to complete the octet or it is most electronegative.
When magnesium was burnt in air, a solid mixture was formed. On addition of water to the mixture a gas which turned moist red litmus paper blue was evolved. Explain these observations.
Zinc reacts with both concentrated and dilute sulphuric (VI) acid. Write equations for two reactions.
a) State the observation made at the end of the experiment when a mixture of iron powder and sulphur is heated in a test tube.
b) Write an equation for the reaction the product in (a) above and dilute hydrochloric acid. c) When a mixture of iron powder and sulphur is heated, it glows more brightly than that of iron fillings and sulphur. Explain this observation
ANSWERS
ANSWERS
Phosphoric acid is manufactured from calcium phosphate according to the following equation.
Ca3(PO4)2(s) + 3H2SO4(l) → 2H3PO4(aq) + 3 CaSO4(s) Calculate the mass in (Kg) of phosphoric acid that would be obtained if 155 Kg of calcium phosphate reacted completely with the acid (Ca=40, P=31, S=32, O=16, H=1)
The structure of a detergent is
a) Write the molecular formula of the detergent.
b) What type of detergent is represented by the formula? c) When this type of detergent is used to wash linen in hard water, spots (marks) are left on the linen. Write the formula of the substance responsible for the spots
When a hydrated sample of calcium sulphate CaSO4 .XH2O was heated until all the water was lost, the following data recorded;
Mass of crucible = 30.296 g Mass of crucible +hydrated salt = 33.111 g Mass of crucible + anhydrous salt = 32.781 g Determine the empirical formula of the hydrated salt (Relative formula mass of CaSO4 =136, H2O =18).
A small crystal of potassium manganate (VII) was placed in a beaker water. The beaker was left standing for two days without shaking. State and explain the observations that were made.
ANSWERS
|
Chemistry Topics
All
Archives
December 2024
|
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |



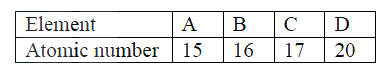

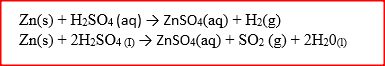
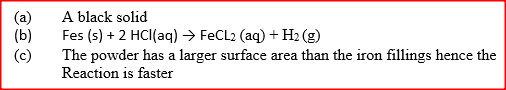









 RSS Feed
RSS Feed

