|
Dilute sulphuric (VI) acid was electrolysed using platinum electrodes.
Name the product formed at the anode and give a reason for your answer.
0 Comments
The atomic number of an element, T is 15.
(a) Write the electronic configuration of the ion T (b) Write the formula of an oxide of T.
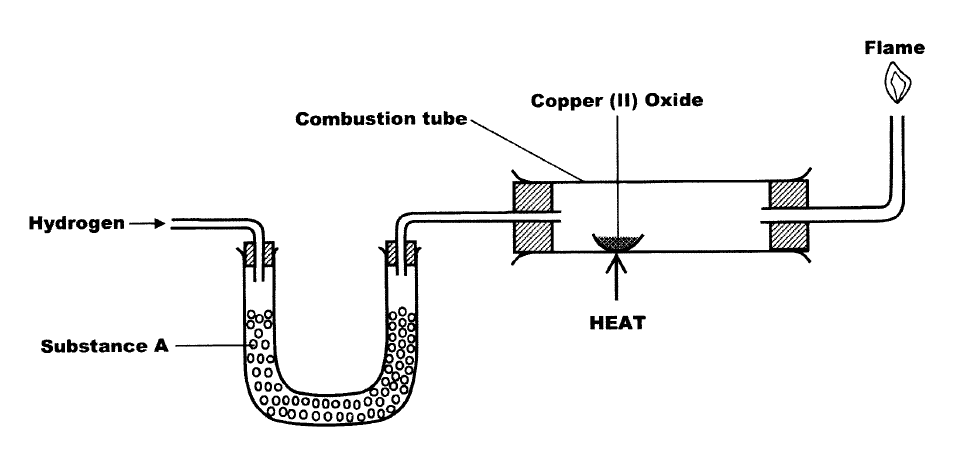
The set up below was used to investigate the reaction between dry hydrogen gas and copper (II) oxide
a) Name substance A
b) state the observation made in the combustion tube c) Explain the observation stated in (b) above.
ANSWERS
(a) Substance A - Calcium Oxide- fused calcium chloride
- Accept any other answer (b) Black Copper (II) Oxide (Solid) changes to brown Colourless liquid formed on the cooler part of the combustion tube. (c) Copper (II) Oxide is reduced to Copper metal.
When solid A was heated strongly, it gave off water and a solid residue. When water was added to the solid residue, the original solid A, was formed
(a) What name is given to the process described? (b) Give one example of solid A
ANSWERS
(a)Type of reaction: Reversible reaction/temporary reaction.
(b)Copper (II) Sulphate salt (Crystals) Copper (II) Chloride hydrated. Any other hydrated salts e.g. Cobalt (II) Chloride
Hydrogen chloride gas can be prepared by reacting sodium chloride with an acid.
(a) Write an equation for the reaction between sodium chloride and the acid. (b) Give two chemical properties of hydrogen chloride gas (c) State two uses of hydrogen chloride
Describe how sodium carbonate is used to remove water hardness
Calculate the mass of Zinc oxide that will just neutralize dilute nitric (V) acid containing 12.6 g of nitric (V) acid in water. (Zn = 65.0; O =16.0, H = 1.0, N = 14.0).
a) State the Boyles Law
b) A gas occupies 500cm3 at 27°C and 100,000 Pa, What will be its volume at O°C and 101325 Pa?
(a) What is meant by lattice energy?
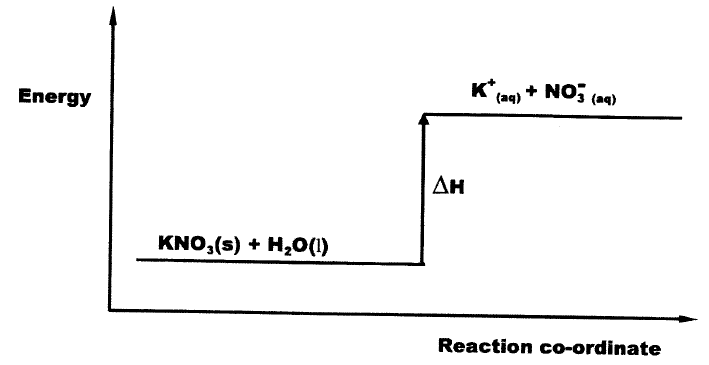
b) Study the energy level diagram below and answer the question that follows
What type of reaction is represented by the diagram?
ANSWERS
(a) Enthalpy change, when one mole of crystal lattice is broken into its ions in gaseous state.
(b) Endothermic reaction (process)
(a) Name the raw material from which sodium is extracted.
(b) Give a reason why sodium is extracted using electrolysis. (c) Give two uses of sodium metal.
ANSWERS
(a)Brine (NaCl)
(b) Sodium is very reactive (use electrolysis) More reactive than carbon. (c) Uses Sodium lamps, coolant in nuclear reactors Sodium cyanide, sodium amalgam Na202 , Extraction of titanium,
(a) Give the name of the first member of the alkene homologous series.
(b) Describe a chemical test that can be used to distinguish butanol from butanoic acid |
Chemistry Topics
All
Archives
December 2024
|
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |







 RSS Feed
RSS Feed

