|
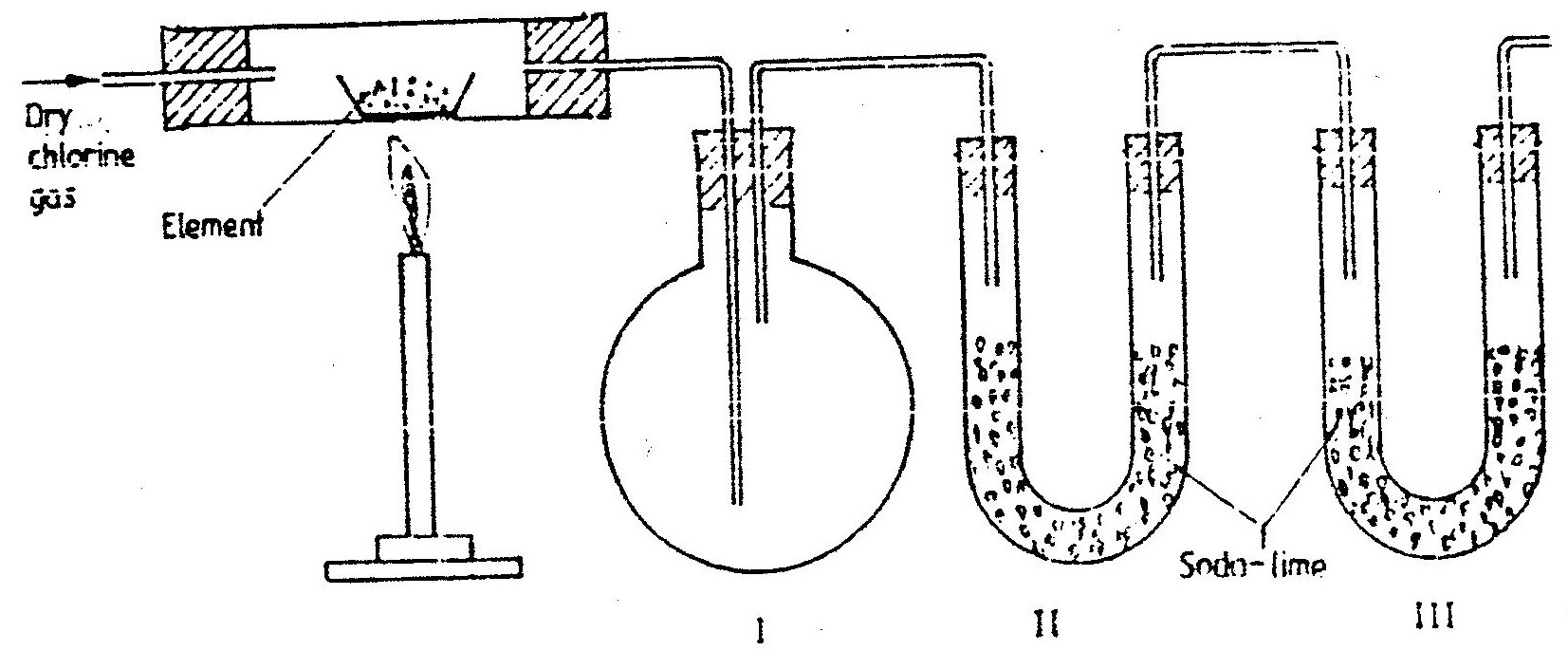
The set – up below was used to prepare anhydrous chlorides of a number of elements in a laboratory where no fine cupboard was available. The chlorides were to be collected in flask
The following table shows the melting and boiling points of the chlorides that were prepared.
More Quiz
(a) The table below gives the solubilities of hydrated copper (II) sulphate in mol dm3 at different temperatures (i) On the grid provided, plot a graph of solubility of copper (II) sulphate (vertical axis) against temperature. (ii) From the graph, determine the mass of copper (II) sulphate deposited when solution is cooled from 700C to 400C. (Molar mass of hydrated copper (II) sulphate = 250g) (b) In an experiment to determine the solubility of sodium chloride, 5.0cm3 of a saturate solution of sodium chloride weighing 5.35g were placed volumetric and diluted to a total volume of 250cm3. 25.0cm3 of the dilute solution chloride completely reacted with 24cm3 of 0.1M silver nitrate solution. AgNO3(aq) + NaCI(aq) → AgCl(s) + NaNO3(aq) Calculate: (i) Moles of silver nitrate in 24cm3 of solution (ii) Moles of sodium chloride in 25.0cm3 of sodium (iii) Moles of sodium chloride in 250cm3 of solution (iv) Mass of sodium chloride in 5.0cm3 of saturated sodium chloride Solution (Na = 23.0, CI = 35.5) (v) Mass of water in 5.0cm3 of saturated solution of sodium chloride (vi) The solubility of sodium chloride in g/100 water
0 Comments
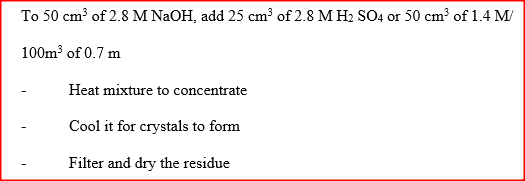
Starting with 50 cm3 of 2.8M sodium hydroxide, describe how a sample of pure sodium sulphate crystals can be prepared.
Describe how a solid sample of lead (II) Chloride can be prepared using the following reagents, dilute nitric acid, dilute hydrochloric acid and lead carbonate.
Expected Response
a) Write an equation to show the effect of heat on the nitrate of:
(i) Potassium b) The table below gives information about elements A1,A2,A3, and A4
(i) In which period of the periodic table is element A2? Give a reason
(ii) Explain why the atomic radius of: I A1 is greater than that of A2; II A4 is smaller than its ionic radius (iii) Select the element which s in the same group as A3 (iv) Using dots (.) and crosses(x) to represent outermost electrons. Draw a diagram to show the bonding in the compound formed when A1 reacts with A4
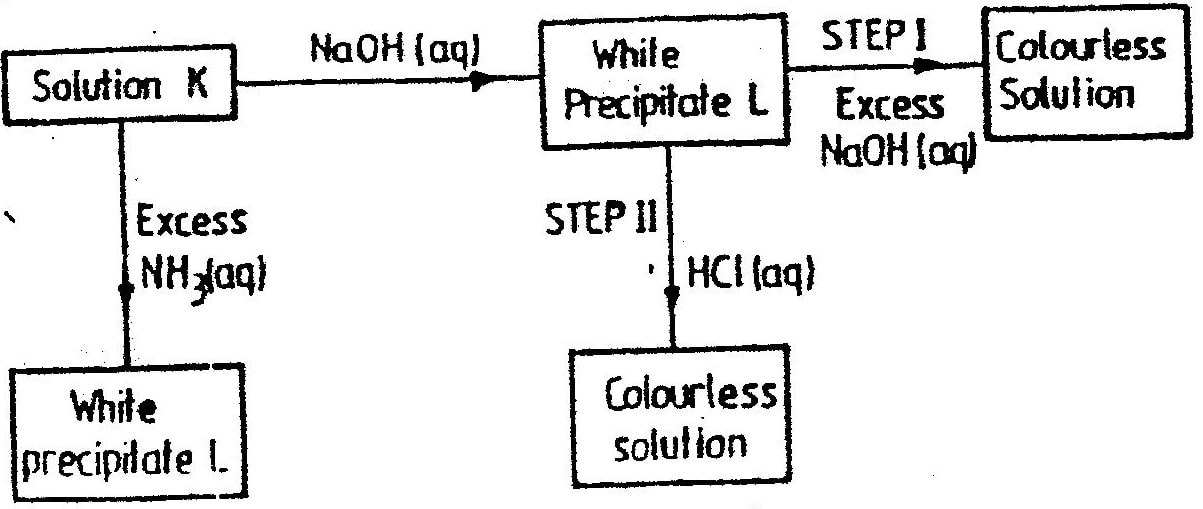
When solid B1 was heated, a gas which formed a white precipitate when passed through lime water was produced. The residue was dissolved in dilute nitric (V) acid to form a colourless solution B2. when dilute hydrochloric acid was added to solution B2 a white precipitate which dissolved on warning was formed.
a) Write the formula of the; I Cation in solid B1 II anion in solid B1 b) Write an ionic equation for the reaction between the resdue and dilute nitric (V) acid.
Starting with copper metal, describe how a sample of crystals of copper (II) chloride may be prepared in the laboratory.
Starting with sodium metal, describe how a sample of crystals of sodium hydrogen carbonate may be prepared.
ANSWERS
ANSWERS
a)Dilute Nitric acid
b)Silver metal c)oxygen In an equation below, identify the reagent that acts as abase. Give a reason. H2O(aq) + H2O(l) → H3O(aq) + HO2-(aq)
Potassium sulphite solution was prepared and divided into two portions. The first portion gave a white precipitate when reacted with barium nitrate. On addition of dilute hydrochloric acid the white precipitate disappeared.
a) Write the formula of the compound which formed as the white precipitate. b) Write the equation for the reaction between dilute hydrochloric acid and the compound whose formula is written in (a) above. c) What observation would be made if one drop of potassium dichromate solution was added to the second portion followed by dilute hydrochloric acid?
Explain why anhydrous magnesium chloride is fairly soluble in organic solvents while anhydrous magnesium chloride is insoluble.
Expected Response
Aluminum chloride is covalent while magnesium chloride is ionic
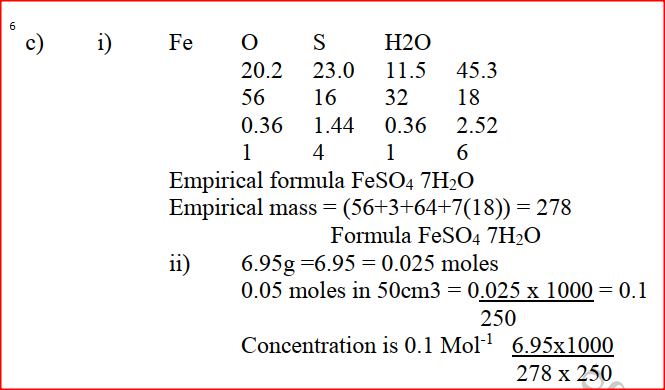
a) Give the name of each of the processes described below which takes place when salts are exposed to air for sometime. i) Anhydrous copper sulphate becomes wet (1mk) ii) Magnesium chloride forms an aqueous solution (1mk) iii) Fresh crystals of sodium carbonate, Na2CO3. 10H2O (1mk) b) Write the formula of the complex ion formed in each of the reactions described below. (i) Zinc metal dissolves in hot alkaline solution (1mk) (ii) Copper hydroxide dissolves in excess ammonia solution. (1 mk) (c) A hydrated salt has the following composition by mass. Iron 20.2% Oxygen 23.0%, sulphur 11.5%, water 45.3 %. Its relative formula mass is 278. (i) Determine the formula of the hydrated salt.. (3mks) (Fe=56, S=32; O = 16, H =1) (ii) 6.95gm of the hydrates salt were dissolved in distilled water and the total volume made to 250 cm3 of solution. Calculate the concentration of the salt solution in moles per litre.
Carbon dioxide can be dissolved in water under pressure to make an acidic solution.
a) What is meant by an acidic solutions? b) aqueous lead (II) nitrate reacts with the acidic solution to form a precipitate. Write anionic equation for the reaction. Explain how you would obtain solid carbonate from a mixture of lead carbonate and sodium carbonate powders. (3mks)
Expected Response
Dissolve in water, filter to remove lead carbonate as a residue, evaporate filter to saturation and allow to cool. Crystallization to take place. Filter the crystals and dry. Evaporate to dryness
Some sodium chloride was found to be contaminated with copper (ll) oxide.
Describe how a sample of sodium chloride can be separated from the mixture
ANSWER
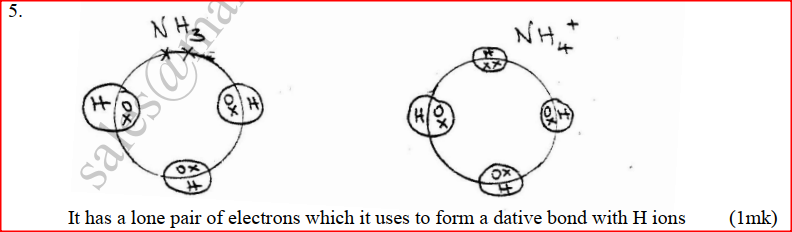
a) Using dots(.) and crosses (x) to represent electrons draw diagram to represent the bonding in: (i) NH3 (ii) NH4+ (1mk) b) State why an ammonia molecule (NH3) can combine with H+ to form NH4+ (Atomic numbers: N=7 and H=1) (1mk)
Describe how a solid sample of Zin (II) carbonate can be prepared starting with zinc oxide
When potassium nitrate is heated, it produces potassium nitrate and gas C
(a) Identify gas C (b) Name the type of reaction undergone by the potassium nitrate
answer
|
Chemistry Topics
All
Archives
December 2024
|
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |




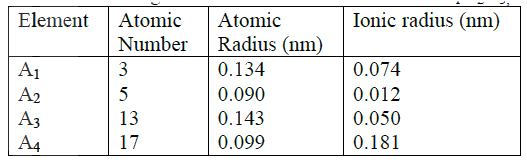

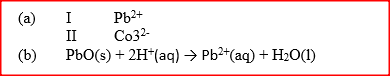









 RSS Feed
RSS Feed

