|
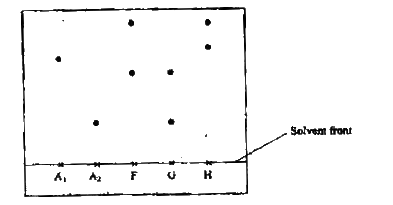
Samples of urine from three participants F, G and H at an international sports meeting were spotted onto a chromatography paper alongside two from illegal drugs A1 and A2.
A chromatogram was run using methanol. The figure below shows the chromatogram.
a) Identify the athlete who had used an illegal drug.
b) Which drug is more soluble in methanol?
ANSWERS
(a)A1
(b)A1 using baseline
0 Comments
ANSWERS
a)They gain energy and move faster. The intermolecular distance increases.
b)XY c)The energy supplied changes molecules of water from liquid to Gaseous state.
ANSWERS
a)V1 and V3
b)Add petrol to the mixture. Filter. V2 is the residue. Filtrate is V4 Distill the filtrate.
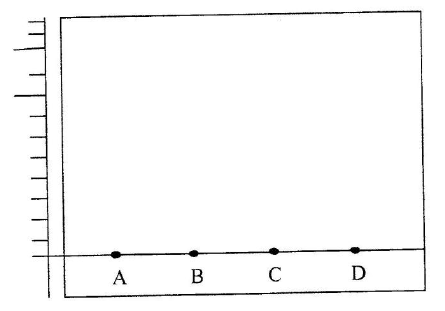
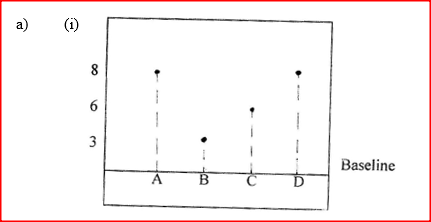
(a) The diagram below shows spots of pure substance A,B, and C on a chromatography paper. Spot D is that of a mixture
After development, A, B and C were found to have moved 8cm, 3cm and 6 cm respectively. D has separated into two spots which had moved 6cm and 8 cm
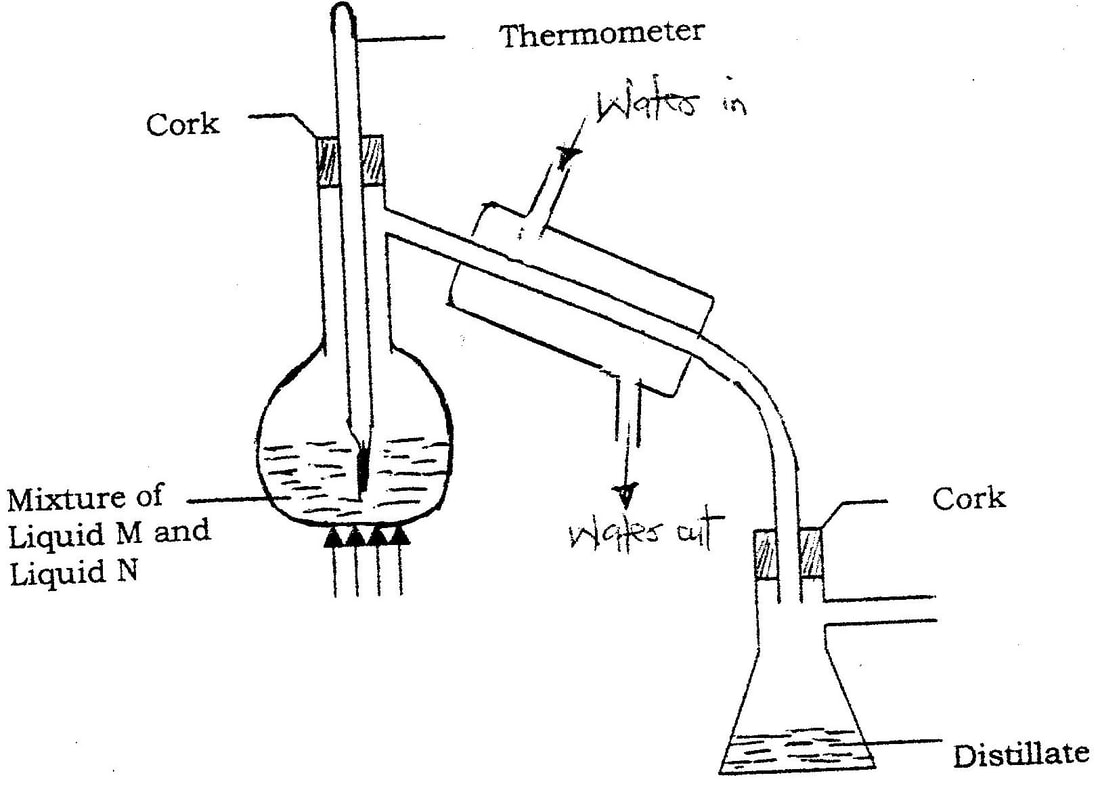
(i) On the diagram I Label the baseline ( origin) II Show the positions of all the spots after development (ii) Identify the substances present in the mixture D (b) Describe how solid ammonium chloride can be separated fro a solid mixture of ammonium chloride and anhydrous calcium chloride (c) The table shows liquids that are miscible and those that are immiscible
Use the information given to answer the questions that follow
(i) Name the method that can be used to separate L1 and L3 from a mixture of two (ii) Describe how a mixture of L2 and L4 can be separated
ANSWERS
(ii) A and C b)Since NH4CL4 sublimes but CaCl2 does not; sublimation process would do. Heat the mixture. Ammonium chloride sublimates into vapour and condenses on the cooler part of the heating tube. Calcium chloride will remain on the bottom of the heating tube. c)i)Fractional distillation ii)Separating funnel method Since the tow liquids are immiscible, pour both the liquids in a separating funnel and allow to settle, the denser liquid will settle down and the less dense will form a second layer on top. Open the tape and run out the liquid in the bottom layer leaving the liquid in the second layer in the funnel.
Apiece of chromatography paper was spotted with coloured inks obtained from pens labeled 1 to 6. The diagram below shows the spots after the chromatogram was developed.
a) Which two pens contained in the same pigment?
b) Which pens contained only one pigment c) According to the chromatogram, which pigments are present in the ink of pen number 6.
ANSWERS
(a) 4 and 5 blue and Green (full) H2SO4 (aq) is on electrolyte
(b)2 and 3 yellow and red (c)Yellow and red 4 – Blue 5 – Green 2 – Yellow 3 – Red award it the colour is tied to the number
answers
Expected Response
W because its solubility decreases with increase in temperature
Explain how you would separate mixture of nitrogen and oxygen gases given that their boiling points are –1960C and 1830C respectively (2mks)
Answer
Cool the mixture to a temperature below – 1960C to form a liquid then start warming, Nitrogen distils off a gas at – 1960 (cool first)
KCSE PAST PAPERS - CHEMISTRY FORM 1 TOPICAL QUESTIONS AND ANSWERSQUESTIONS
|
Chemistry Topics
All
Archives
December 2024
|
||||||||||||||||||||||||||||||
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |


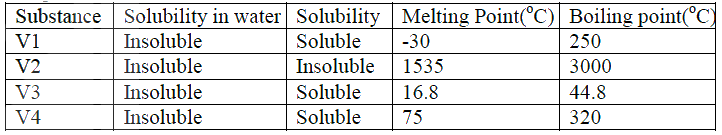
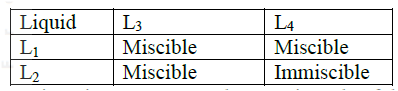





 RSS Feed
RSS Feed

