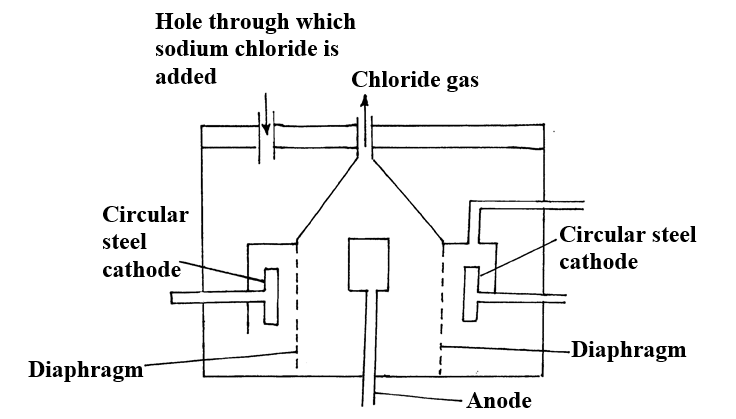
a) Name the substance the anode is made of (1mk)
Graphite
b) Explain your answer in (a) above (1mk)
Reject carbon
Graphite does not react with chlorine liberated at the anode
c) What is the role of the diaphragm in Down’s cell (1mk)
To prevent metal and chlorine gas from recombining
d) In Down’s cell for the manufacture of Sodium metal, Calcium chloride salt is added to lower the melting point from 8000C to 6000C. Explain why it is necessary to lower the melting point (1mk)
To reduce the cost of energy used to melt the sodium metal
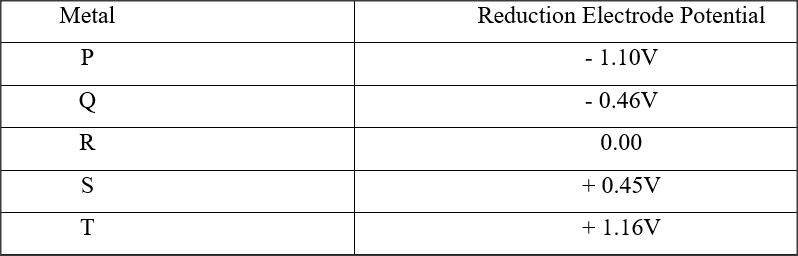
e) Calculate the mass of sodium metal produced if a current of 50 amperes is passed through the molten Sodium chloride for two (2) hours (Na = 23, F = 96500C) (2mks)f) Below is a list of potential differences obtained when metal P, Q, R, S and T are used in the following electrochemical cell
|
Chemistry Topics
All
Archives
December 2024
|
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |
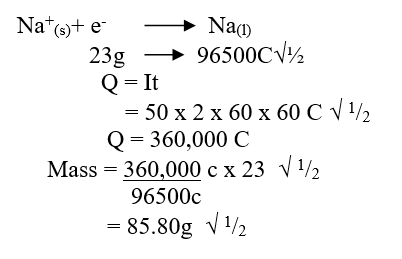

 RSS Feed
RSS Feed

