|
The diagram below shows a set up for the laboratory preparation and collection of dry chlorine gas
Expected Response
Molar heats of vaporization increase down the group with increase in van der waals forces of attraction as the number of electrons increase/increases in size/ volume/mass/energy levels/ density/ No. of protons (atomic number)
0 Comments
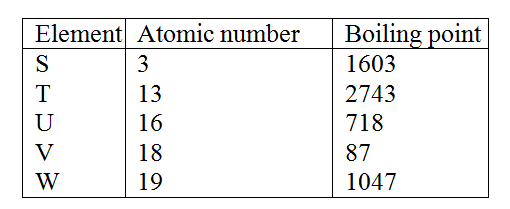
Use the information in the table below to answer the questions that follow. The letters do not represent that actual symbols of the elements.
a) Give the reasons why the meling point of:
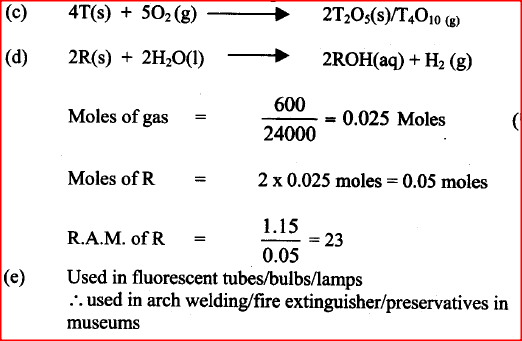
i) S is higher than that of R ii) V is lower than that of U b) How does the reactivity of W with chlorine compare with that of R with chlorine? Explain, c) Write an equation for the reaction between T and excess oxygen
d) When 1.15g of R were reacted with water, 600cm3 of gass was produced.
Determine the relative atomic mass of R. (Molar gas volume = 24000cm3) e) Give one use of element V
ANSWERS
(a)(i) Metallic bonds in S are stronger than in R.
(ii) V is monoatomic (independent) hence weaker van der waals forces while U is diatomic hence stronger van der waals forces V has less van der waals forces while U has more van der waal forces (b) reacts more vigorously/more reactive/reacts faster - reactivity of group 1 elements increases down the group/Ionisation energy is less than that of R - easier to lose outer electron in W than in R W is more electropositive than R
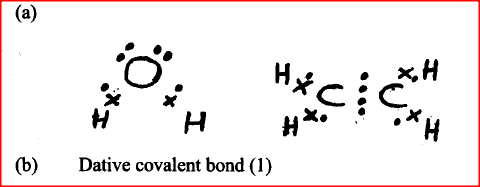
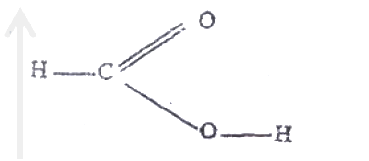
Using electrons in thee outermost energy level, draw the dot (.) and cross (x) diagrams for the molecules H2O and C2H4. (H = 1, C = 6, O = 8)
i) H2O ii) C2H4 b) The formula of a complex ion is Zn(NH3)42+. Name the type of bond that is likely to exist between zinc and ammonia in the complex ion.
What type of bond is formed when lithium and Fluorine react? Explain
(Atomic numbers: Li = 3 and F = 9)
Study the information given in the table below and answer the questions that follow. The letters do not represents the actual symbols of the elements
(a) Select the elements which belong to the same
(e) The aqueous sulphate of element W was electrolyzed using inert electrodes Name the products formed at the:
ANSWERS
(a) SiH4 it has a higher boiling point
(b) No hydrogen bonding in CH4 and SiH4 while the hydrogen bond in H2O is stronger than that in H2S
ANSWERS
Using dots (.) and crosses (x) , show bonding in:
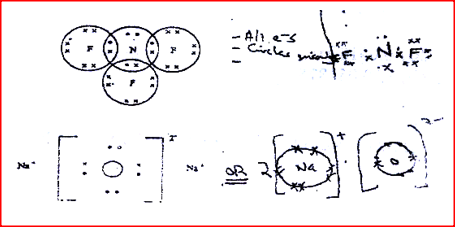
a) The compound formed when nitrogen reacts with fluorine (Atomic numbers F=9, N=7); b) Sodium oxide.(Atomic numbers Na= 11, 0 = 8)
An isotope of element E has 34 neutrons and its mass number is 64. E forms a cation with 28 electrons. Write the formula of the cation with 28 electrons. Write the formula of the cation indicating the mass and atomic numbers.
In terms of structure and bonding, explain why the melting point of oxygen is much lower than that of sodium.
ANSWER
ANSWERS
(i) 2.8.8 (ii) 2.8.2
Diamond and graphite are allotropes of carbon. In terms of structure and bonding explain the following.
The table below gives atomic numbers of elements represented b the letters A, B, C and D.
Use the information to answer the questions that follow.
a) Name the type of bonding that exists in the compound formed when A and D react b) Select the letter which represents the best oxidizing agent. Give a reason for your answer.
ANSWERS
(a)Ionic/ electrovalent
(b)Has 7 electrons in its outermost energy level and hence easily gains an electron to complete the octet or it is most electronegative.
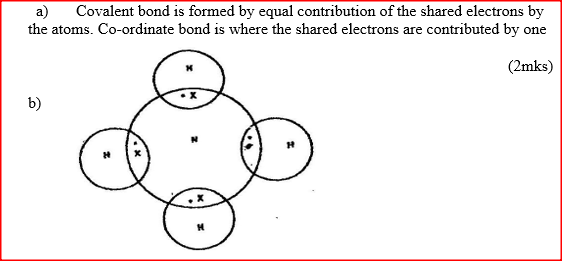
a) Distinguish between a covalent bond and a co-ordinate bond
b) Draw a diagram to show bonding in an ammonium ion. Using dots (.)and crosses (x) to represent outermost electrons, draw diagrams to show the bonding in CO2 and H3O+ (Atomic numbers; H = 1.0,C= 14.0, O = 8 ).
(a) An atom Q can be represented as
What does the number 52 represent?
(b) Study the information in the table below and answer the equations that follow (Letters are not the actual symbols of the elements)
(i) Write the formula of the compound formed when N reacts with P. (atomic numbers are N = 20; P = 17)
(ii) Identify the elements which belong to the third period of the periodic table. Explain (iii) Which of the element identified in b (ii) above comes first in the third period? Explain (iv) Select two elements which are non- metals (c) The table below gives some properties of substances I, II, III, and IV. Study it and answer the questions that follow
(i) What type of bonding exists in substances I and II
I II (ii) Which substances is likely to be sulphur? Explain
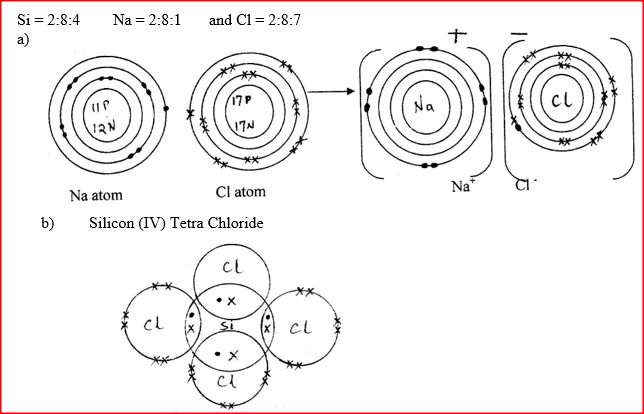
Using dots (.) and crosses(x) to represent electrons, show bonding in the compounds formed when the following elements react: (si = 14, Na = 11 and Cl = 17)
a) Sodium and chlorine b) Silicon and chlorine
a) The chart below is an outline of part of the periodic table.
With the help of vertical and horizontal lines, indicate the direction of increasing metallic nature of the elements.
Which types of elements are represented in the shaded area? b) i) Element A is the same group of the periodic table as chlorine. Write the formula of the compound formed when A reacts with potassium metal. ii) What type of bonding exists in the compound formed in (b) (l) above? Give a reason for your answers. c) Starting with aqueous magnesium sulphate, describe how you would obtain a sample of magnesium oxide. d) Write two ionic equations to show that aluminum hydroxide is amphoteric
expected response
6.
a) G b) E
With reference to iodine, distinguish between covalent bonds and Van Der Waals forces
answers
Oxygen and sulphur belong to group (VI) of the periodic table. Explain why there is a big difference their (melting points of oxygen is –2160C while that of sulphur is 440C.
ANSWER
|
Chemistry Topics
All
Archives
December 2024
|
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |



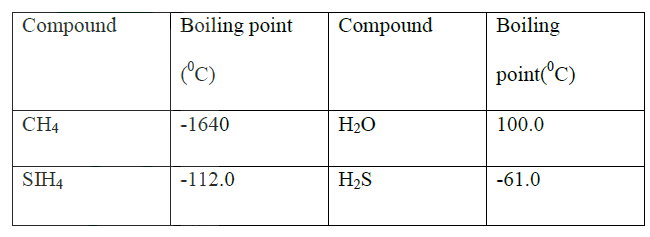

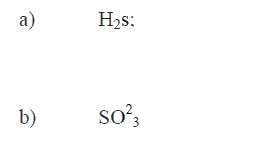


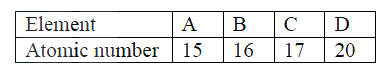

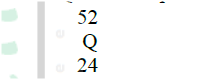

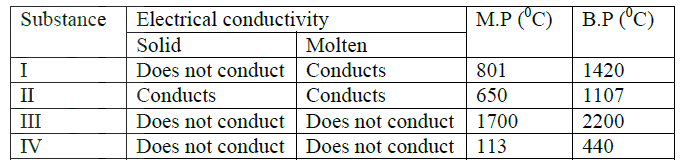

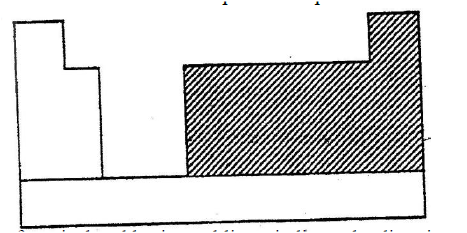


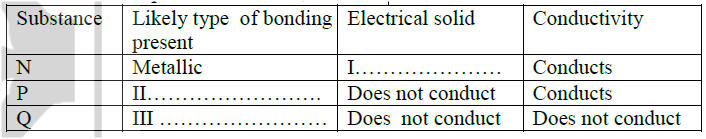



 RSS Feed
RSS Feed

