|
Urea, (NH2)2CO is prepared by the reaction between ammonia and carbon dioxide. 2NH3(g) + CO2(g) → (NH2)2CO(aq) + H2O(l). In one process, 680 kg of ammonia were reacted with excess carbon dioxide. Calculate the mass of urea that was formed. (H = 1.0, c+ 12.0, N =14.0, O = 16.0 and relative molecular mass of ammonia = 17) Calculate the mass of Urea that was formed
Expected Response
0 Comments
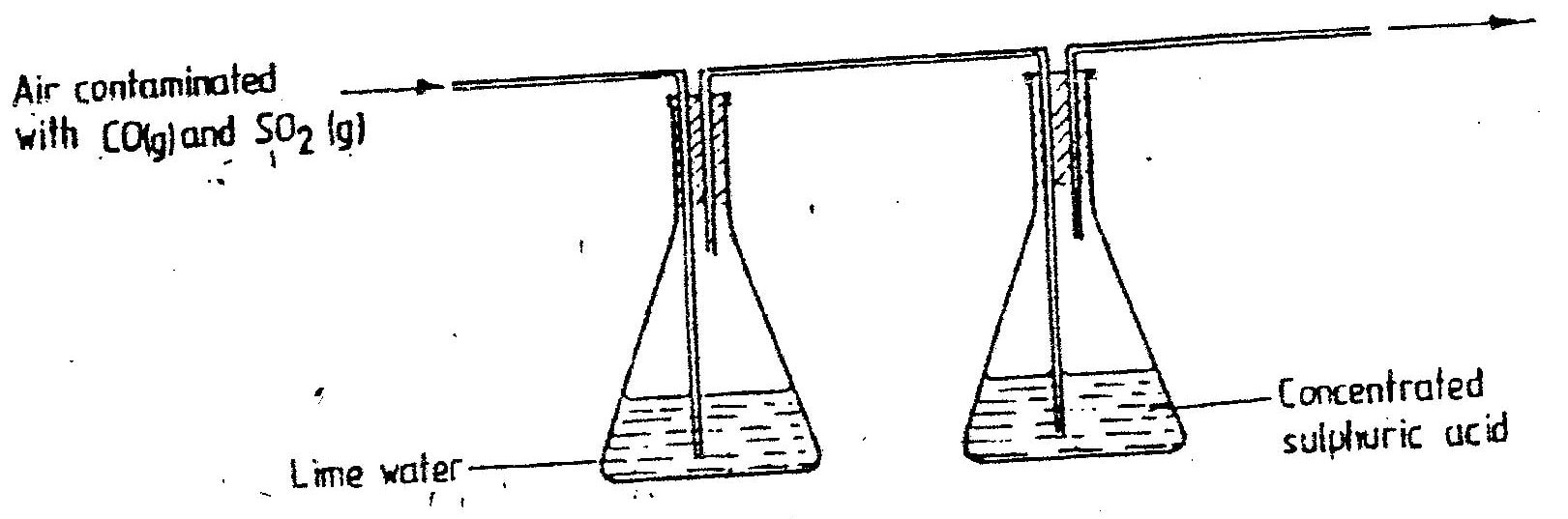
Crude oil contains sulphur. What would be the effect to the environment of using fuel containing sulphur?
ANSWERS
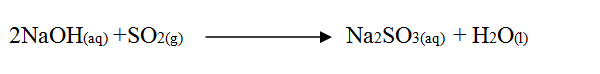
Zinc reacts with both concentrated and dilute sulphuric (VI) acid. Write equations for two reactions.
a) State the observation made at the end of the experiment when a mixture of iron powder and sulphur is heated in a test tube.
b) Write an equation for the reaction the product in (a) above and dilute hydrochloric acid. c) When a mixture of iron powder and sulphur is heated, it glows more brightly than that of iron fillings and sulphur. Explain this observation
ANSWER
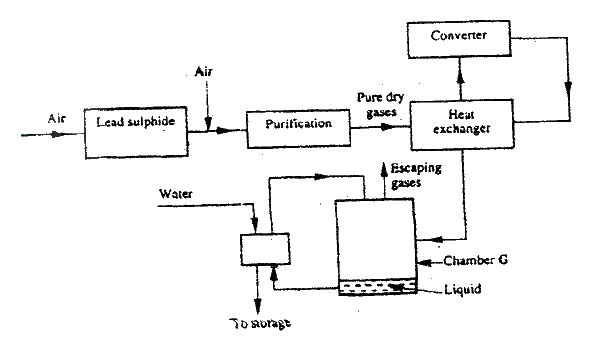
a) The diagram below shows some processes that take place during the industrial manufacture of sulphuric acid.
(i) Write the equation for the reaction in which sulphur dioxide gas is produced.
(ii) Why is it necessary to keep the gases pure and dry ? (iii) Describe the process that takes place in chamber G. (iv) Name the gases that escape into the environment. (v) State and explain the harmful effect on the environment of one of the gases named in (iv) above (vi) Give one reason why it is necessary to use a pressure of 2-3 atmospheres and not more. b) (i) Complete the table below to show the observations made when concentrated sulphuric acid is added to the substances shown.
(ii) Give reasons for the observations made using:
I iron fillings II Crystals of white sugar. (c) Name one fertilizer made from sulphuric acid. (d) Suggest a reason why BaSO4(a pigment made from sulphuric acid) would be suitable in making paint for cars.
ANSWERS
a)Conc. H2SO4
b)Heat the solution to concentrate it. Allow for crystal for form. Filter. c)Anhydrous copper(II) Sulphate
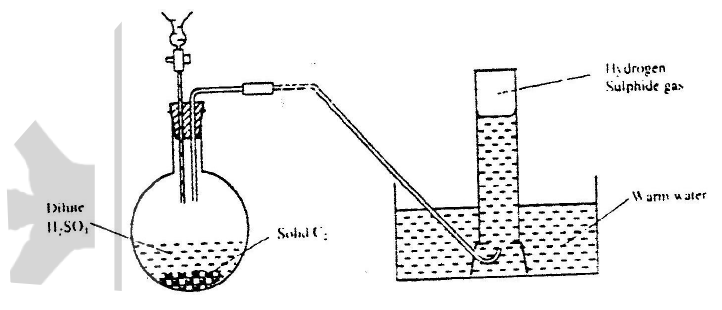
When hydrogen sulphide gas was bubbled into an aqueous solution of iron (III) chloride, a yellow precipitate was deposited.
a) The standard reduction potential for Fe2+ (aq) is -0.44 volts. Select the element which would best protect iron from rusting. b) Write an equation for the reaction that took place. c) What type of reaction was undergone by hydrogen by hydrogen sulphide in this reaction?
In an experiment to study the properties of concentrated sulphuric acid, a mixture of the acid and wood charcoal was heated in a boiling tube.
(a) write the equation of the reaction that took place in the boiling tube. (b) Using oxidation numbers, show that reduction and oxidation reactions took place in the boiling tube
In an experiment, a gas jar containing most sulphur dioxide was inverted over another gas jar containing hydrogen sulphide gas
a. State and explain the observation that was made b. State the precaution that should be taken when carrying out this experiment
ANSWERS
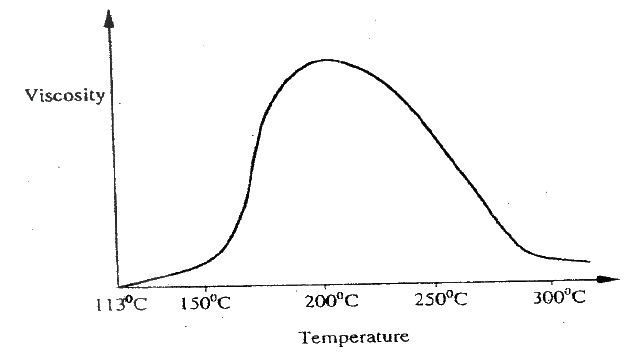
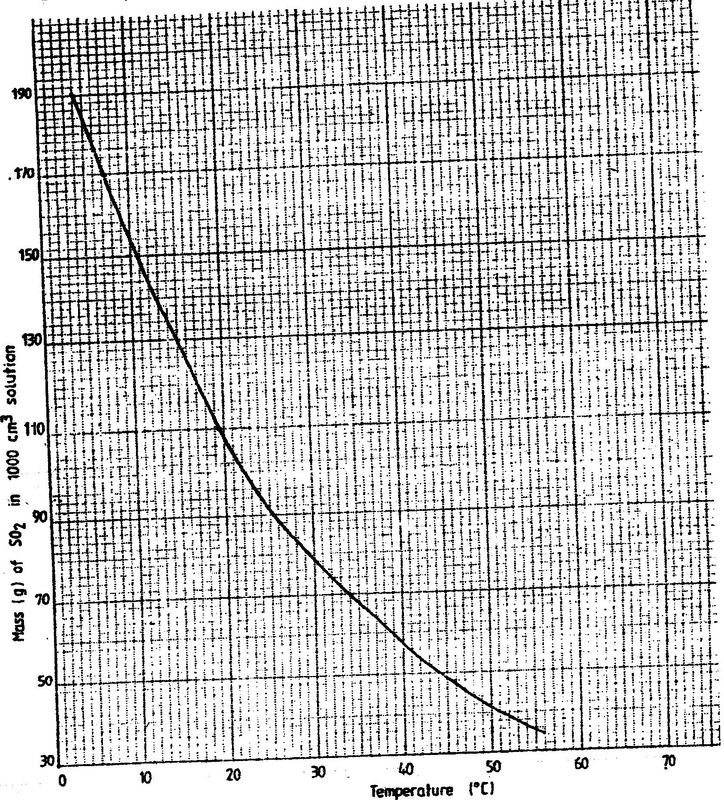
(a) The graph below shows the solubility of sulphur dioxide gas at different temperatures. Use the following in it to answer the questions that follow.
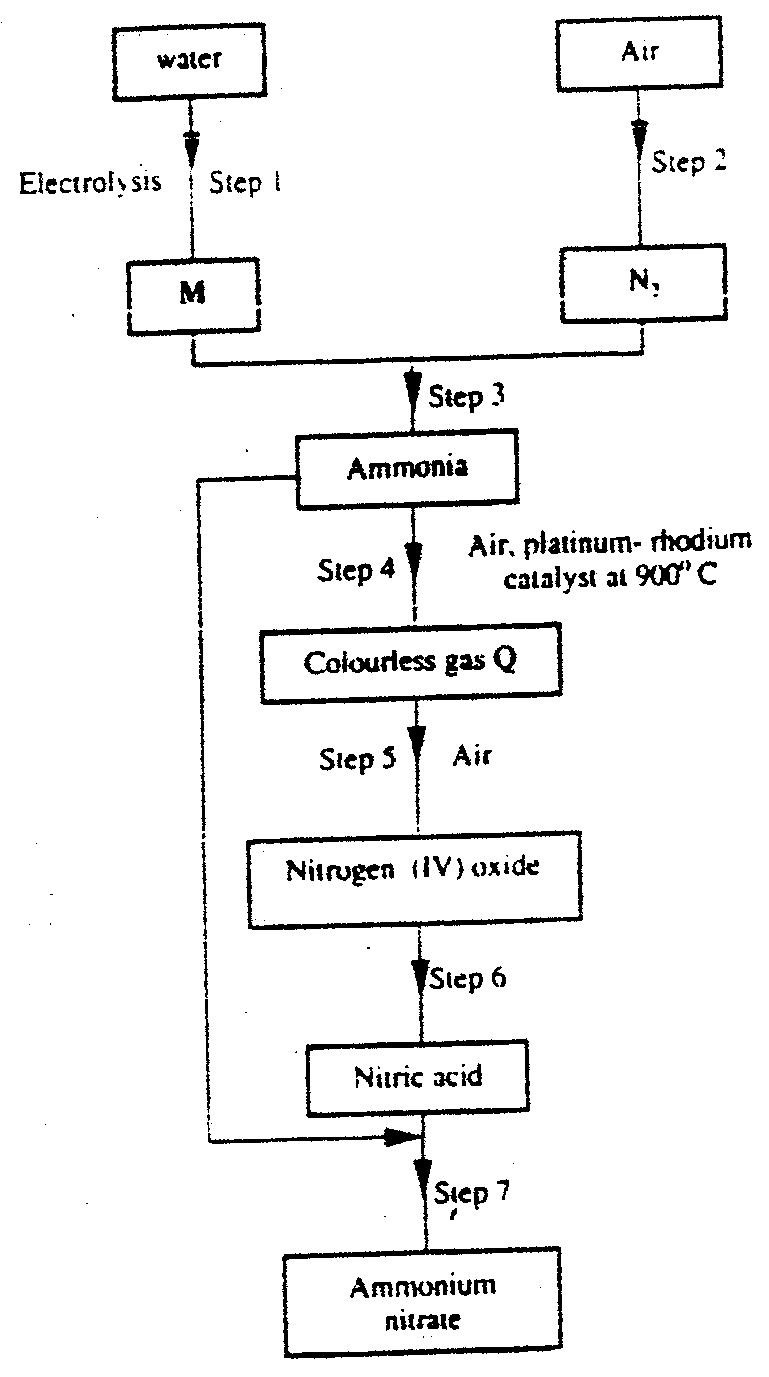
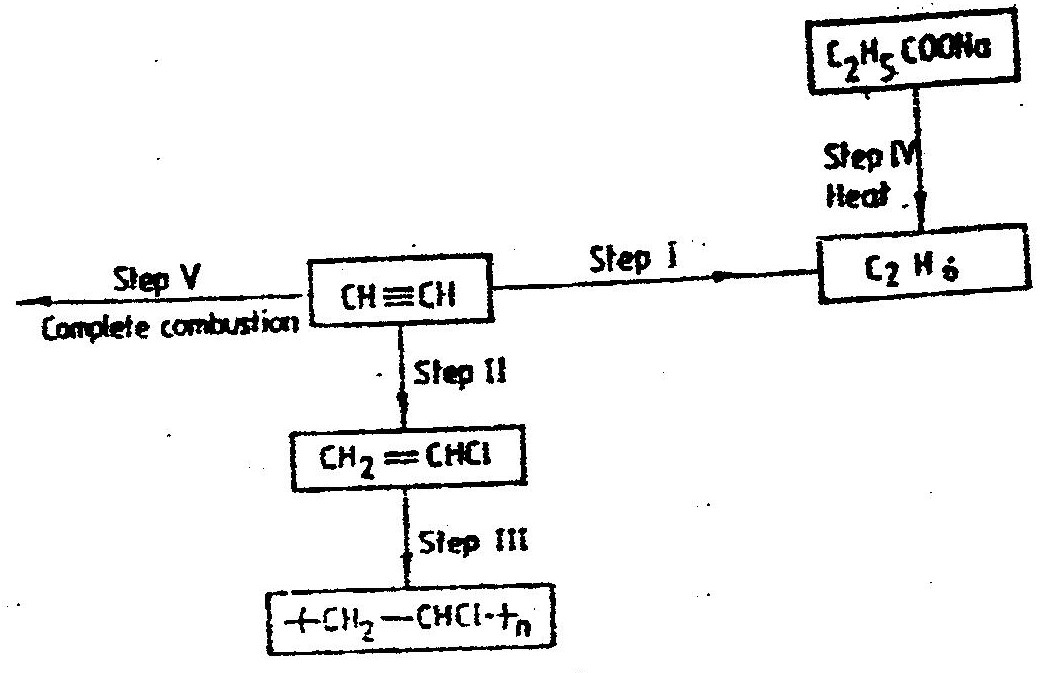
(i) From the graph determine: I The lowest temperature at which 1,000cm3 of solution would contain 116g of sulphur dioxide. II The maximum mass of sulphur dioxide that would dissolve in 15 litres of solution at 100C (ii) Sodium hydroxide reacts with sulphur trioxide according to the following equation. Using the information in the graph determine the volume of 2M sodium hydroxide required to completely neutralize on one litre of saturated sulphur dioxide solution 230C. (S=32.0; O; O = 16.0) (3mks) (b) Study the flow chart below and answer the questions that follow. Write equation for the reaction taking place at: I The roasting furnace (1mk) II The absorption tower (1mk) III The diluter (1mk) (ii) The reaction taking place in chamber K is I Explain why it is necessary to use excess air in chamber K II Name another substance used in chamber K (c) Study the scheme given below and answer the questions that follow: (i) Name the reagents used in: Step I……………… (1mk) Step II……………… (1mk) Step IV…………… (1mk) (ii) Write an equation for the complete combustion of CH = CH (1mk) Explain one disadvantage of the continued use of items made from the compound formed in step III (2mks)
Determine the oxidation state of sulphur in the following compounds:
a) H2S……………………… b) Na2S……………………
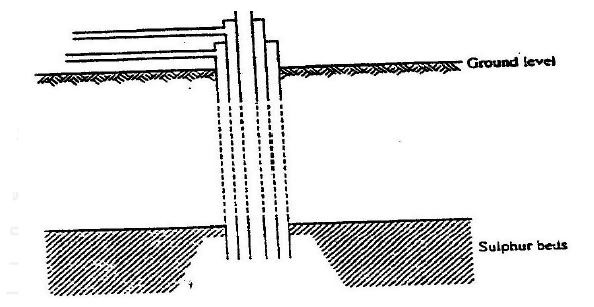
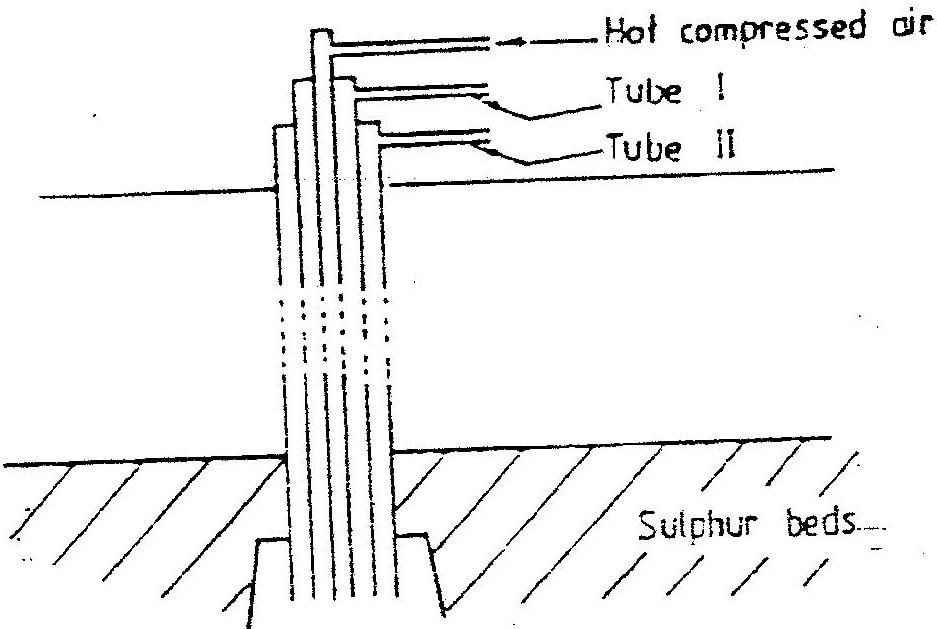
a) The diagram below illustrates how Sulphur/extracted by the Frisch process.
Label the pipe through which superheated water is pumped in
b. The equation below shows the oxidation of sulphur dioxide to sulphur trioxide in the contact process.
(i) Name one catalyst for this reaction
(ii) State and explain the effect on the yield of sulphur trioxide when: I The temperature increased II The amount of oxygen is increased (iii) Describe how sulphur trioxide is converted to sulphuric acid in the contact process. (c) State two disadvantages of having sulphur dioxide in the environment (d) Ammonia sulphate is a fertilizer produced by passing ammonia gas into concentrated sulphuric acid. (i) Write the equation for the reaction (ii) Calculate the mass in Kg of sulphuric acid required to produce 25kg of the fertilizer (S = 32.0; O= 16.0; N = 1.0)
answers
expected response
a)
i) Iron (II) Suphide ii) Hydrogen Sulphide
b) Darker paper soaked in lead acetate
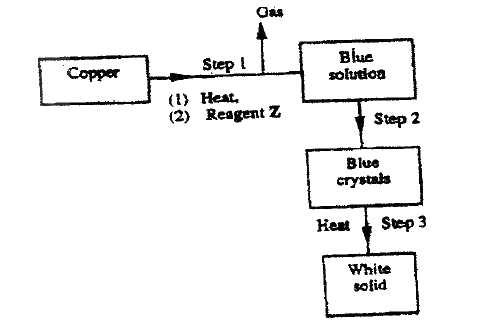
Oleum (H2S2O7) is an intermediate product in the industrial manufacture of sulphuric acid
(a) How is oleum converted into sulphuric acid? (b) Give one use of sulphuric acid
answer
(a) Sulphur exists in two crystalline forms
(b) Name one crystalline form of sulphur
expected response
Sulphur dioxide, it reacts with lime-water being an acid gas
In an experiment, sulphur dioxide gas was bubbled into water followed by chlorine gas. The resulting clear solution gave a white precipitate when mixed with a acidified barium chloride solution. Explain these observations
answers
State what would be observed when dilute hydrochloric acid is added to the products formed when a mixture of iron fillings and sulphur? (1mk)
Expected Response
A gas with a smell of rotten eggs is formed H2S gas is formed / A greenish solution is formed? Effervescence / A gas is produced / Black solid dissolves. (1mk)
Expected Response
a) I – Molten sulphur
II – Super heated water / water
b) To force the molten sulphur out
|
Chemistry Topics
All
Archives
December 2024
|
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |
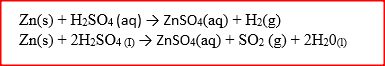
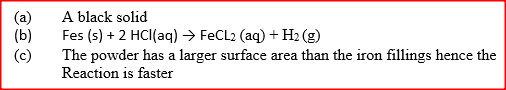
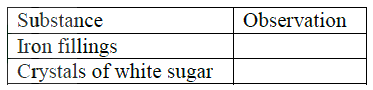



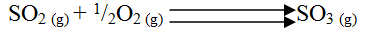

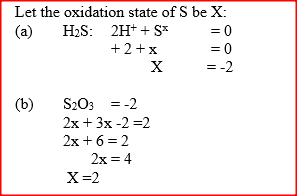
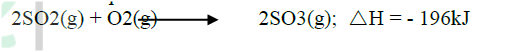

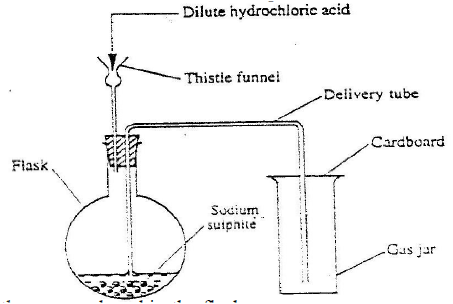

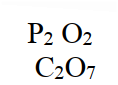


 RSS Feed
RSS Feed

