|
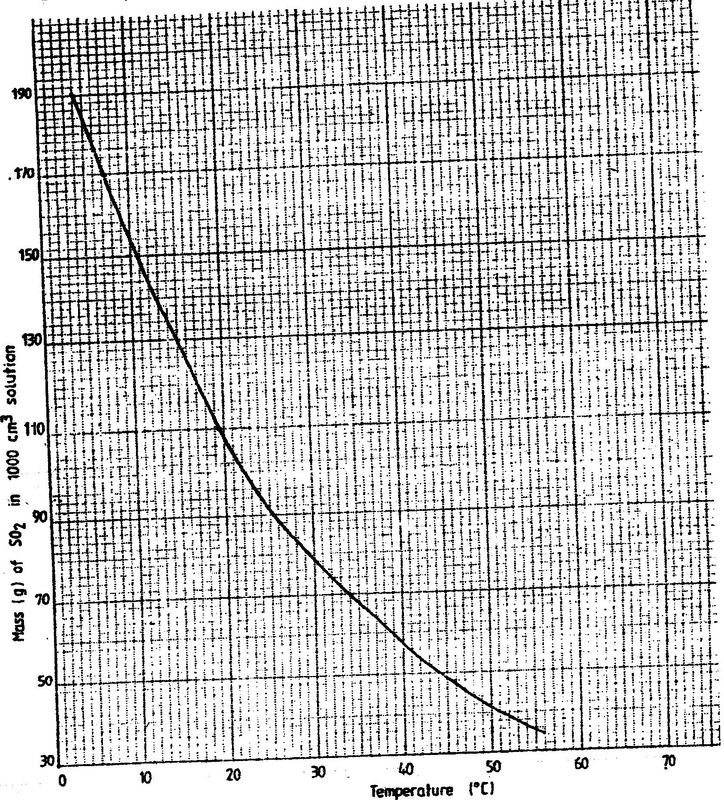
(a) The graph below shows the solubility of sulphur dioxide gas at different temperatures. Use the following in it to answer the questions that follow.
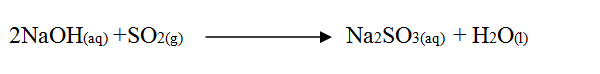
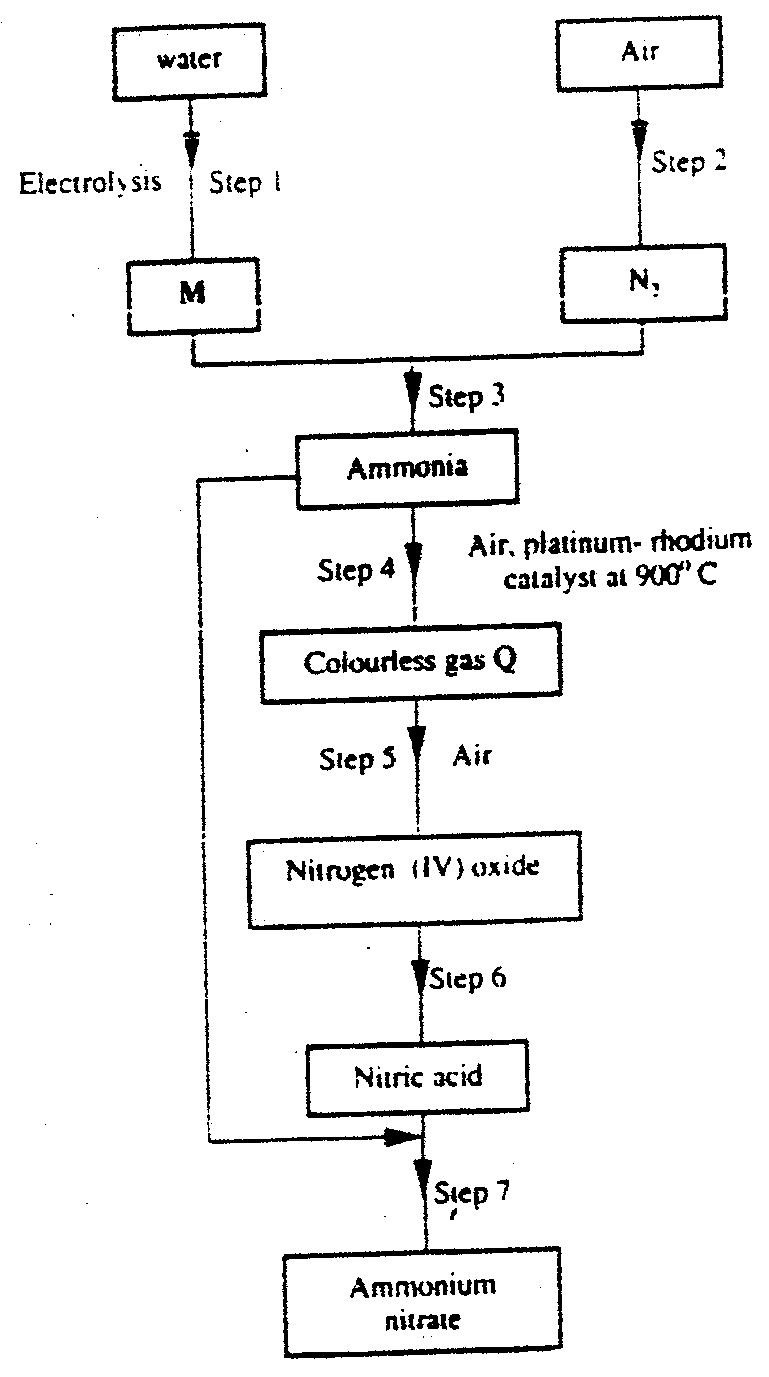
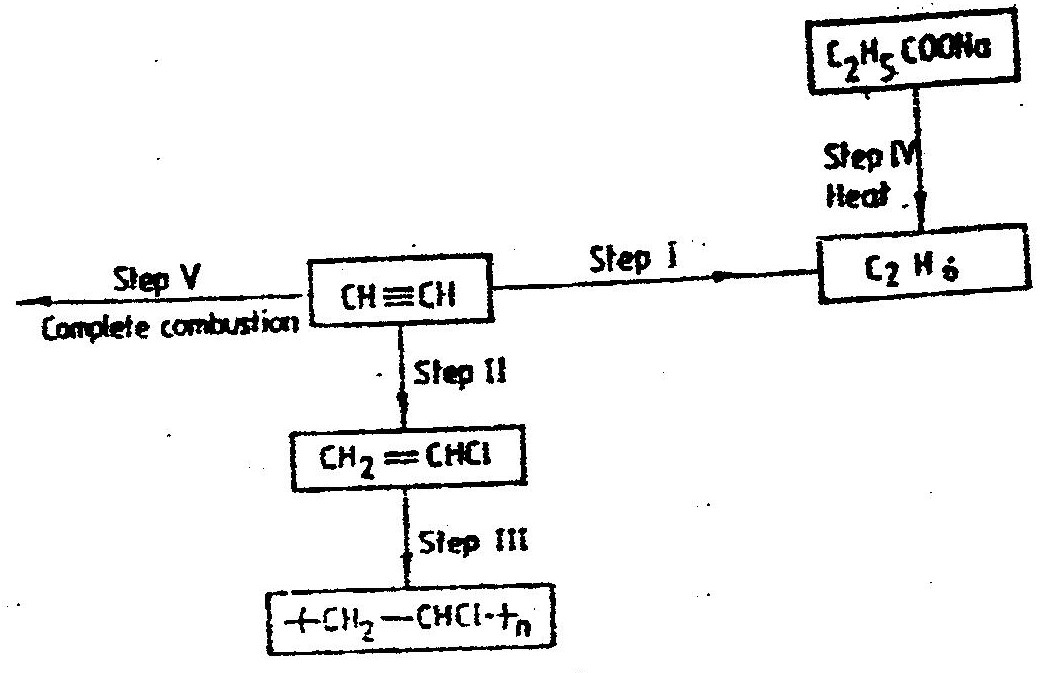
(i) From the graph determine: I The lowest temperature at which 1,000cm3 of solution would contain 116g of sulphur dioxide. II The maximum mass of sulphur dioxide that would dissolve in 15 litres of solution at 100C (ii) Sodium hydroxide reacts with sulphur trioxide according to the following equation. Using the information in the graph determine the volume of 2M sodium hydroxide required to completely neutralize on one litre of saturated sulphur dioxide solution 230C. (S=32.0; O; O = 16.0) (3mks) (b) Study the flow chart below and answer the questions that follow. Write equation for the reaction taking place at: I The roasting furnace (1mk) II The absorption tower (1mk) III The diluter (1mk) (ii) The reaction taking place in chamber K is I Explain why it is necessary to use excess air in chamber K II Name another substance used in chamber K (c) Study the scheme given below and answer the questions that follow: (i) Name the reagents used in: Step I……………… (1mk) Step II……………… (1mk) Step IV…………… (1mk) (ii) Write an equation for the complete combustion of CH = CH (1mk) Explain one disadvantage of the continued use of items made from the compound formed in step III (2mks) Related Chemistry Questions and Answers on Sulphur and it's Compounds Form 3 Level
0 Comments
Leave a Reply. |
Chemistry Topics
All
Archives
December 2024
|
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |
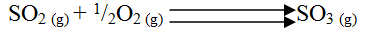


 RSS Feed
RSS Feed

