|
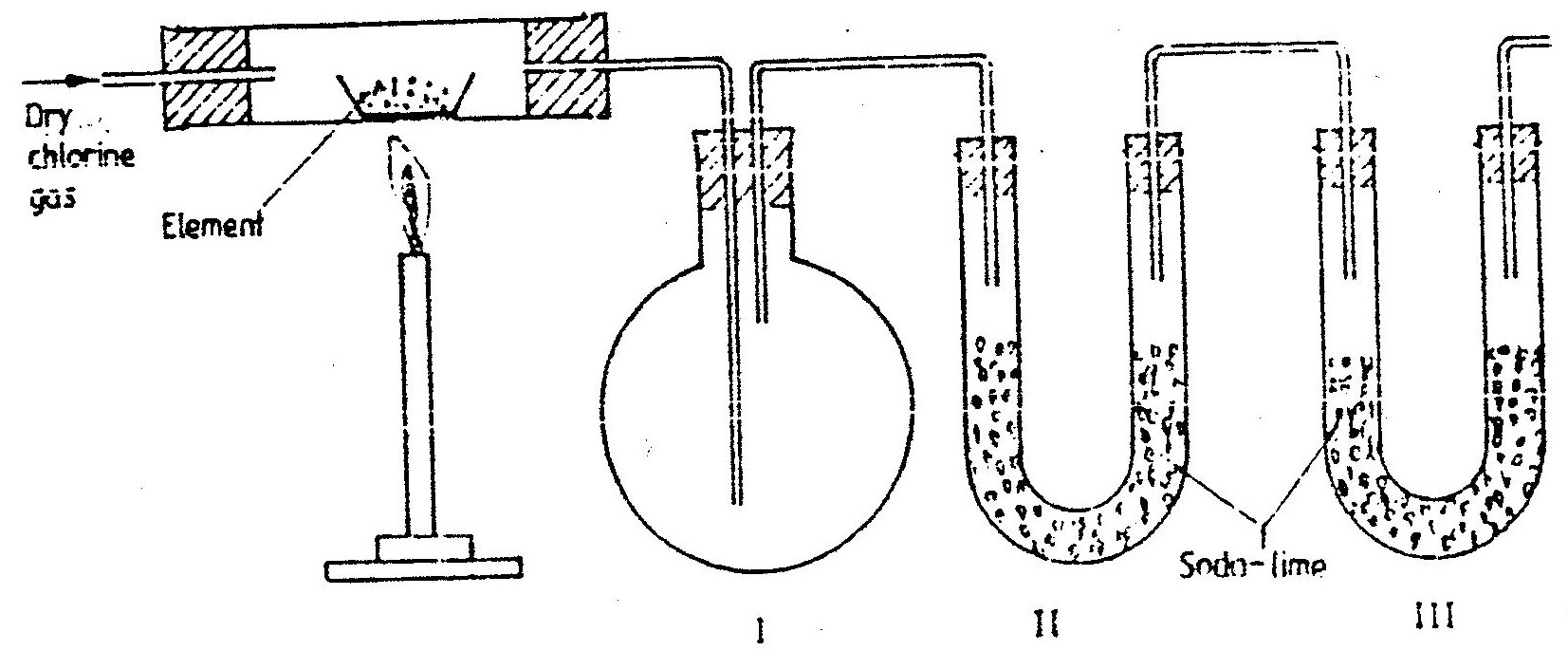
The set – up below was used to prepare anhydrous chlorides of a number of elements in a laboratory where no fine cupboard was available. The chlorides were to be collected in flask
The following table shows the melting and boiling points of the chlorides that were prepared.
More Quiz
(a) The table below gives the solubilities of hydrated copper (II) sulphate in mol dm3 at different temperatures (i) On the grid provided, plot a graph of solubility of copper (II) sulphate (vertical axis) against temperature. (ii) From the graph, determine the mass of copper (II) sulphate deposited when solution is cooled from 700C to 400C. (Molar mass of hydrated copper (II) sulphate = 250g) (b) In an experiment to determine the solubility of sodium chloride, 5.0cm3 of a saturate solution of sodium chloride weighing 5.35g were placed volumetric and diluted to a total volume of 250cm3. 25.0cm3 of the dilute solution chloride completely reacted with 24cm3 of 0.1M silver nitrate solution. AgNO3(aq) + NaCI(aq) → AgCl(s) + NaNO3(aq) Calculate: (i) Moles of silver nitrate in 24cm3 of solution (ii) Moles of sodium chloride in 25.0cm3 of sodium (iii) Moles of sodium chloride in 250cm3 of solution (iv) Mass of sodium chloride in 5.0cm3 of saturated sodium chloride Solution (Na = 23.0, CI = 35.5) (v) Mass of water in 5.0cm3 of saturated solution of sodium chloride (vi) The solubility of sodium chloride in g/100 water Related Chemistry Questions and Answers on Salts Form 2 Level
0 Comments
Leave a Reply. |
Chemistry Topics
All
Archives
December 2024
|
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
Can't find what you want? Use this Search box. |
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |



 RSS Feed
RSS Feed

