|
(a) What is meant by molar heat of combustion?
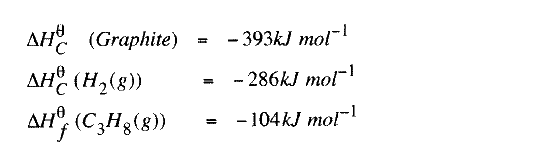
(b) State the Hess's Law. c) Use the following standard enthalpies of combustion of graphite, hydrogen and enthalpy of formation of propane.
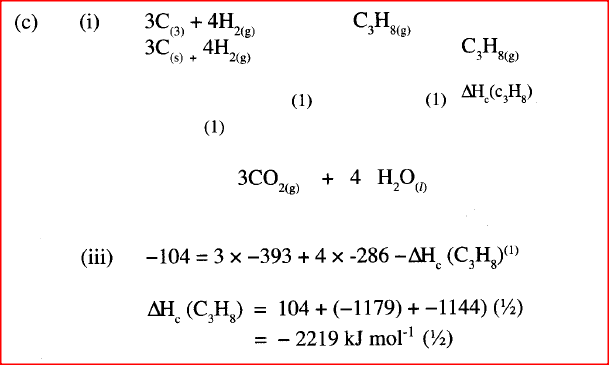
(i) Write the equation for the formation of propane.
(ii) Draw an energy cycle diagram that links the heat of formation of propane with its heat of combustion and the heats of combustion of graphite and hydrogen. (iii) Calculate the standard heat of combustion of propane. Other than the enthalpy of combustion, state one factor which should be considered when choosing a fuel. (e) The molar enthalpies of neutralization for dilute hydrochloric acid and dilute nitric (V) acid are -57.2kJ/mol while that of ethanoic acid is -55.2kJ/mol. Explain this. observation.
ANSWERS
(a) The amount of heat liberated when one mole of a substance is burnt in excess Oxygen.
(b) The heat evolved are absorbed in a chemical change is the same whether the change occurs in one step or through many steps.
(d) cost
effect on environment availability storage (e) Ethanoic acid is a weak acid therefore heat is used to ionize it before neutralization occurs . It value is therefore lower than that of hydrochloric acid which is fully ionized Related Chemistry Questions and Answers on Energy Changes in Chemical and Physical Processes Form 4 Level
0 Comments
Leave a Reply. |
Chemistry Topics
All
Archives
December 2024
|
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |

 RSS Feed
RSS Feed

