|
(a) (i) what is meant by the term Enthalpy of formation?
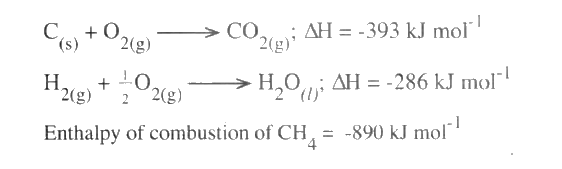
(ii) The enthalpies of combustion of carbon, methane and hydrogen are indicated below:
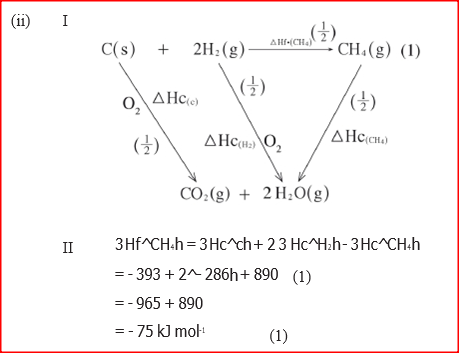
(i) Draw an energy cycle diagram that links the enthalpy of formation of methane to enthalpies of combustion of carbon, hydrogen and methane
(ii) Determine the enthalpy of formation of methane (b) An experiment was carried out where different volumes of dilute hydrochloric acid and aqueous sodium hydroxide both at 25°C were mixed and stirred with a thermometer. The highest temperature reached by each mixture was recorded in the table below
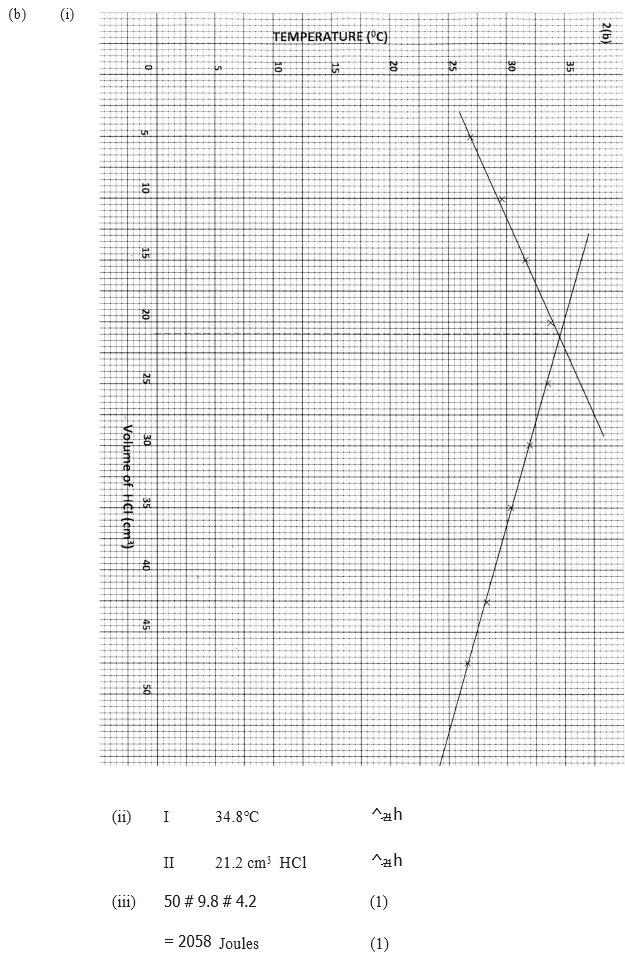
(i) On the grid provided. plot a graph of highest temperature (vertical axis) against volume of hydrochloric acid ( horizontal axis)
(ii) Using your graph , determine the (a) Highest temperature reached (b) Volume of acid and base reacting when highest temperature is reached
(iii) Calculate the amount of heat liberated during the neutralization process .(specify heat capacity is 4.2 j g -1 k-1 and the density of solution is 1.0 g cm-3)
(c) The molar enthalpy of neutralization between hydrochloric acid and ammonia solution was found to be -52.2 kJ mol-1, while that of hydrochloric acid and sodium hydroxide was – 57.1 Kj mol-1. Explain the difference in these values.
ANSWERS
(a)(i)This is the heat absorbed or evolved when one mole of any substance is formed from its constituent elements in their normal states.
(c)The molar heat of neutralisation between a strong acid and a weak base is low because some of the heat is used to ionise (1) the weak base before neutralization.
For strong acid and strong base they are completely ionised. Related Chemistry Questions and Answers on Energy Changes in Chemical and Physical Processes Form 4 Level
0 Comments
Leave a Reply. |
Chemistry Topics
All
Archives
December 2024
|
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |


 RSS Feed
RSS Feed

