Calculating Enthalpy Change using Hess's Law - A Comprehensive GuideTo calculate Hess's Law, we need to understand the concept of enthalpy change. Enthalpy change is the heat energy transferred during a chemical reaction. Hess's Law states that the total enthalpy change of a reaction is independent of the pathway taken. Here is the step-by-step process to calculate the enthalpy change using Hess's Law:
By following these steps, you can calculate the enthalpy change of a reaction using Hess's Law. It is important to note that accurate and reliable data for the enthalpy changes of the simpler reactions is crucial for obtaining an accurate result. Formula for Calculating Enthalpy Change using Hess's LawThe formula to calculate the enthalpy change using Hess's Law is as follows:
ΔH = ΣnΔHf(products) - ΣmΔHf(reactants) In this formula, ΔH represents the enthalpy change of the target reaction that we want to calculate. The enthalpy change is the heat energy transferred during the reaction. The ΣnΔHf(products) term represents the sum of the enthalpy of formation of the products, multiplied by their stoichiometric coefficients. The enthalpy of formation (ΔHf) is the enthalpy change when one mole of a compound is formed from its constituent elements in their standard states. The stoichiometric coefficients (n) represent the number of moles of each product involved in the reaction. Similarly, the ΣmΔHf(reactants) term represents the sum of the enthalpy of formation of the reactants, multiplied by their stoichiometric coefficients. The enthalpy of formation (ΔHf) of the reactants is also determined when one mole of the compound is formed from its constituent elements in their standard states. The stoichiometric coefficients (m) represent the number of moles of each reactant involved in the reaction. By plugging in the values of the enthalpy of formation for the products and reactants, along with their respective stoichiometric coefficients, you can calculate the enthalpy change (ΔH) of the target reaction. It's important to note that the enthalpy of formation values can be found in reference books or databases, and they are typically given for standard conditions (25°C and 1 atm). Also, make sure to consider the sign of the enthalpy change (positive or negative) based on whether the reaction is exothermic or endothermic. Using this formula, along with the step-by-step process mentioned earlier, you can accurately calculate the enthalpy change of a reaction using Hess's Law.
0 Comments
he Relationship Between Hydration, Lattice, and Heat of Solution in DissolutionHydration, Lattice, and Heat of Solution: Exploring the Relationship The process of dissolution involves the interaction between a solute and a solvent, resulting in the formation of a homogeneous mixture. In the case of a solid solute dissolving in a liquid solvent, hydration, lattice, and heat of solution play crucial roles. Hydration: Hydration refers to the process in which water molecules surround and interact with solute particles, leading to the formation of hydrated ions. Water is an excellent solvent due to its unique properties, such as polarity and ability to form hydrogen bonds. When a solute dissolves in water, the water molecules surround the solute particles and establish hydrogen bonding interactions. This hydration process weakens the attractive forces between the solute particles, facilitating their separation and dispersion throughout the solvent. Lattice: In a solid solute, the particles are arranged in a regular, repeating pattern called a lattice structure. The lattice structure determines the strength of the forces holding the particles together. These forces can be ionic, covalent, or metallic, depending on the nature of the solute. When a solute dissolves, the lattice structure is disrupted as the solute particles separate from each other. This separation allows the hydration process to occur and leads to the formation of hydrated ions. Heat of Solution: The heat of solution is a measure of the energy change that occurs during the dissolution process. It represents the amount of heat energy absorbed or released when a solute dissolves in a solvent. The heat of solution can be either exothermic or endothermic, depending on the nature of the solute-solvent interaction. Relationship between Hydration, Lattice, and Heat of Solution:The relationship between hydration, lattice, and heat of solution can be understood as follows:
Overall, understanding the relationship between hydration, lattice, and heat of solution is essential in comprehending the process of dissolution and the behavior of solutes in solvents. Mastering the Art of Heat Transfer: A Comprehensive Guide to Efficient Energy ExchangeHeat transfer is the process of energy exchange between objects or systems due to the temperature difference between them. It occurs in three main ways: conduction, convection, and radiation.
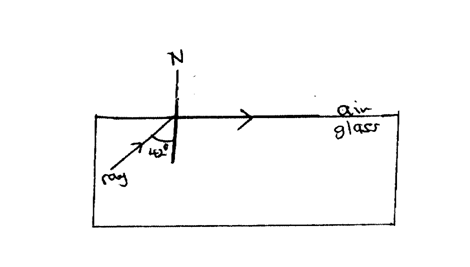
The figure below shows the path of a ray of light passing through a rectangular glass block placed in airCalculate the refractive index of glass (2mks)
Two capacitors of capacitance 2 μF 1μF are connected in parallel. A p.d of 3V is applied across them. Find the energy stored in the combination. (2mks)
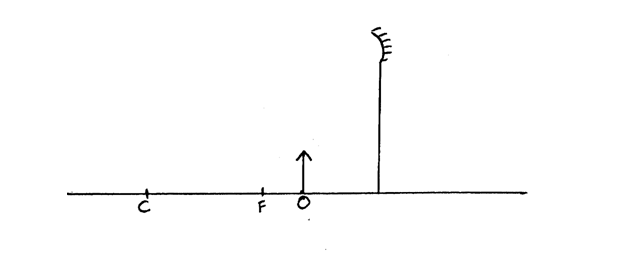
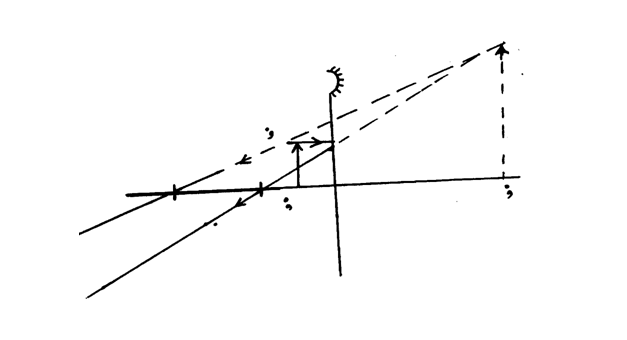
The figure below shows an object placed in front of a concave mirror. By use of correct ray diagram, locate the position of imageSolution
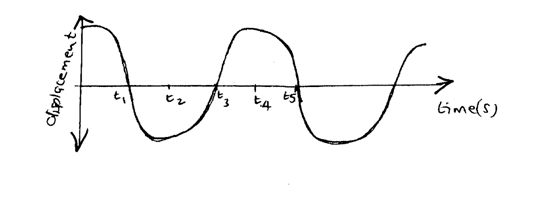
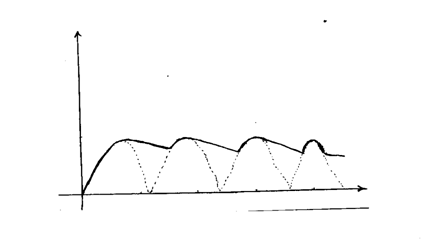
The figure bellows shows a wave profile for a wave whose frequency is 2HZDetermine the value of t3(s) (2mks)Solution
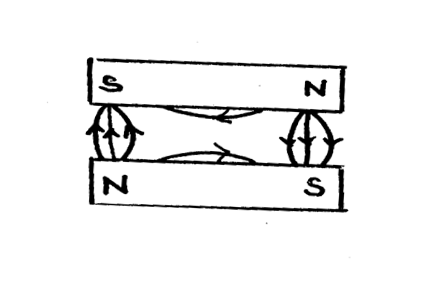
The figure below shows two parallel magnets with unlike poles adjacent to each other. Sketch the magnetic field pattern around the magnets (2mks)Solution
(a) Define Radio-activity (2mks)
Radioactivity is the spontaneous disintegration of a nucleus releasing
alpha, beta and gamma radiations accompanied by the release of energy (b) When carrying out experiments with radioactive substances the source should never be held with bare hands but with forceps. Explain? (1mk)
Radioactive substances are harmful to body cells when ingested
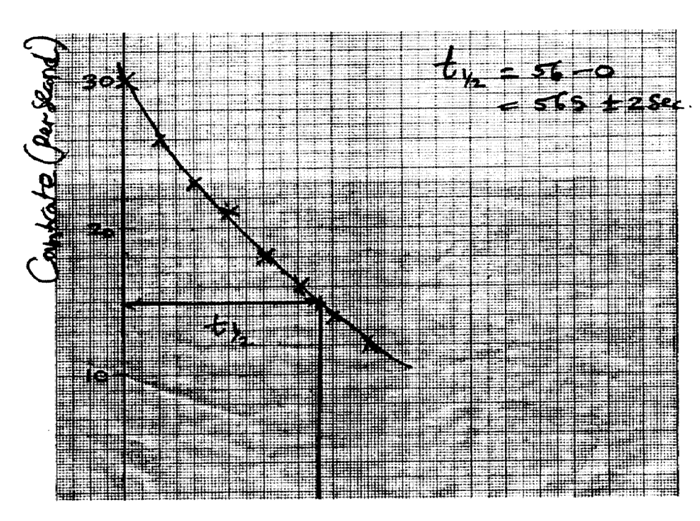
(i) Plot on the graph paper provided the graph of the count rate (y-axis) against time(ii) From the graph, determine the half-life of Radon-220 (1mk)
Half-life of radon 220=54 sec + 2 (showing on graph a must)
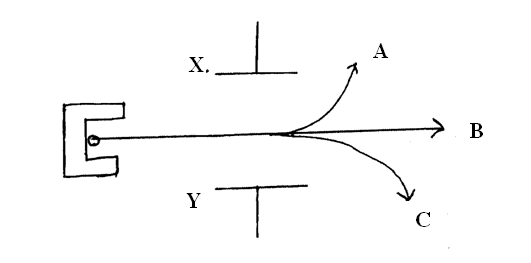
(e) The diagram in the figure below shows paths taken by three radiations A,B and C from a radioactive isotope through an electric field.(i) State the charge on plate Y (1mk)
(ii) Identify the radiations A and C (2mks)
(iii) Give a reason why C deviates more than A (1mk)
C more massive than A
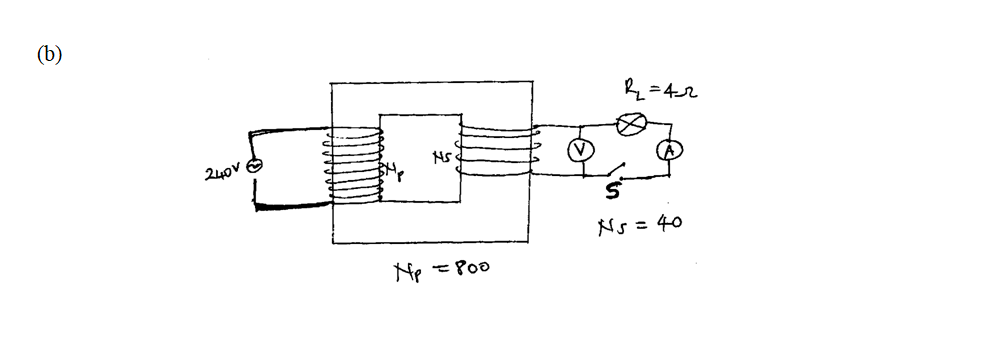
(a) A 240v mains transformer has 800 turns in the primary and N turns in the secondary. It is used to supply energy to a 12v/21w lamp. (i) Calculate the number of turns in the secondary (2mks)(ii) What is the efficiency of the transformer if the current drawn from the 240V mains is 100mA (2mks)
Calculate.(i) the reading of voltmeter, V, with, S, open (2mks)(ii) the reading of the ammeter A with S closed (neglect the effective resistance of the secondary winding) (2mks)(iii) the power dissipated in the lamp (2mks)(iv) the primary current if the transformer is 90% efficient (3mks)
(a) State two advantages of a C.R.O as a voltmeter
(b) A television tube is a cathode ray tube modified. State two modifications
(c) State the functions of the following parts of a C.R.O(i) Grid
(ii) X-plates
(iii) Fluorescent screen
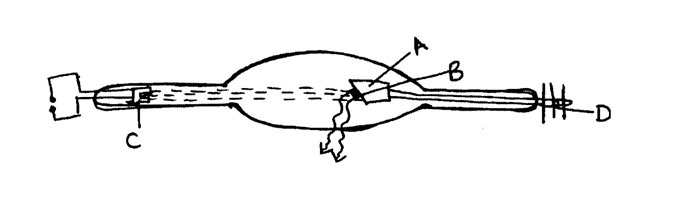
(d) A modern X-ray tube is shown below(i) Name the parts labeled A-D (2mks)A
Copper Anode
B
Tungsten Target
C
Filament
D
Cooling fins
(ii) Give the functions of:- (1mk)(i) Shielding with lead
Lead absorbs the radiation (1mk)
(ii) Evacuating the tube
Prevents collisions, avoid secondary electrons (1mk)
(a) Define doping (1mk)
Process of introducing foreign element (impurity) into a pure semi-conductor,
(b) Distinguish between a p-type and n-type semi conductors (2mks)
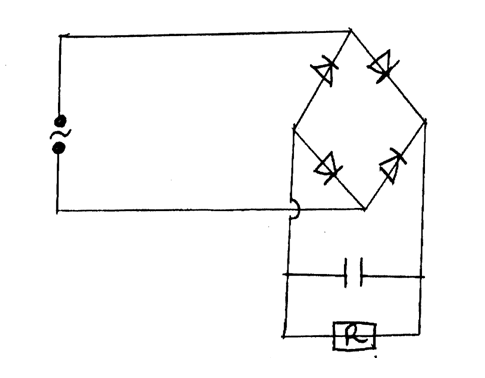
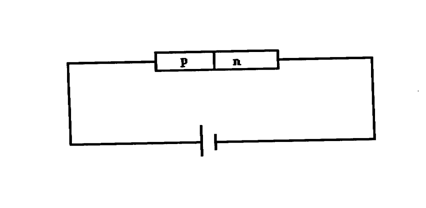
(c) Using a diagram illustrate a forward P-N junction (2mks)(d) The figure below shows a bridge circuit(i) Sketch of the figure below the wave form when a CRO is connected across the resistor, R(ii) On the same axes, sketch a wave form when C.R.O is connected across R when the capacitor has been removed (1mk)
(a) Explain why bulbs in along corridor are not wired in series (2mks)
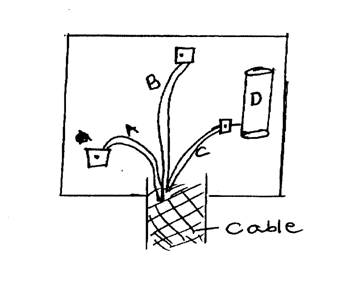
(b) An electric cooker has an oven rated 2500w, a grill rated 1500w and two plates each of 1000W. This cooker operates directly from 240v supply. What is the costs of operating all the parts at once for 30 minutes if electricity costs sh.10.00 per unit? (3mks)(c) Study the figure below(i) What name is given to the fitting in the diagram (1mk)
3 pin plug
(ii) Identify the parts labeled (2mks)A
Neutral wire
B
Earth wire
C
Live wire
D
Fuse
Give two advantages of a circuit breaker instead of a fuse in domestic wiring (2mks)
State the conditions for generation of e.m.f from a magnet (2mks)
The following is a part of a radio-active seriesIdentify the radiation r, find the values of c and d (3mks)r
r is βeta particle
c
c is 206
d
d is 82
State two uses of optical fibres whose working relies on total internal reflection (2mks)
Give a reason why gases are poor transmitters of sound (1mk)
Molecules in gases are much further apart
Other than penetrating powers, state two differences between soft and hard X-rays (2mks)
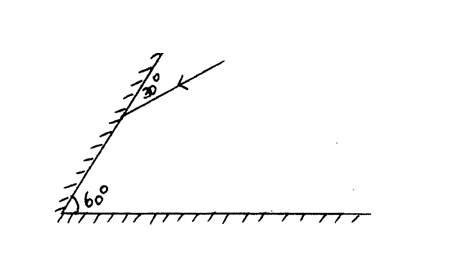
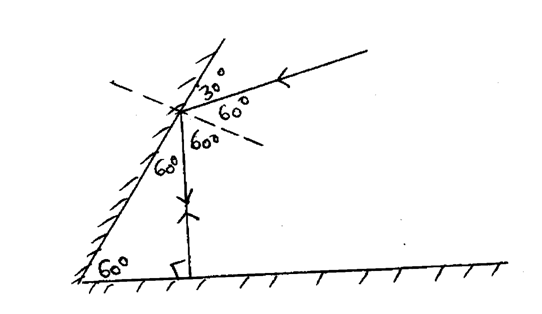
Two mirrors are inclined at 60o to each as shown:
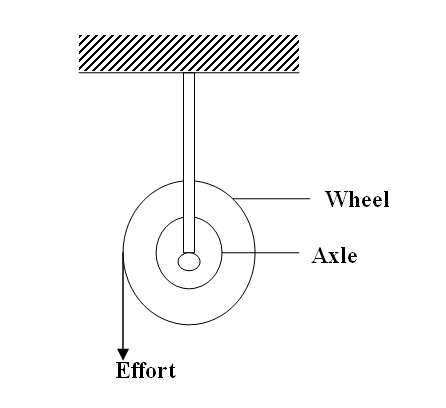
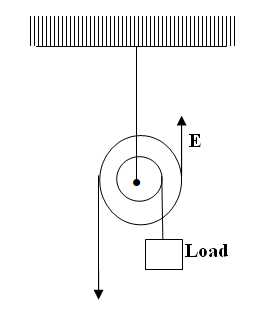
Complete the ray diagram to show how it travels after striking the two mirrors and find the angle of reflection on each surface (2mks)Solution(a) The diagram in the figure below represents a wheel and axle used as a machine, whose efficiency is 80% to raise 400N of building materials. The wheel and axles have diameters of 75cm and 15cm respectively.
(i) Mark on the diagram the correct position and direction of the load to be lifted (1mk)(ii) Name the principle on which this machine works (1mk)
The principle of moments
(iii) Calculate the effort needed to raise the load (3mks)
(iv) The machine is operated manually and raises the load to a height of 5m in 20 seconds. Calculate the power developed by the operator (2mks)(b)
|
CATEGORIES
Categories
All
Topics
FORM I - PHYSICS SYLLABUSFORM II - PHYSICS SYLLABUSTOPICS
FORM III - PHYSICS SYLLABUSFORM IV - PHYSICS SYLLABUSARCHIVES
RSS FEEDS
AUTHOR
M.A NyamotiMy passion is to see students pass using right methods and locally available resources. My emphasis is STEM courses
|
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |
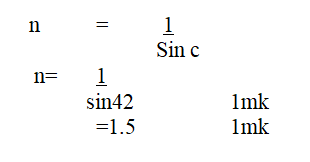
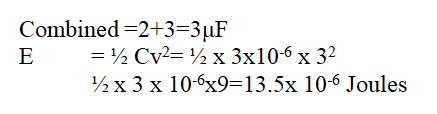
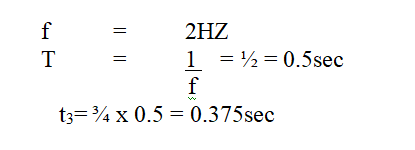

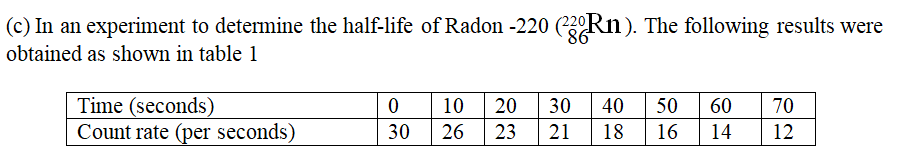


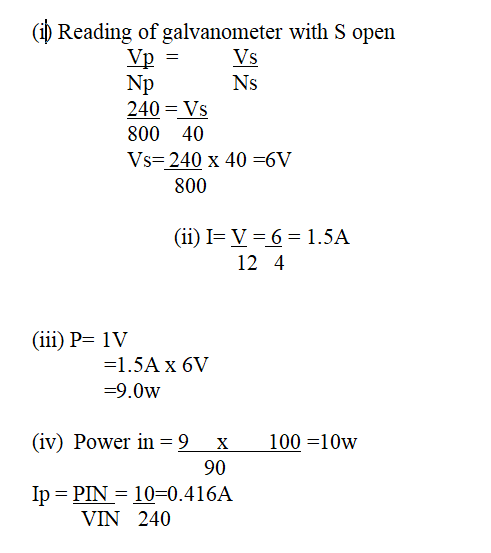

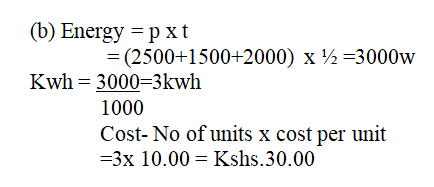
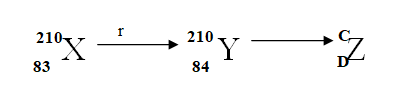
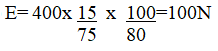
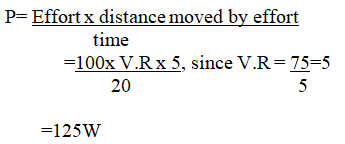
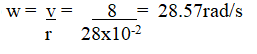
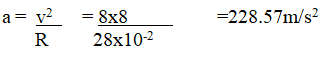

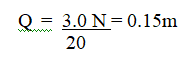
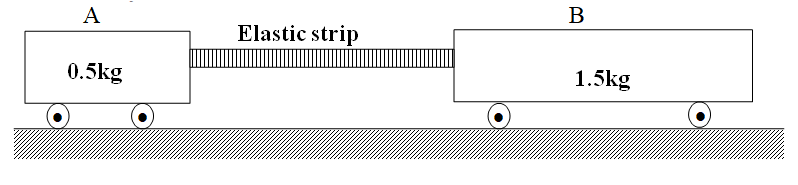
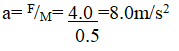
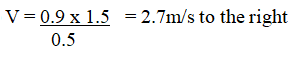
 RSS Feed
RSS Feed

