PHYSICS HIGH SCHOOL NOTES FOR KCSE PREPARATIONS
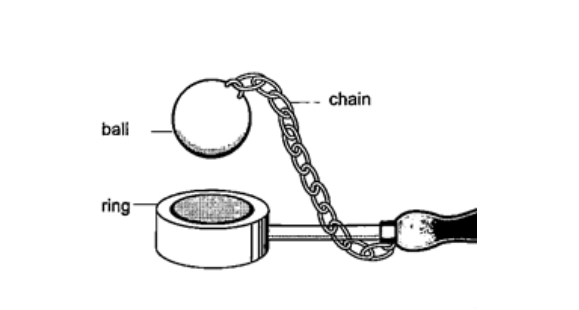
THERMAL EXPANSION.IntroductionTemperature is the degree of hotness or coldness of a body. Both Celsius scale (0C) and Kelvin scale (thermodynamic scale) are used to measure temperature. The Kelvin scale is also known as the absolute scale temperature and is measured from absolute zero (0 K).Expansion of solidsWhen solids are heated they expand. The expansion is so small such that we can’t see them.The following experiments will demonstrate actual expansion of solids. Experiment 1:- Ball and ring experiment Procedure
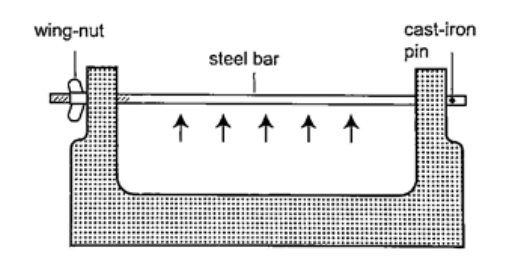
DiscussionWhen the ball is heated it expands and increases in diameter. This makes the ball not to pass through the ring. After cooling it is found that the ball slips through the ring easily again.Experiment 2:- The bar-breaker Procedure
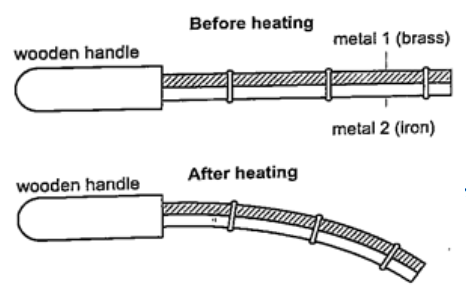
DiscussionWhen the bar cools the cast-iron pin breaks. This shows that as the bar cools it contracts and strong forces pull against the pin. These forces makes the pin to break. Experiment 3:- Heating a bimetallic strip Procedure
DiscussionWhen a brass-iron bimetallic strip is heated it bends towards the iron. This means that brass expands more than iron and this causes the strip to bend towards the iron side. This shows that different materials expand at different rates when heated.Applications of the expansion of solids
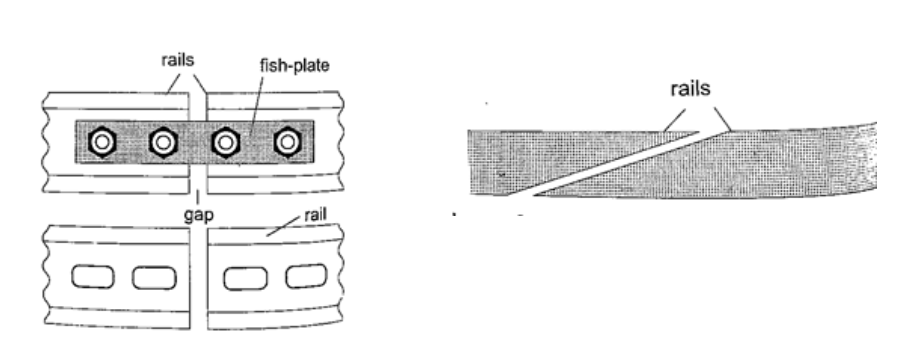
1. Construction of railway lines– an expansion joint is allowed between any two rails to accommodate expansion. A fish plate is used to join two rails. Modern railway system use the overlapping joint at the end of rails.
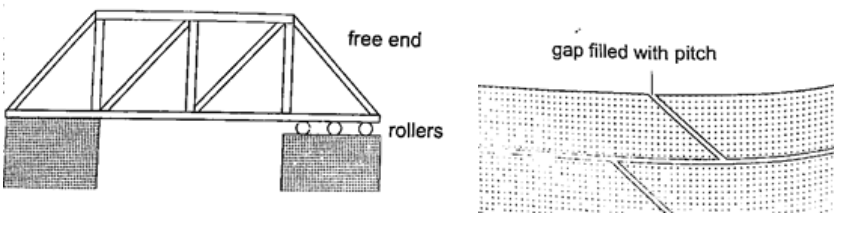
2. Construction of bridges and roof tops (steel girders) – for bridges one side has rollers while the other is fixed to allow for expansion. Concrete slabs are also laid on the ground leaving space filled with pitch to allow for expansion.
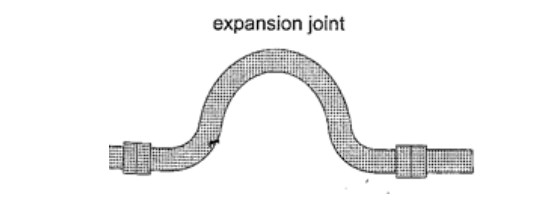
3. Hot water pipes – pipes carrying hot water (steam) from boilers are fitted with expansion joints for expansion.
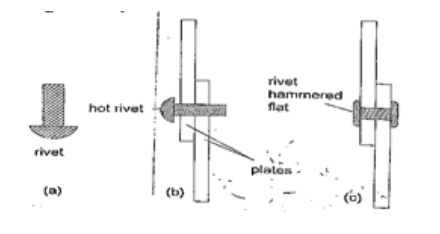
4. Riveting – used to join two pieces of metal together i.e. bimetallic strips, car bodies, drums etc. Fitting rail cart wheel using heat uses the principle of rivets. Bimetallic strips are used in thermostats (control temperature) – electric iron box, alarm systems, car flasher units etc.
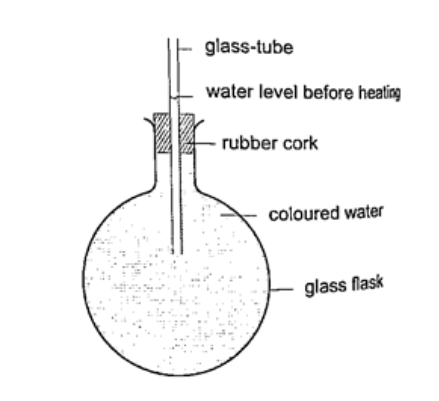
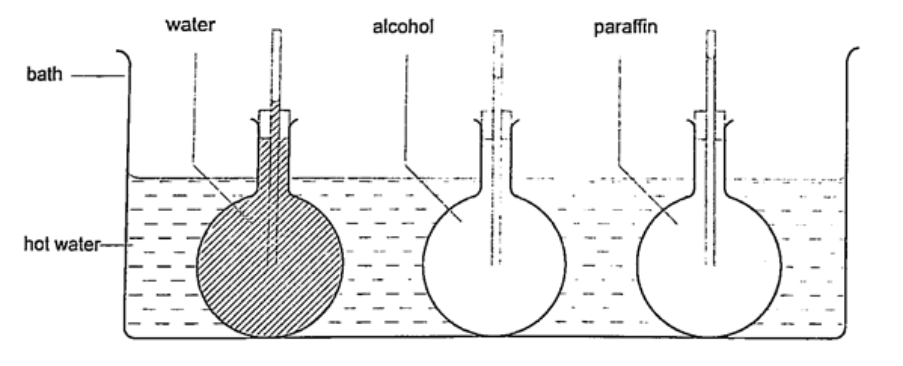
Expansion of liquids and gases.Expansion of liquids.Liquids expand more than solids so it is easy to observe and see clearly as they expand. We use the hot water bottle to demonstrate the expansion of water. Water is put in the bottle as shown below.
When the bottle is immersed in hot water, initially there is a drop in the level of water in the glass tube then it steadily rises after a while. This shows that liquids expand with increment in volume as shown by the hot water bottle. Different liquids expand at different rates as shown below.
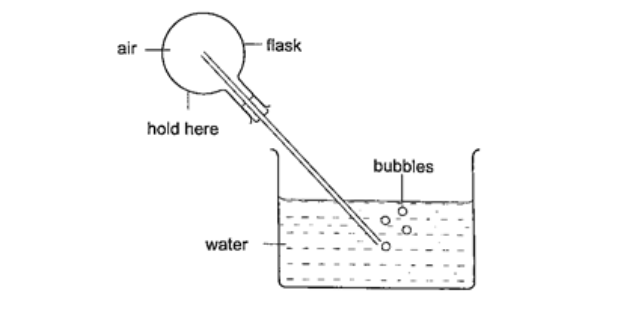
Expansion of gasesThey are the easiest to observe since they expand the most.Experiment: - Expansion of air Procedure
DiscussionThe heat produced by the hands makes the air inside the flask to expand. This makes the volume to increase and therefore force the excess air out as bubbles.Applications of the expansion of gases and liquids.
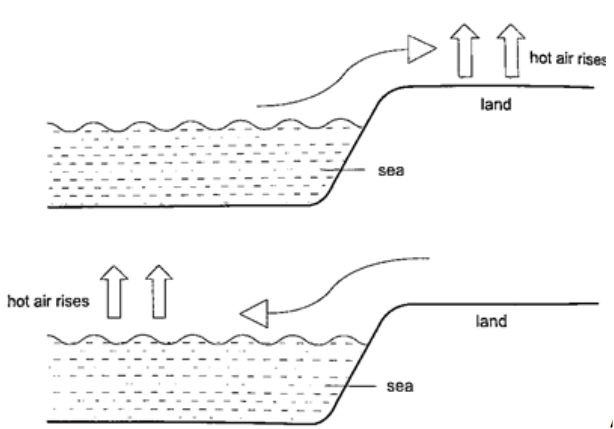
1. Land and sea breeze– during the day the land is heated by the sun causing the air above it to expand. The air becomes less dense therefore it rises. The space left is quickly filled by another cool air (generally from the sea since the land gets hot faster). This causes a cool breeze form the sea during the day. At night the land loses heat faster than the sea. The air above the sea rises since it is less dense and cool air from the land rushes to fill the gap. This causes a breeze blowing from the land to the sea.
Thermometers
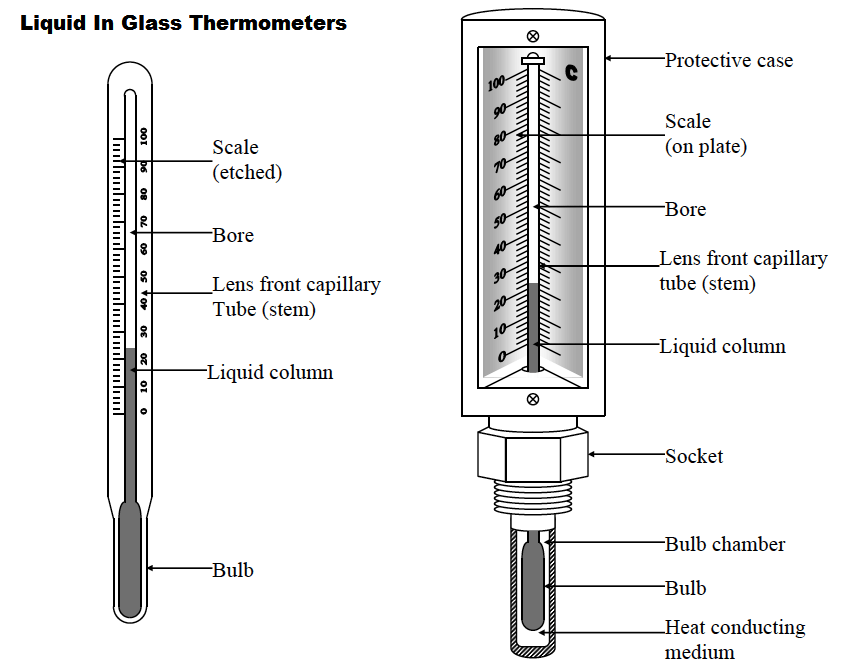
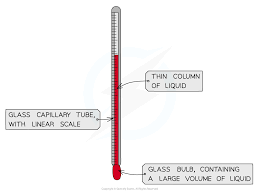
1. Liquid-in-glass thermometer–this applies to the expansion of a liquid in a thin-walled glass-tube. The liquid moves up the tube when the bulb is heated. The liquid must be a good conductor, visible and be able to contract and expand quickly and uniformly over a wide range of temperatures. It should also not stick on the sides of the tube.
Liquids commonly used are mercury and coloured alcohol. The scale is obtained by choosing two temperature points called fixed points. In Celsius lower point is taken to be 0oC (when placed in ice) and the upper point as 100oC (boiling steam). The two points are therefore divided into 100 equal parts (calibration). The melting and boiling points of both mercury and alcohol are (-39oC – 357 oC) and (-112 oC - 78 oC) respectively.
2. Clinical thermometer– this is a special type of mercury-in-glass thermometer used to measure body temperature. Since body temperature is normally 37 oC the scale is only a few degrees below and above 37oC. It has a constriction which prevents mercury from going back after expansion for convenient reading of temperature.
This thermometer has a narrow bore for greater sensitivity and accuracy.
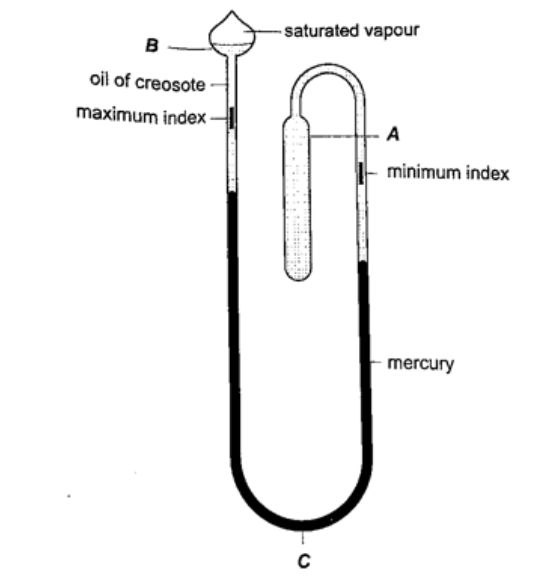
3. Six’s maximum and minimum thermometer– it is used to measure temperature of surroundings of an area or a place. It can record both maximum and minimum temperatures attained.
Consists of a large bulb (A) containing oil of creosote connected to U-shaped stem which connects to a second bulb (B) containing the same liquid. The base (C) contains a thin thread of mercury. The range of this thermometer is between -20 oC and 50 oC. After each reading the indices are pulled down to the level of mercury by use of a magnet.
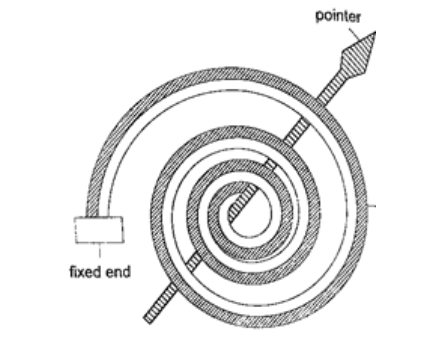
4. Bimetallic thermometer– it is made up of a bimetallic strip with one end fixed and the other connected to a pointer. Metals used are usually brass and invar. As temperatures increase the strip unwinds and moves the pointer over a calibrated scale. It is used to measure high temperatures.
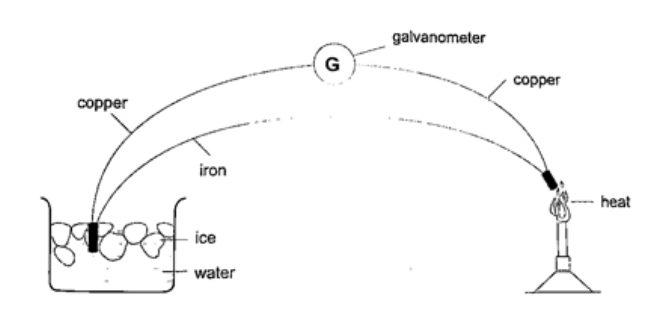
5. Thermocouple thermometer– thermocouple is a junction made of copper and iron looped at both ends. In practice a sensitive millivoltmeter is used instead of a galvanometer. A cold junction is maintained in melting ice (0oC) while the other junction is heated steadily. This thermometer does not apply the principle of expansion.
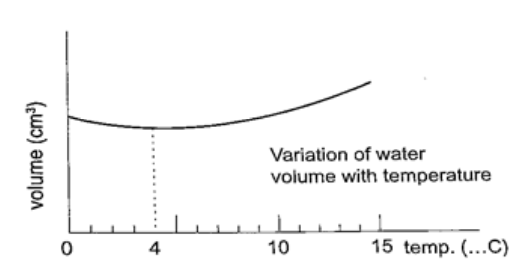
Unusual expansion of water.
If water is heated let’s say from -15oC it expands normally like any solid but only up to 0oC.
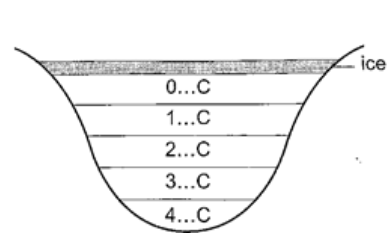
At this point it starts to melt and it contracts. This contraction will be observed up to 4oC. When heated further water starts to expand up to boiling point. This is the unusual expansion of water. This makes the top of water to freeze (0oC) in temperate countries allowing the one below to remain liquid (4oC). This supports marine life during winter. Molecules and heat.1. Solids – when heated molecules in solids absorb heat energy and vibrate. They push against one another and this causes expansion. Further expansion may result to collapse as melting in ice. 2. Liquids – besides vibrating particles in a liquid move short distances. As they move they collide by hitting each other and this results to more expansion. For boiling to occur molecules absorb enough energy to be able to escape from the liquid. 3. Gases – individual particles are free of one another and in rapid motion. When heated there are collisions with the walls of the container. This results to high pressure in the container.
1 Comment
|
Archives
December 2024
Categories
All
Physics notes form 1 to 4
|
||||||||||||||||||||||||
We Would Love to Have You Visit Soon! |
Hours24 HR Service
|
Telephone0728 450425
|
|
8-4-4 materialsLevels
Subjects
|
cbc materialsE.C.D.E
Lower Primary
Upper Primary
Lower Secondary
Upper Secondary
|
teacher support
Other Blogs
|

 RSS Feed
RSS Feed