|
Just incase any document encrypted, use this password 'siri2u!'
FORM 1
FORM 2
FORM 3
FORM 4
PHYSICS SYLLABUS TOPICS
2 Comments
Specific ObjectivesBy the end of this topic, the learner should be able to:
ELECTROSTATICS I (12 LESSONS)Sub-Topics Covered:
ELECROSTATICS I.Some substances get charged when rubbed against other substances i.e. nylon, plastic, paper etc. the charge acquired stays within the body i.e. it does not move and therefore known as electrostatic charge or static electricity.The law of charges – types of chargesThere are two types of charges i.e. negative and positive charges. The negative charge consists of electrons which are mobile. The law of charges in summary states that “like charges repel, unlike charges attract’’.Just like in magnetism attraction is not a sure way of testing for charge but repulsion because it will only occur if the bodies are similarly charged. Charges, atoms and electronsThe atom is made up of a central part called the nucleus, containing positively charged ions called protons and outwardly surrounded by negatively charged electrons.The nucleus also contain the particles called neutrons which are not charged. When an atom is not charged the number of protons equals the number of electrons. When a material is rubbed with another i.e. acetate with silk, electrons are transferred from one body to another. The body accepting or receiving electrons becomes negatively charged while the one donating or losing electrons becomes positively charged. Protons and neutrons in the nucleus do not move. The SI unit for charge is the Coulomb (Coul.)
1 Coul. = charge on 6.25 × 1018 electrons.
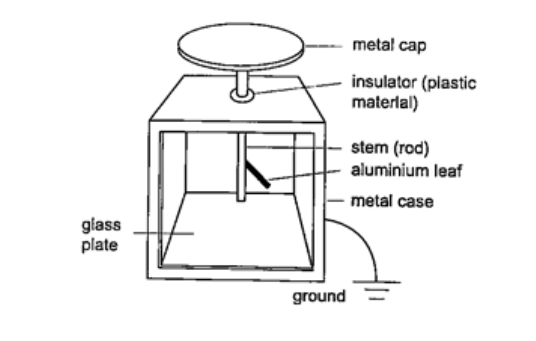
Charge on one electron = 1.60 × 10-19 Coul. 1 Coul. = 1 Ampere second (As). The leaf electroscopeThis is a sensitive instrument for measuring charge. It consists of a metal cap connected to a stem whose lower part is flattened into a plate with a thin strip of aluminium foil attached to it.The plate and the leaf are enclosed in a metal casing which is earthed. The sides of the metal are made of glass to allow the leaf to be seen.
Other leaf electroscopes are made using gold strips and are referred to as gold leaf electroscope.
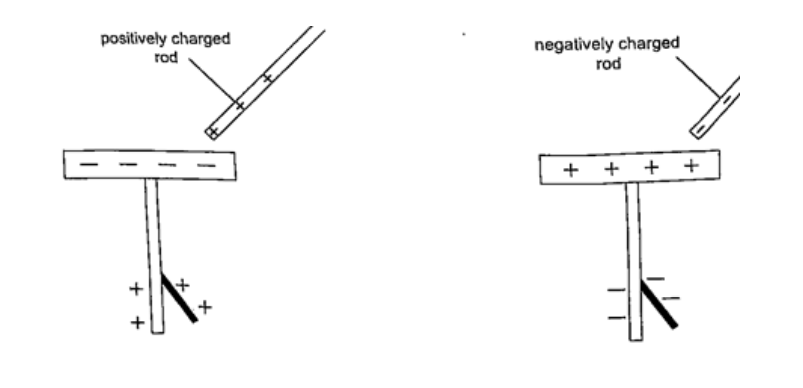
Charging and discharging an electroscopeWhen a charged body is brought near the cap of the electroscope the leaf diverges, and when removed it collapses. When a negatively charged body is brought near the metal cap electrons are repelled from the cap to the lower parts of the stem and the leaf.This concentration of negative charges makes the leaf to diverge. Similarly when a positively charged body comes near the metal cap the electrons are attracted by the protons and move up the stem, leaving a high concentration of positive charges which make the leaf to diverge.
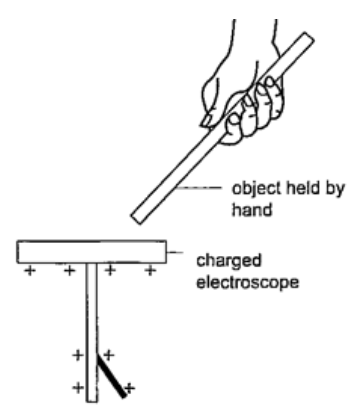
If you touch the metal cap with your finger the leaf collapses showing that the charges have been discharged through your body. An uncharged body will always cause the leaf of a charged electroscope to collapse regardless of the charge on the electroscope. This shows that charge moves from the charged electroscope to the uncharged body.
Conductors and insulatorsConductors are those substances which allow easy passage of a charge. Insulators do not allow a charge to pass through easily. A charged electroscope can be used to classify objects into conductors and insulators.Experiment: Arranging objects into conductors and insulators. Procedure
Examples of Conductors and Insulators
We have seen that when a charged body is brought near a leaf electroscope, charges are transferred to the electroscope and the leaf diverges. This method of transferring charge without actual contact is called induction.
Uses of the electroscope
Applications of electrostatic charges.
Dangers of electrostaticsAs liquid flows through a pipe its molecules get charged due to rubbing against inner surface. If the liquid is flammable then this can cause sparks and explode. The same happens to fuels carried in plastic cans therefore it is advisable to carry fuel in metallic cans to leak out the continuously produced charges. |
Archives
December 2024
Categories
All
Physics notes form 1 to 4
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |

 RSS Feed
RSS Feed

