|
60 cm3 of oxygen gas diffused through a porous partition in 50 seconds. How long would it take 60cm3 of sulphur (IV) oxide gas to diffuse through the same partition under the same conditions? (S = 32.0, 0 = 16.0)
0 Comments
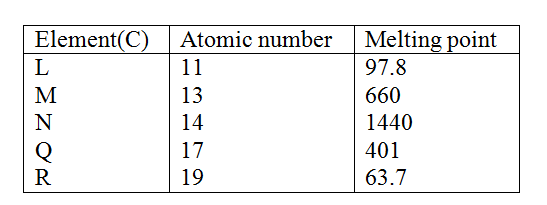
Study the information in the table below and answer the questions that follow. The letters do not represent the symbols of the elements.
a) Write the electrons arrangement for the atom formed by elements and M and Q
b) Select an element which is
d) Element R lodes its outermost electron more readily than I. Explain e) Using dots(.) and crosses (x) to represent outermost electrons show bonding in the compound formed elements N and Q. f) Explain why the melting point elements M is higher than that of element . g) Describe how a solid mixture of sulphate of R and lead sulphate can be separated into solid samples.
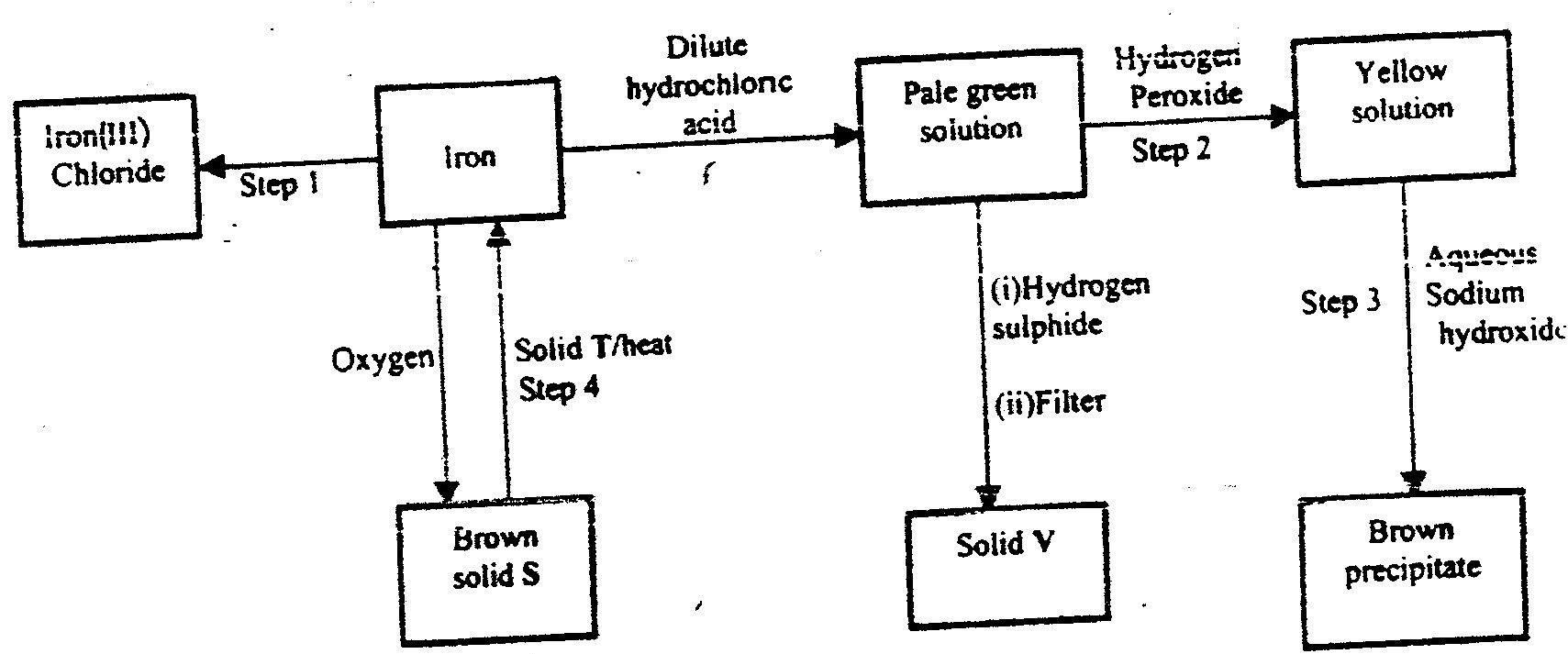
a) The flow chart below shows a sequence of reactions starting with.
Study and answer the questions that follow.
Cu = 63.5, Fe = 56.0
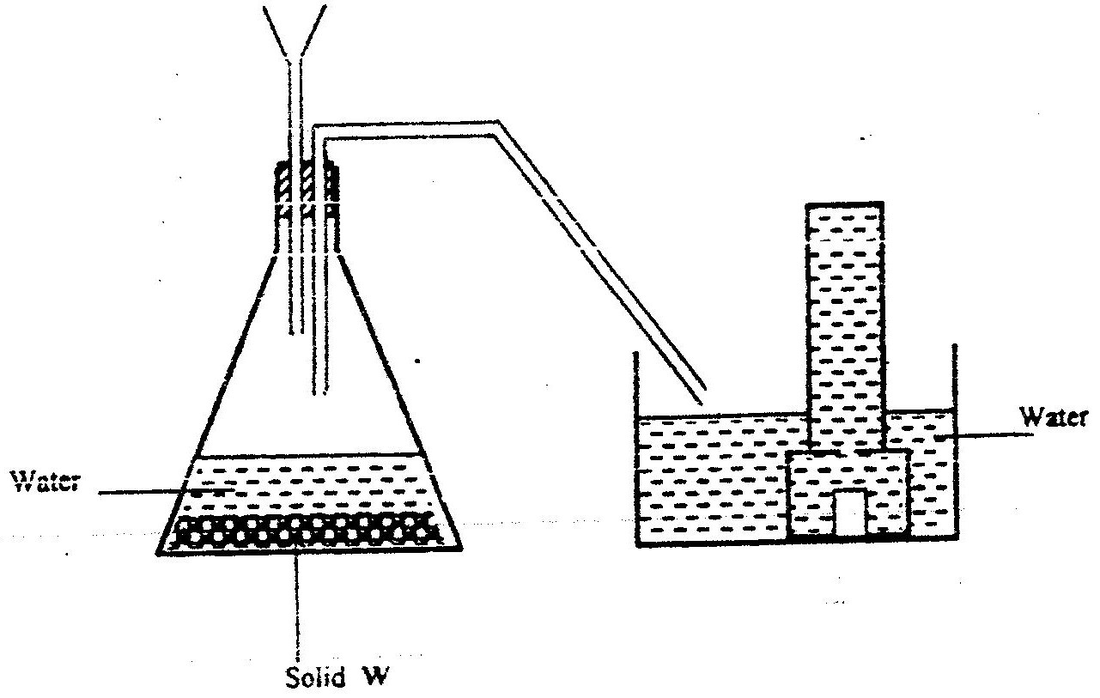
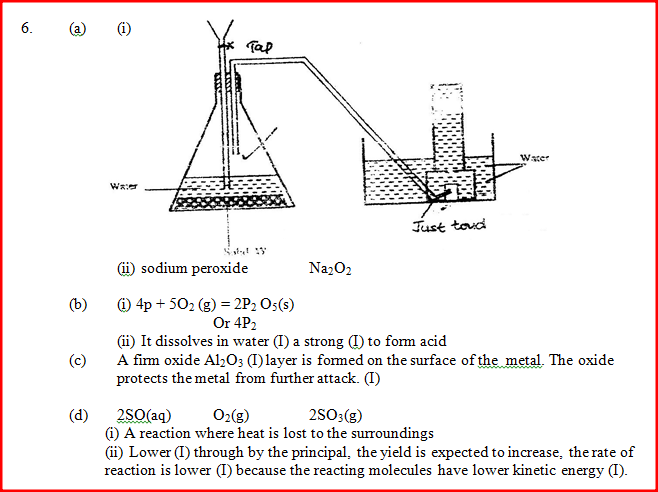
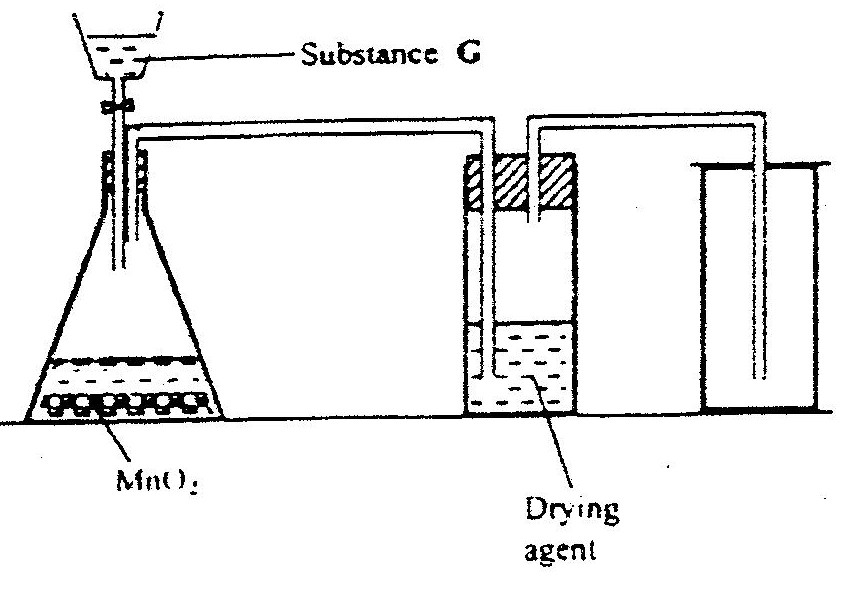
a) The diagram below shows a set –up used by as a student in an attempt to prepare and collect oxygen gas
c) The reaction between sulphur dioxide and oxygen to form trioxide in the contact process in exothermic. Factory manufacturing sulphuric acid by contact process produces 350kg of sulphur trioxide per day (conditions) for the reaction catalyst. 2 atmospheres pressure and temperatures between. (400 – 5000C) i) What is meant by an exothermic reaction? ii) How would the yield per day of sulphur trioxide be affected Temperatures lower than 40000C are used? Explain.
a) When an organic compound Y is reacted with aqueous sodium carbonate, it produces carbon dioxide reacts with propanol to form a sweet smelling compound Z whose formula is.
c) The scheme below was used to prepare a cleaning agent. Study and answer the questions that follow.
i) What name is given to the type of cleaning agent prepared by the method shown in the scheme?
ii) Name one chemical substance added in step II iii) What is the purpose of adding the elements substance names in C(ii) above. iv) Explain how an aqueous solution of the cleansing removes oil from utensils during washing.
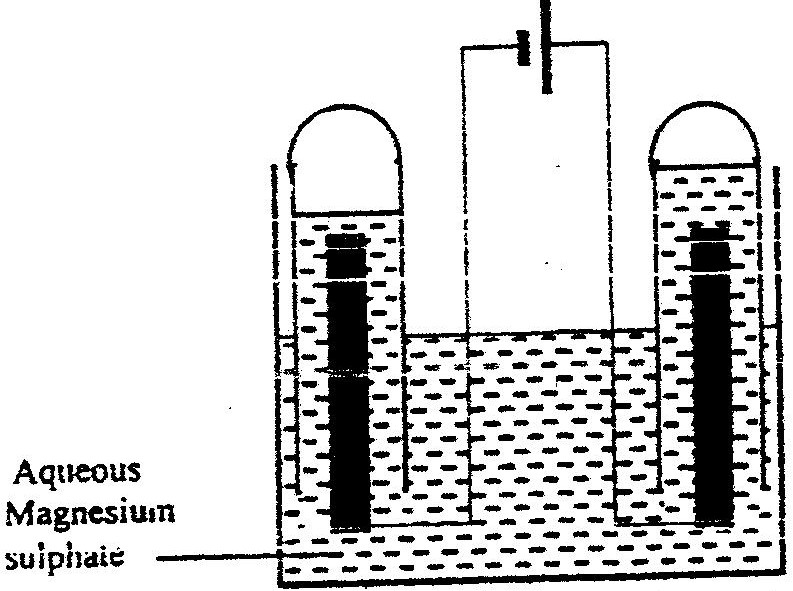
The set – up below was used during the electrolysis of aqueous magnesium sulphate using inert electrodes
i) Name a suitable pair of electrodes for this experiment ii) Identify the ions and cations in the solution iii) On the diagram label the cathode iv) Write ionic equations for the reactions that took place at the anode. v) Explain the change that occurred to the concentration of magnesium sulphate solution during the experience. vi) During the electolysis a current of 2 amperes was passed through the solution for 4 hours. Calculate the volume of the gas produced at the anode.(1 faraday 96500 coulombs and volume of a gas at room temperature is 24000cm3) vii) One of the uses of electrolysis is electroplating What is meant by electroplating? Give tow reasons why electroplating is necessary.
The table below gives the volume of the gas provided when different volumes of 2M hydrochloric were reacted with 0.6g of magnesium powder at room temperature
(a) Write an equation fro the reaction between magnesium and hydrochloric acid
(b) On the grid provided plot a graph of the volume of gas produced (vertical axis), against the volume of acid added (Note the reaction comes to completion, the volume of the gas produced directly proportional to completion, the acid added). From the graph determine c)
e) Given that one mole of the gas occupied 24000cm3 at room temperature. The table below shows the PH values of solutions I, II, III and IV.
(a) Which solution is likely to be that of calcium hydroxide?
(b) Select the solution in which a sample of aluminium oxide is likely to dissolve. Give reason for your answer
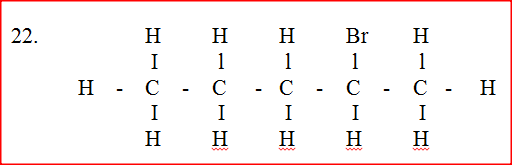
Bromine reacts with ethane as shown below
(a) What condition is necessary for this reaction to occur?
(b) Identify the bonds which are broken and those that are formed
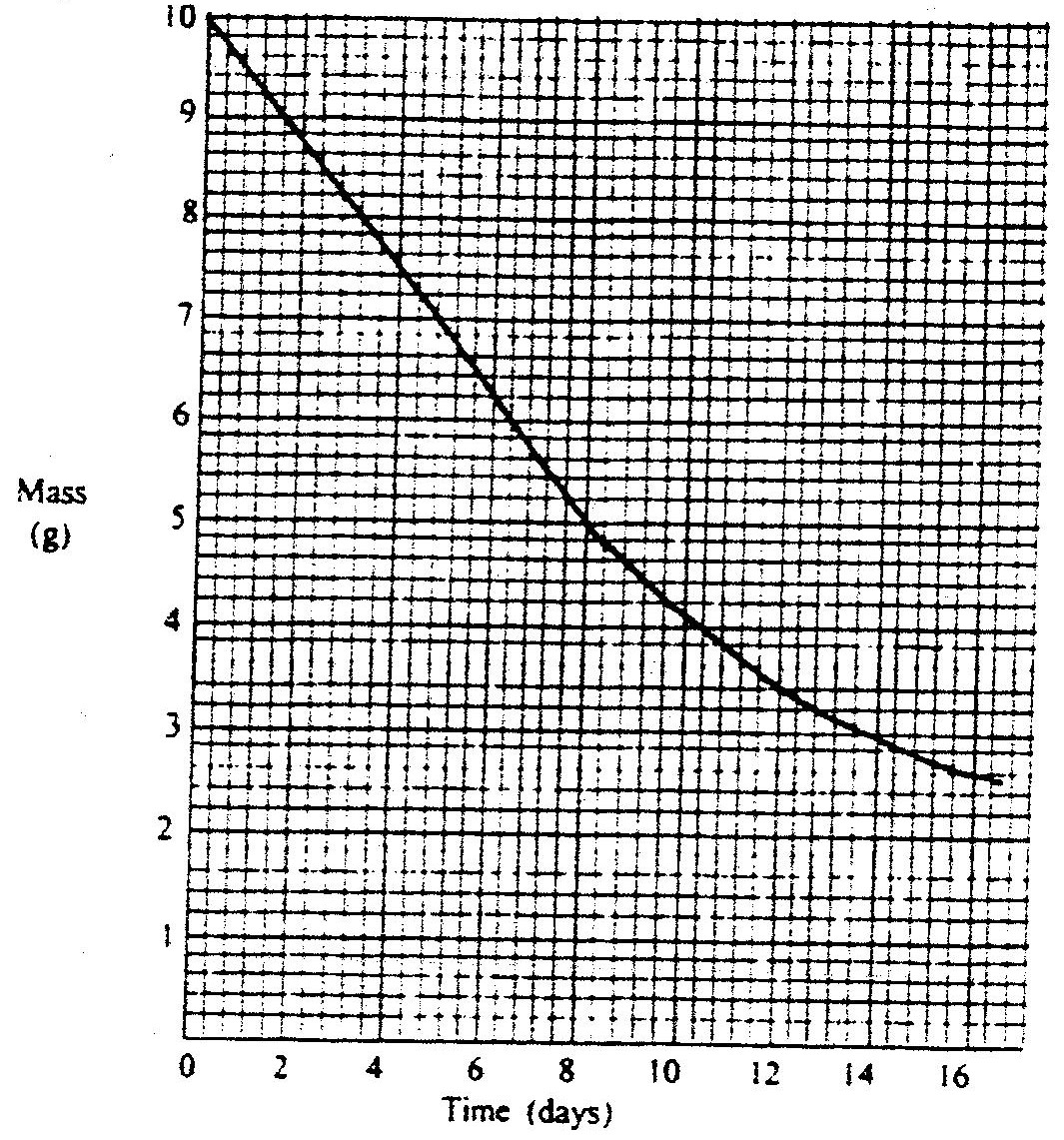
The graph below shows the mass of a radioactive isotope plotted against time
(a) Using the graph, determine the half – life of the isotope
(b) Calculate the mass of the isotope present after 32 days
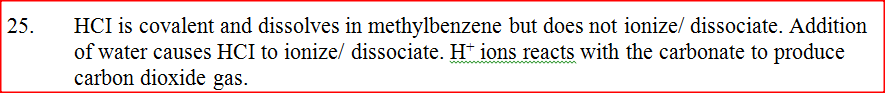
When solid magnesium carbonate was added to a solution of hydrogen chloride in methylbenzene, there was no apparent reaction. On addition of water to the resulting mixture, there was vigorous effervescence. Explain these observations
One of the fuels associated with crude oil is natural gas. Name the main constituent of natural gas and write an equation for its complete combustion
Name: Equation:
When excess carbon monoxide gas was passed over heated lead (II) oxide in combustion tube, lead (II) oxide was reduced
(a) Write an equation for the reaction, which took place (b) What observation was made in the combustion tube when the reaction was complete? (c) Name another gas, which could be used to reduce lead (II) oxide
Expected Response
(a) Pb(s) + CO(g) →Pb(s) + CO2(g)
(b) Silvery white or grey metal/ shiny grey metal (c) Hydrogen gas / ammonia
Compound L reacts with hydrogen bromide gas to give another compound whose structure is
(a) Give the structural formula and name of compound L
(b) Write an equation for the reaction, which takes place between ethane and excess chlorine gas
State and explain one disadvantages of using hard water in boilers
Expected Response
On boiling hard water deposits CaCO3 which forma fur/ scales in the boiler. This reduces conductivity
Under certain conditions, carbon dioxide reacts with water to form methanol (CH3OH) and oxygen as shown below
What would be the effect on the yield of methanol if the temperature of the reaction mixture is increased? Explain
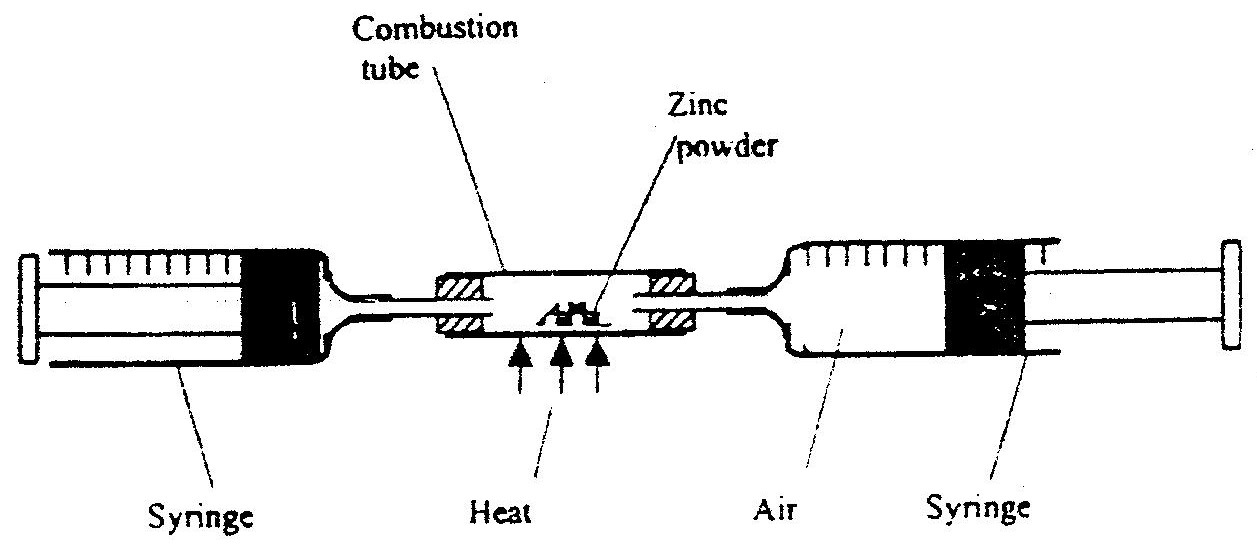
In an experiment a certain volume of air was passed from syringe to syringe over heated zinc powder as shown in the diagram below
The experiment was repeated using excess magnesium powder. In which of two experiments was the change in volume of the air greatest. (Give reasons)
The set up below was used to investigate some properties of two gases M and N
The diagram below shows a set up for the laboratory preparation and collection of dry chlorine gas
Expected Response
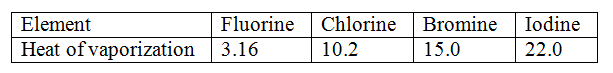
Molar heats of vaporization increase down the group with increase in van der waals forces of attraction as the number of electrons increase/increases in size/ volume/mass/energy levels/ density/ No. of protons (atomic number)
Use the information in the table below to answer the questions that follows
Explain the trend in the molar heats of vaporization
Expected Response
Change was greatest with magnesium. Both react with oxygen to form oxides but magnesium reacts with nitrogen to form a nitrite
The set up below was used to investigate some properties of two gases M and N
When beaker A was filed with gas M, the level of water in the glass tube rose to
point II. When the experiment was repeated using gas N, the level of water dropped to point III. Explain these observations.
Use the information below to answer the question that follows
(a) A few drops of freshly prepared iron (II) sulphate solution were added to potassium nitrate solution in a test – tube. Concentrated sulphuric acid was then carefully added to the mixture. State the observation that were made
(b) Write an equation for the reaction that occurs when solid potassium nitrate is strongly heated
Use the cell representation below to answer the questions that follow
In an attempt to prepare sulphur dioxide gas, dilute sulphuric acid was reacted with barium sulphuric. The yield of sulphur dioxide was found to be negligible explain
D gm of potassium hydroxide were dissolved in distilled water to make 100cm3 of solution. 50cm3 of the solution- required 50cm3 of 2 M nitric acid for complete neutralization. Calculate the mass D, of potassium hydroxide (KOH)(aq) + HNO3(aq) →KNO3 (aq) + H2O (I) Relative formula mass of KOH = 56 |
Chemistry Topics
All
Archives
December 2024
|
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |



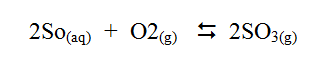
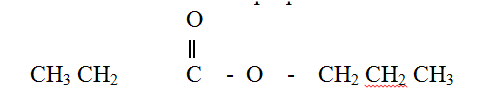





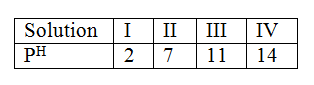

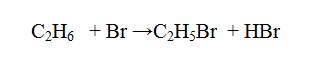

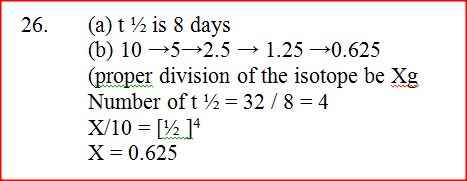
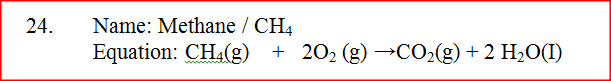
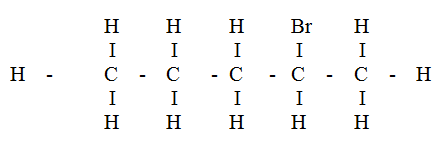



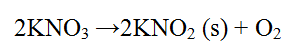
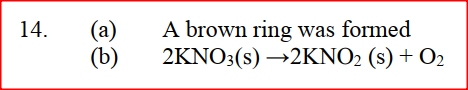





 RSS Feed
RSS Feed

