|
Write the formula of sulphide of an element C, whose atomic number is 5. (C is not the actual symbol of the element)
0 Comments
Give the name of the product formed when magnesium reacts with phosphorus.
ANSWERS
The table below gives the energy required to remove the outermost electron for some group I elements
Arrange the elements in order of their reactivity starting with the most reactive
Expected Response
ANSWERS
(a) Energy required to remove 1 mole of electrons from 1 mole of gaseous atoms
(b) B 418??? It loses electrons most readily Reject lowest i.e. Mg (HCO3) 2 aq →MgCS O3 + H2O + CO2 (g)
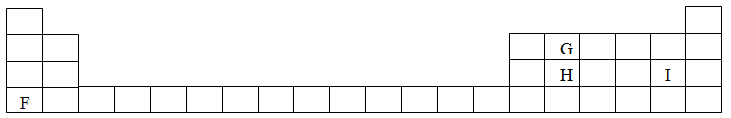
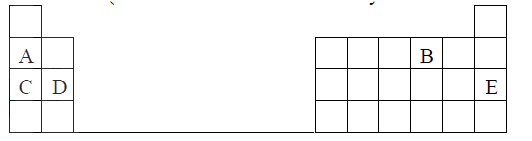
The grid below shows part of a periodic table. The letters do not represent the actual symbols of the elements
Expected Response
The grid below is part of the periodic table. Use it to answer the questions that follow, (the letters are not the actual symbols of the elements).
a) Indicate on the grid the position of an element represented by letter V whose atomic number is 14.
b) Select a letter which reaction between Q and T.
ANSWERS
(a) Metallic bonding
(b) Group 1 Each atom contains one electron in its outer most energy level
Both chlorine and iodine are halogens.
a) What are halogens? b) In terms of structure and bonding, explain why the boiling point of chlorine is lower than that of iodine.
ANSWERS
(a) Group (VIII) elements
(b) Chlorine molecule is smaller and the strength of Vander Waals forces between molecules of chlorine is weak as compared to iodine.
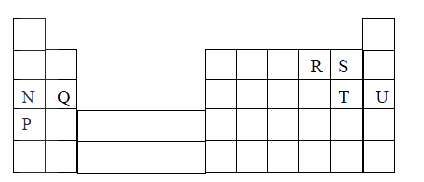
a) Distinguish between isotopes and allotropes.
b) The chart below is part of the periodic table. Study it and answer the questions that follow.(The letters are not the actual symbols of the elements).
i) Select the element in period three which has the shortest atomic radius. Give a reason for you answer.
ii) Element F has the electronic structure, 2.8.18.4 on the chart above, indicate the position of element F. iii) State one use of the elements of which E is a member. iv) Write an equation to show the action of heat on the nitrate of element C. c) When 3 litres of chlorine gas were completely reacted with element D, 11.875g of the product were formed. Determine the relative atomic mass of element D. (Atomic mass of chlorine = 35.5; molar gas volume = 24litres)
The atomic numbers of elements C and D are 19 and 9 respectively. State and explain the electrical conductivity of the compound CD in:
(a) Solid state (b) aqueous state.
ANSWERS
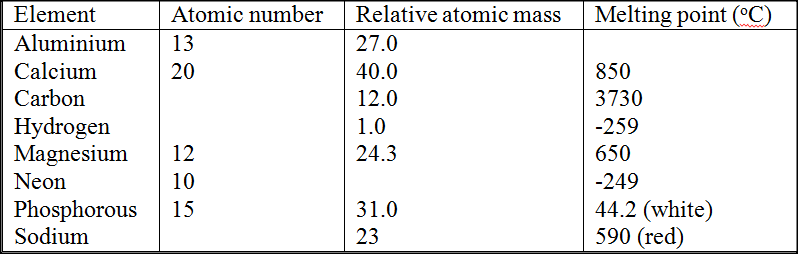
Study the table below and answer the questions that follow:
a) Complete the table by filling in this missing atomic numbers and atomic mass. (2mks) b) Write the electron arrangement for the following ions (2mks) Ca+ P3+ c) What is the melting point of hydrogen in degrees Kelvin? (1mk) d) Which of the allotropes of phosphorous ha a higher density? Explain (2mks) e) The mass numbers of three isotopes of magnesium are 24, 25 and 26. What is the mass number of the most abundant isotope of magnesium? Explain (2mks) f) Give the formula of the compound formed between aluminium and carbon.(1mks) g) Explain the difference in the melting points of magnesium and sodium.(2mks)
ANSWERS
Four metal F,G,H and J were each separately added to cold water, and steam. The table below is a summary of the observations made and the formulae of the hydroxides formed.
a) Which two elements are likely to be in the same group of the periodic table?
b) Arrange the metals in the order of their reactivity starting with the most reactive
answers
answer
Explain why the reactivity of group (VII) elements decreases down the group
answer
An oxide of element F has the formula F2O5
(a) Determine the oxidation state of F (b) In which group of the periodic- table is element F
Use the reactions given below to answer the questions that follow. The letters do not represent the actual symbols of the elements
(a) What name is given to the type of reaction given above?
(b) Arrange the elements D, E, F and G in the order of their reactivity starting with the most reactive (c) Complete the equation below
Name One property of neon that makes it possible to be used in electric lamps
ANSWERS
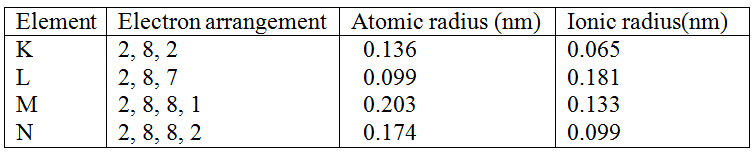
The table below gives information on four elements by letters K, L, M and N. Study it and answer the questions that follow. The letters do not represent the actual symbols of the elements. a) Which two elements have two similar properties? Explain (2mks) With reference to atomic number of one, explain why hydrogen can be placed in either group I and VII of the periodic table. K.N.E.C Marking Scheme
Hydrogen forms compounds by losing one electron like group I elements or by gaining one electron like group VII element /Hydrogen has one electron in outermost shell. |
Chemistry Topics
All
Archives
December 2024
|
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |

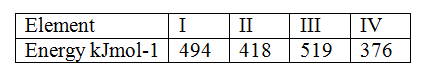
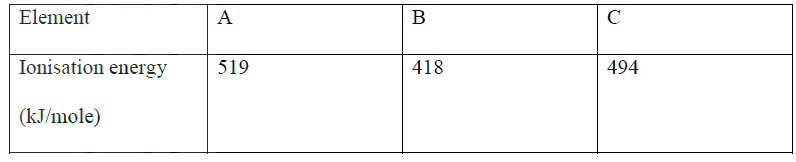



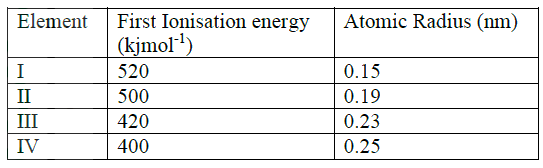

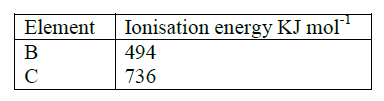




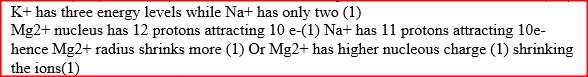
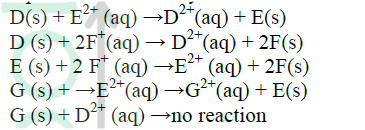
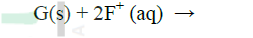




 RSS Feed
RSS Feed

