KCSE CHEMISTRY QUESTIONS AND ANSWERS PER TOPIC
|
The table below shows the volumes of nitrogen dioxide gas produced when different volume of 1M nitric acid were each reacted with 2.07 g of lead at room temperature.
a) Give a reason why nitric acid is not used to prepare hydrogen gas. b) Explain how the rate of the reaction between lead and nitric acid would be affected if the temperature of the reaction mixture was raised. c) On the grid provided below, plot a graph of the volume of the gas produced (Vertical axis) against volume of acid.
d) Using the graph, determine the volume of
:i) Nitrogen dioxide produced when 30cm3 of 1 M nitric acid were reacted with 2.07 g of lead ii) 1M nitric acid which would react completely with 2.07g of lead. e) Using the answer in d(i) above, determine: i) The volume of 1M nitric acid that would react completely with one mole of lead (Pb=207) ii) The volume of nitrogen dioxide gas produced when one mole of lead reacts with excess 1 M nitric room temperature. f) Calculate the number of moles of: i) 1M nitric acid that reacted with one mole of lead ii) nitrogen dioxide produced when one mole of lead were reacted with excess nitric acid. (Molar gas volume of 2400cm3) g) Using the answers obtained in f (i) and (ii) above, write the equation for the reaction between lead and nitric acid given that one mole of lead nitrate and two moles of water were also produced.
0 Comments
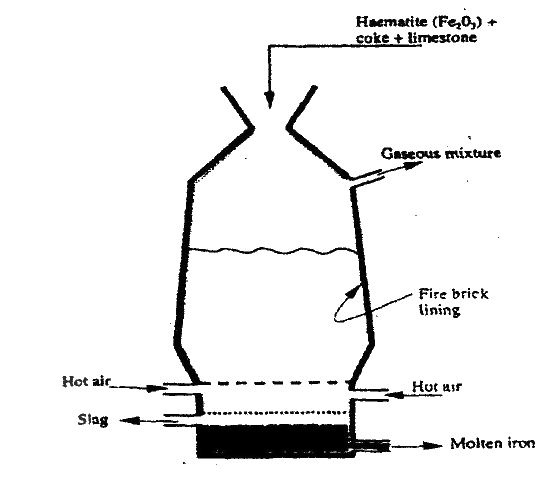
The extraction of iron from its ores takes place in the blast furnace. Study it and answer the questions that follow.
a) Name
(i) One of the substances in the slag (ii) Another iron ore material used in the blast furnance. (iii) One gas which is recycled. b) Describe the process which leacd to the formation of iron in the blast furnace c) State the purpose of limestone in the blast furnace. d) Give a reason why the melting point of the iron obtained from the blast furnace is 12000 C while tat of pure iron is 15350C (e) State two uses of steel
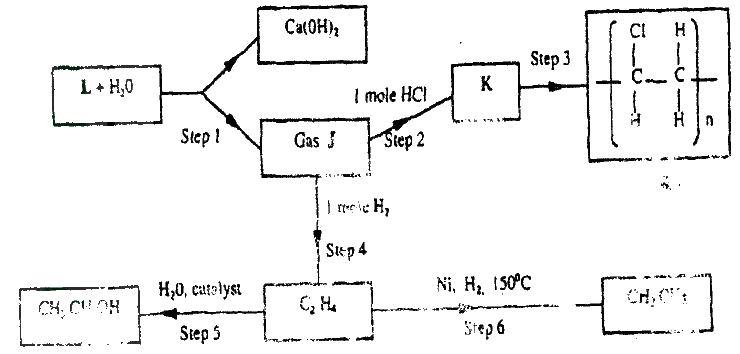
a) What name is given to a compound that contains carbon and hydrogen only?
b) Hexane is a compound containing carbon and hydrogen. (i) What method is used to obtain hexane from crude oil? (ii) State one use of hexane c) Study the flow chart below and answer the questions that follow.
(i) Identify reagent L.
(ii) Name the catalyst used in step 5. (iii) Draw the structural formula of gas J. (iv) What name is given to the process that takes place in step 5? d) (i) write the equation for the reaction between aqueous sodium hydroxide and aqueous ethanoic acid. (ii) Explain why the reaction between 1g of sodium carbonate and 2M hydrochloric acid is faster than the reaction between 1g of sodium carbonate and 2M ethanoic acid.
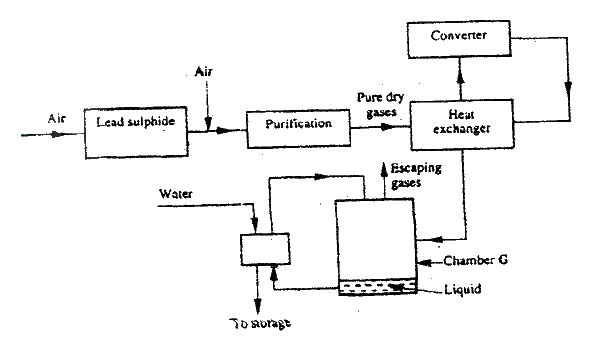
a) The diagram below shows some processes that take place during the industrial manufacture of sulphuric acid.
(i) Write the equation for the reaction in which sulphur dioxide gas is produced.
(ii) Why is it necessary to keep the gases pure and dry ? (iii) Describe the process that takes place in chamber G. (iv) Name the gases that escape into the environment. (v) State and explain the harmful effect on the environment of one of the gases named in (iv) above (vi) Give one reason why it is necessary to use a pressure of 2-3 atmospheres and not more. b) (i) Complete the table below to show the observations made when concentrated sulphuric acid is added to the substances shown.
(ii) Give reasons for the observations made using:
I iron fillings II Crystals of white sugar. (c) Name one fertilizer made from sulphuric acid. (d) Suggest a reason why BaSO4(a pigment made from sulphuric acid) would be suitable in making paint for cars.
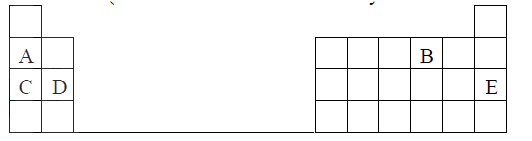
a) Distinguish between isotopes and allotropes.
b) The chart below is part of the periodic table. Study it and answer the questions that follow.(The letters are not the actual symbols of the elements).
i) Select the element in period three which has the shortest atomic radius. Give a reason for you answer.
ii) Element F has the electronic structure, 2.8.18.4 on the chart above, indicate the position of element F. iii) State one use of the elements of which E is a member. iv) Write an equation to show the action of heat on the nitrate of element C. c) When 3 litres of chlorine gas were completely reacted with element D, 11.875g of the product were formed. Determine the relative atomic mass of element D. (Atomic mass of chlorine = 35.5; molar gas volume = 24litres)
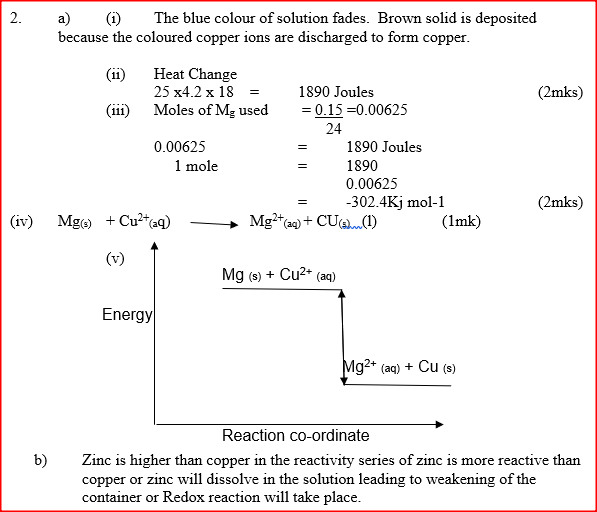
a) In an experiment to determine the molar heat of reaction when magnesium displaces copper ,0.15g of magnesium powder were added to 25.0cm3 of 2.0M copper (II) chloride solution. The temperature of copper (II) chloride solution was 25°C.While that of the mixture was 43°C.
i) Other than increase in temperature, state and explain the observations which were made during the reaction. ii) Calculate the heat change during the reaction (specific heat capacity of the solution = 4.2jg-1k-1and the density of the solution = 1g/cm3 iii) Determine the molar heat of displacement of copper by magnesium.(Mg=24.0). iv) Write the ionic equation for the reaction. v) Sketch an energy level diagram for the reaction. b) Use the reduction potentials given below to explain why a solution containing copper ions should not be stored in a container made of zinc.
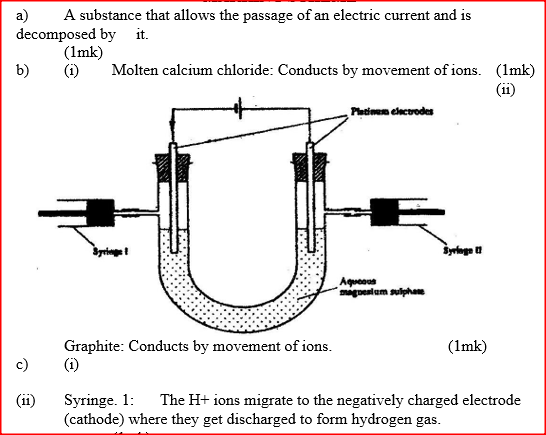
a) What is an electrolyte?
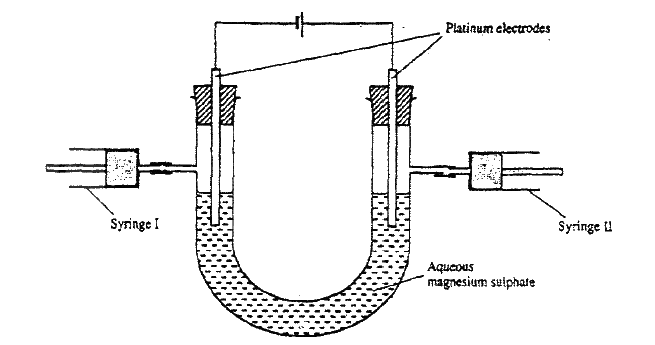
b) State how the following substances conduct electricity. i) Molten calcium chloride ii) Graphite. c) The diagram below shows a set up that was used to electrolyse aqueous magnesium sulphate.
i) On the diagram above, using an arrow, show the direction of flow of electrons.
ii) Identify the syringe in which hydrogen gas would be collected. Explain d) Explain why the concentration of magnesium sulphate was found to have increased at the end of the experiment. e) During the electrolysis, a current of 0.72A was passed through the electrolyte for 15 munities. Calculate the volume of gas produced at the anode.(1 Faraday = 96 500 coulombs; molar gas volume is 24000cm3 at room temperature).
ANSWERS
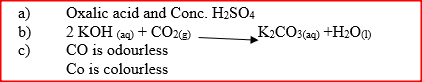
a)Conc. H2SO4
b)Heat the solution to concentrate it. Allow for crystal for form. Filter. c)Anhydrous copper(II) Sulphate
ANSWERS
a)They gain energy and move faster. The intermolecular distance increases.
b)XY c)The energy supplied changes molecules of water from liquid to Gaseous state.
ANSWERS
a)V1 and V3
b)Add petrol to the mixture. Filter. V2 is the residue. Filtrate is V4 Distill the filtrate.
Explain why the boiling point of ethanol is higher than that of hexane.
(Relative molecular mass of ethanol is46 while that of hexane is 86)
ANSWERS
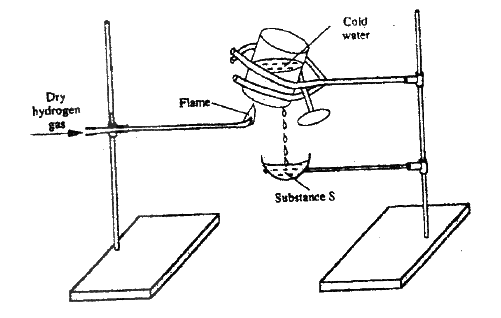
The diagram below represents part of a set – up used to prepare and collect gas T.
a) Name two reagents that are reacted to produce both carbon (IV) oxide and carbon(II) oxide.
b) Write the equation for the reaction which takes place in the wash bottles. c) Give a reason why carbon(II) oxide is not easily detected.
(a) Explain why the metals magnesium and aluminium are good conductors of electricity.
b) Other than cost, give two reasons why aluminium is used for making electric cables while magnesium is not
ANSWERS
a)They have delocalized valency electrons
b)Aluminium has three delocalized electrons. It is resistant to corrosion
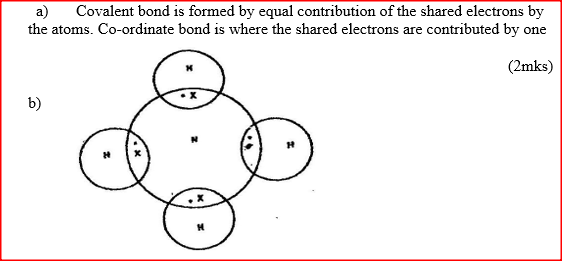
a) Distinguish between a covalent bond and a co-ordinate bond
b) Draw a diagram to show bonding in an ammonium ion.
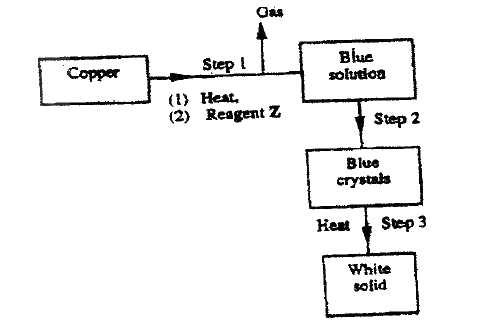
a) starting from solid magnesium hydroxide.
b) Give one use of magnesium hydroxide.
ANSWERS
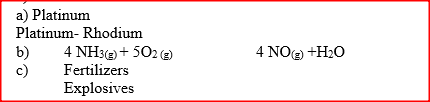
The first step in the industrial manufacture of nitric acid is the catalytic oxidation of ammonia gas.
(a) What is the name of the catalyst used? (b) Write the equation for the catalytic oxidation of ammonia gas. (c) Nitric acid is used to make ammonium nitrate, state two uses of ammonium nitrate.
When hydrogen sulphide gas was bubbled into an aqueous solution of iron (III) chloride, a yellow precipitate was deposited.
a) The standard reduction potential for Fe2+ (aq) is -0.44 volts. Select the element which would best protect iron from rusting. b) Write an equation for the reaction that took place. c) What type of reaction was undergone by hydrogen by hydrogen sulphide in this reaction?
Study the standard reduction potential given and answer the questions that follow.
(The letters are not the actual symbols of the elements).
a) The standard reduction potential for Fe 2+(aq) is -0.44 volts. Select the element which would best protect iron from rusting.
b) Calculate the E ø value for the cell represented as M(s) / M2+ (aq) // P+(aq) /P(s).
Below is a list of oxides.MgO, N2O, K2O, CaO, and Al2O3.
Select: a) A neutral oxide b) A highly water soluble basic oxide c) An oxide which can react with both sodium hydroxide solution and dilute hydrochloric acid.
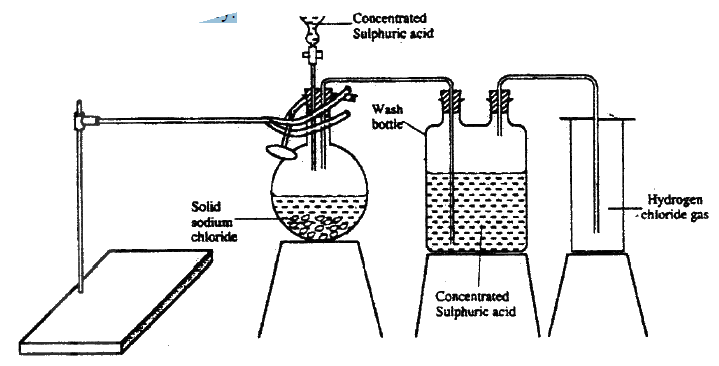
The diagram below represents the set-up that was used to prepare and collect hydrogen chloride gas in the laboratory.
a) State the purpose of concentrated sulphuric acid in the wash bottle.
b) Write an equation for the reaction between dry hydrogen chloride gas and heated iron c) Hydrogen chloride gas is dissolved in water to make hydrochloric acid. State one use of hydrochloric acid.
The table below shows the relative atomic masses and the percentage abundance of the isotopes L1 of element L.
a) water from a town in Kenya is suspected to contain chloride ions but not sulphate ions.
Describe how the presence of the chloride ions in the water can be shown. b) State one advantage of drinking hard water rather than soft water.
ANSWERS
a)
Acidify water with nitric acid. Add aqueous lead nitrate. Formation of white PPt shows presence of CT b)provides essential minerals e.g Ca2+ |
Chemistry Topics
All
Archives
December 2024
|
We Would Love to Have You Visit Soon! |
Hours24 HR Service
|
Telephone0728 450425
|
|
8-4-4 materialsLevels
Subjects
|
cbc materialsE.C.D.E
Lower Primary
Upper Primary
Lower Secondary
Upper Secondary
|
teacher support
Other Blogs
|
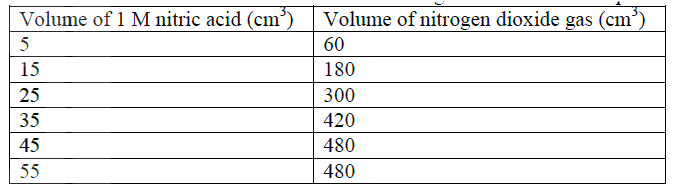



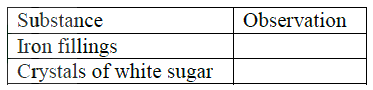


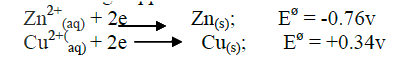



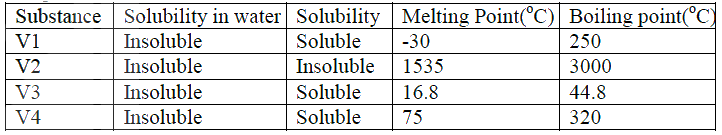



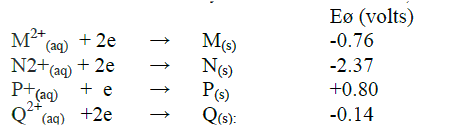



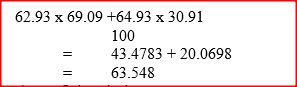

 RSS Feed
RSS Feed