|
Explain the observation made when chlorine gas is passed through a solution of potassium iodide.
0 Comments
20 cm3 of ethanoic acid was diluted to 400 cm3 of solution. Calculate the concentration of the solution in moles per litre. (C = 12.0 ; H = 1.0 ; 0 =16.0) (Density of ethanoic acid = 1.05 g/cm3)
Copper(II) ions react with excess aqueous ammonia to form a complex ion.
(a) (i) Write an equation for the reaction that forms the complex ion. (ii) Name the complex ion. (b) Explain why CH4 is not acidic while HCl is acidic yet both compounds contain hydrogen.
(a) State Charles' Law.
(b) Explain why the pressure of a fixed mass of a gas increases, when the volume of the gas is reduced at constant temperature.
ANSWERS
(a)The volume of a fixed mass of a gas is directly proportional to the absolute temperature at constant pressure.
(b)As the volume decreases, there is increased bombardment / collisions of the molecules against the walls of the container, hence increased pressure.
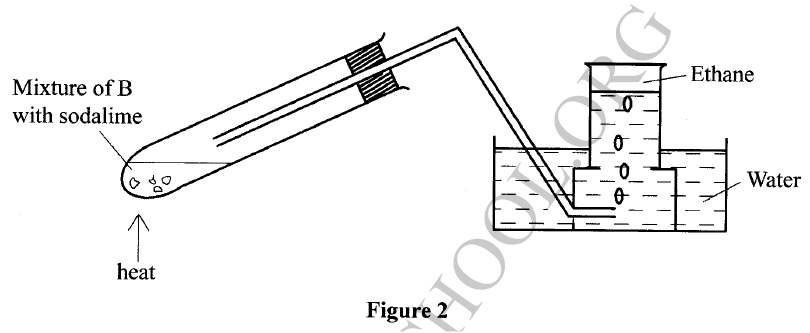
The set-up in Figure 2 was used to prepare a sample of ethane gas. Study it and answer the questions that follow.
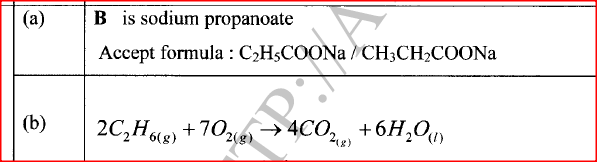
(a) Name B
(b) Write an equation for the complete combustion of ethane. (c) State one use of ethane.
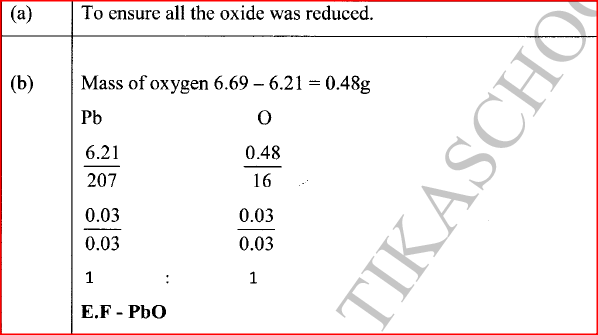
The empirical formula of lead(II) oxide was determined by passing excess dry hydrogen gas over 6.69g of heated lead(II) oxide.
(a) What was the purpose of using excess dry hydrogen gas? (b) The mass of lead was found to he 6.21g. Determine the empirical formula of the oxide. (Pb = 207.0 0 = 16.0)
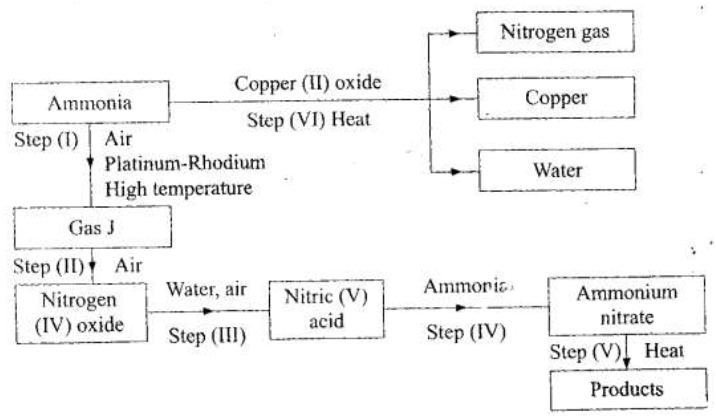
(a) Describe the process by which Nitrogen is obtained from air on a large scale.
(b) Study the flow chart below and answer the questions that follow.
(i) Identify gas J.
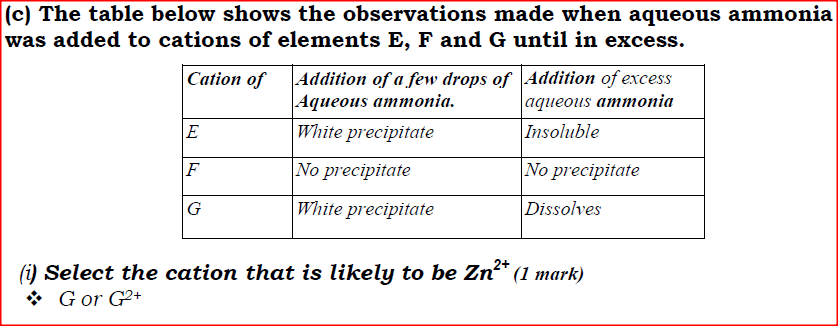
(ii) Using oxidation numbers, show that ammonia is the reducing agent in step (VI) (iii) Write the equation for the reaction that occurs in step (V). (iv) Give one use of ammonium nitrate. (c) The table below shows the observations made when aqueous ammonia was added to cations of elements E, F and G until in excess.
(i) Select the cation that is likely to be Zn2+
(ii) Given that the formula of the cation of element E is E 2+ , write the ionic equation for the reaction between E2+(aq) and aqueous ammonia.
Use the information in the table below to answer the questions that follow. The letters do not represent the actual symbols of the elements.
(a) Give reasons why the melting point of:
(i) S is higher than that of R; (ii) V is lower than that of U. (b) How does the reactivity of W with Chlorine compare with that of R with chlorine? Explain. (c) Write an equation for the reaction between T and excess oxygen. (d) When 1.15g of R were reacted with water, 600cm3 of gas was produced. Determine the relative atomic mass of R. (Molar gas volume = 24 000 cm3). (e) Give one use of element V.
(a) What is meant by standard electrode potential of a an element? (1 mark)
(b) use the standard electrode potentials given below to answer the questions that follow.
(i) State whether acidified MnO4- can oxidise M2+. Give a reason. (2 marks) (ii) Select two half-cells which when combined will give the highest e.m.f. (1 mark) (iii) Write the cell representation for the cell formed in b (ii). (I mark) (iv) Calculate the E0 value for the cell formed in b (iii). (2 marks) (c) A mass of 1.24g of a divalent metal was deposited when a current of 6A was passed through a solution of the metal sulphate for 12 minutes. Determine the relative atomic mass of the metal.
(1 Faraday = 96,500 C mol-1 (3 marks)
(d) State two applications of electrolysis. (I mark)
Hydrogen sulphide is a highly toxic and flammable gas. It is normally prepared in a fume chamber.
(a) Name two reagents that can be used to prepare hydrogen sulphide in the laboratory. (b) One of the uses of hydrogen sulphide is to produce sulphur as shown in the following equation: 2H2S(g) + S02 (g) -> 3S(s) + 2H20(1) Identify the reducing agent in this reaction and give a reason for your answer. (c) Other than production of sulphuric (VI) acid, state one commercial use of sulphur.
ANSWERS
(a)Iron (II) sulphide or conc sulphide / copper sulphide (Accp formula: Fes/ HCl)
Hydrochloric acid or lead (II) sulphide/ HNO3 (b)Hydrogen sulphide The sulphur changes from -2 to zero/ (it reduces SO2 to S) i.e. +4 to 0 / sulphur lost e’s in the H2S to form sulphur (c)Vulcanization of rubber Manufacture of sulphur drugs Manufacture of gun powder/ match sticks / explosives/ fungicides
Draw and name the isomers of pentane.
60 cm3 of oxygen gas diffused through a porous partition in 50 seconds. How long would it take 60cm3 of sulphur (IV) oxide gas to diffuse through the same partition under the same conditions? (S = 32.0, 0 = 16.0)
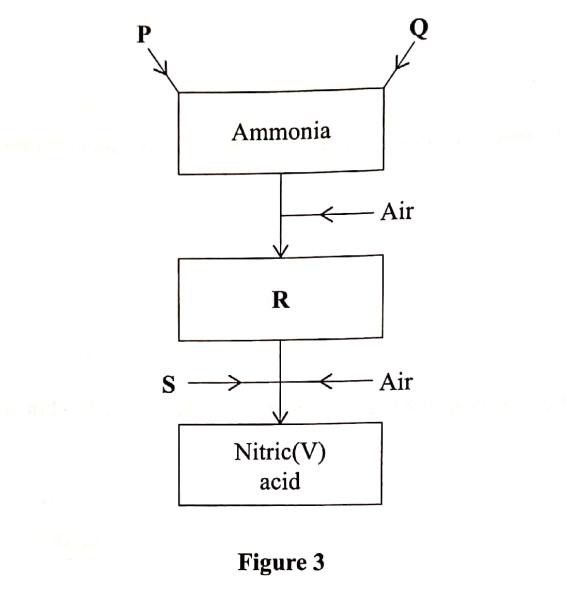
Figure 3 is a flow chart that shows the process that occurs in the manufacture of nitric (V) acid.
(a) Name substance P, Q, R and S.
To obtain substance R, ammonia is heated at 9000C in the presence of air and a catalyst.
The product is then cooled in air.
(e) When ammonia ¡s reacted with nitric(V) acid, it produces a nitrogenous fertiliser.
(a) Explain the following observations:
(b) A sample of water is suspected to contain aluminium ions (AI3+) . Describe a laboratory experiment that can be carried out to show that AI3+ ions are present in the water sample. (3 marks) (c) In an experiment to determine the number of moles of water of crystallisation of a hydrated compound, Na2S04. X H2O, 5g of the compound were heated strongly to a constant mass.
1.
(a) Alkanes are said to be saturated hydrocarbons.
(b) When the alkane, hexane, is heated to high temperature, one of the products is ethene.
Related Chemistry Questions and Answers on Organic Chemistry II Form 3 Level
When fuels burn in the internal combustion engine at high temperature, one of the products formed is nitrogen (II) oxide.
(a) Write the equation for the formation of nitrogen (II) oxide. (b) Give a reason why nitrogen (II) oxide is not formed at room temperature. (c) Describe how formation of nitrogen (II) oxide in the internal combustion engine leads to gaseous pollution.
ANSWERS
(a)N2(g) + 02(g) 2NO(g)
(b)Nitrogen atoms in the molecule are joined by strong triple covalent bond that requires a lot of energy to break than provided at room temperature (c) Nitrogen (II) oxide reacts with oxygen in air to form nitrogen (IV) oxide that dissolves in water vapour causing acid rain.
During an experiment, chlorine gas was bubbled into a solution of potassium iodide.
(a)State the observations made. (b)Using an ionic equation, explain why the reaction is redox
ANSWERS
(a)Solution turned from colourless to dark brown
Greenish yellow / pale green colour of Cl2 disappears Brown solution / black solid is deposited (b) Cl2 (aq) +2I – (aq) → I2 (aq)+2CI- (aq) Explanation; Iodine oxidation state changes from -1 to 0 hence oxidation while Cl2 0.5 changes from 0 to -1 hence reduction / increase is ON and decrease is ON or movement of electrons Cl2 gains e’s where lose
The diagram below represents the set-up that was used to prepare and collect hydrogen chloride gas in the laboratory.
State the purpose of concentrated sulphuric (VI) acid in the wash bottle.
Write an equation for the reaction between dry hydrogen chloride gas and heated iron.
ANSWERS
It is a drying agent.
Fe(s) + 2HCI(g) → FeCI2(s) +H2(g)
When 8.53g of sodium nitrate were heated in an open test-tube, the mass of oxygen gas produced was 0.83 g . Given the equation of the reaction as;
2NaNO 3 (s) -> 2NaNO 2 (s) + O 2 (g) Calculate the percentage of sodium nitrate that was converted to sodium nitrite. (Na = 23.0, N = 14.0, O = 16.0)
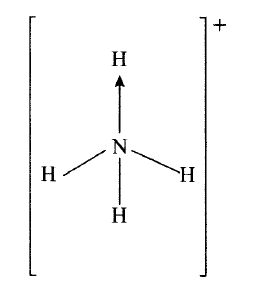
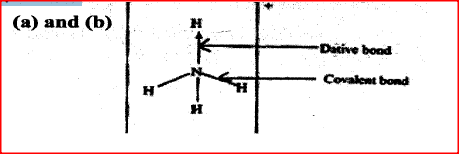
Ammonium ion has the following structure
Label on the structure:
(a) covalent bond; (b) coordinate (dative) bond.
When burning magnesium ribbon is introduced into a gas jar full of nitrogen, it continues to burn producing a greenish yellow powder.
140cm3 of nitrogen gas diffuses through a membrane in 70 seconds. Flow long will it take 200cm3 of carbon(IV) oxide gas to diffuse through the same membrane under the same conditions of temperature and pressure. (3 marks)
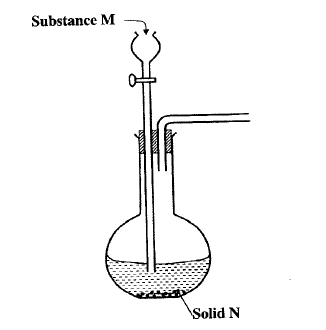
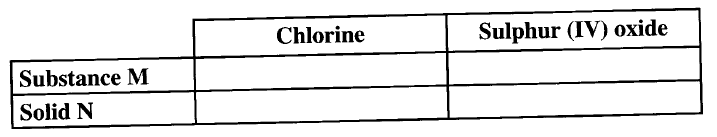
The set up below can be used to generate a gas without heating. This occurs when substance M reacts with solid N.
a i) Complete the table below giving the names of substance M and solid N if the gasses generated are chlorine and sulphur (IV) oxide.
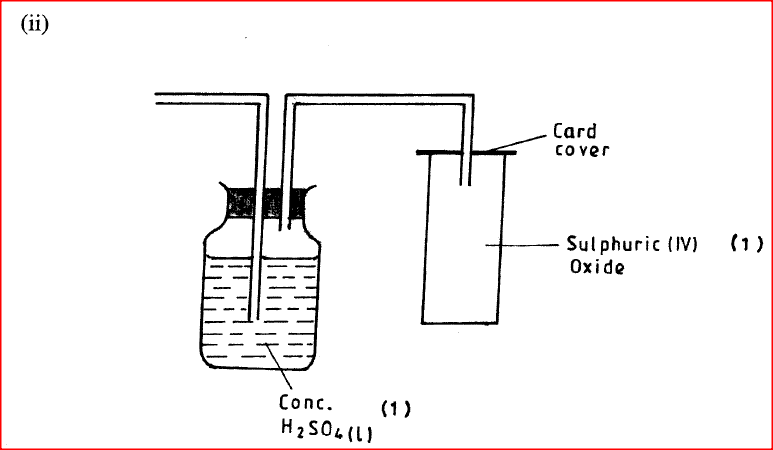
(ii) Complete the diagram above to show how a dry sample of sulphur (IV) oxide can be collected
(b) Describe two chemical methods that can be used to test the presence of sulphur (IV) oxide. (c) Other than the manufacture of sulphuric (VI) acid, state two uses of sulphur (IV) oxide.
ANSWERS
(b) Presence of SO2
- Use of acidified potassium dichromate (VI) which turns from orange to green. - Bubble gas through acidified potassium manganate (VII) which decolourises /changes i.e from purple to colourless. - Iron (III) sulphate solution - yellow/brown changes to green - Bromine water colour changes from yellow/brown / orange to colourless (c) - Fumigation - Bleaching agent - Preservative . - Disinfectant - Antioxidant
(a) State one source of alkanes.
(b) Ethane gas was reacted with 1 mole of bromine gas. State one observation made during this reaction.
ANSWERS
(a) Sources of alkanescrude oil/petroleum natural gas/biogas
(b) The brown/red/orange/yellow colour of bromine is discharged/discoloured
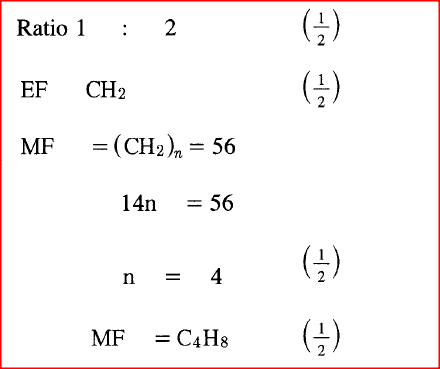
A hydrocarbon contains 14.5% of hydrogen. If the molar mass of the hydrocarbon is 56, determine the molecular formula of the hydrocarbon.
(C = 12.0; H = 1.0) |
Chemistry Topics
All
Archives
December 2024
|
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |








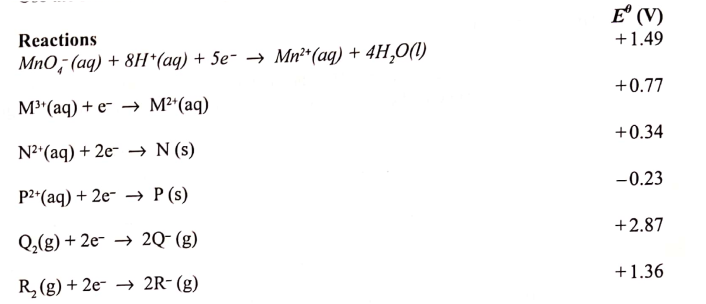

















 RSS Feed
RSS Feed

