|
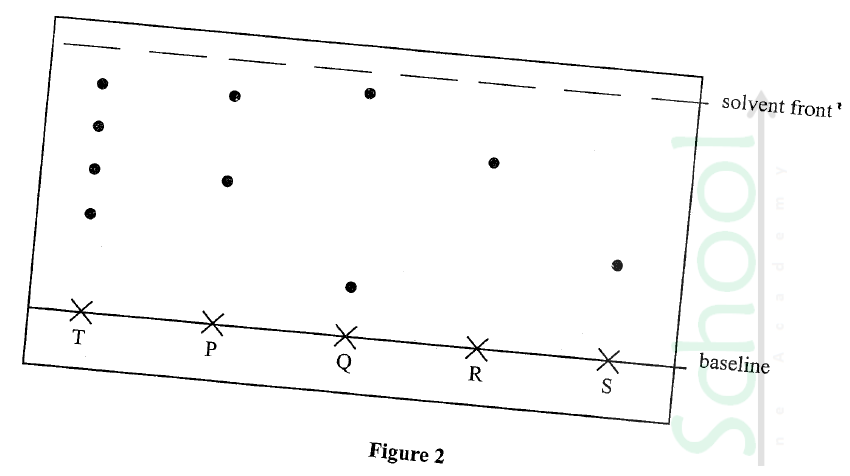
An experiment was Carried out to determine the presence of Substances P, Q, R and S in mixture T. The results obtained are shown in Figure 2.
(a) Ne the method of separation illustrated in Figure 2.
(b) Select: (i) one Substance Which Contains a Component not present in T. (ii) a substance which is least Soluble in the solvent used.
ANSWERS
(a)Chromatography/paper chromatography
(b)(i)Q (ii)S
0 Comments
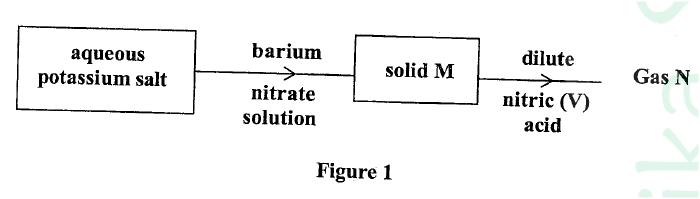
Study the flow chart in Figure 1 and answer the questions that follow.
Gas N forms a while suspension with aqueous calcium hydroxide.
(a) Name the anion present in the potassium salt. (b) Write an ionic equation for the formation of solid M. (c) Give one use of gas N.
30.0 cm3 of aqueous sodium hydroxide containing 8.0 g per litre of sodium hydroxide were completely neutralized by 0.294 g of a dibasic acid. Determine the relative formula mass of the dibasic acid. (Na = 23.0 ; 0 = 16.0; H 1.0)
(a) State Graham’s law of diffusion
(b) Explain why a balloon filled with helium gas deflates faster than a balloon of the same size filled with argon gas.
ANSWERS
(a) Graham’s Law of diffusion
The rate of diffusion of a gas is inversely proportional to the square root of its density at constant temperature and pressure. (b)Helium is less dense than argon hence it diffuses out of the balloon faster than argon.
Describe an experiment to show that group one elements react with cold water to form alkaline Solutions
ANSWERS
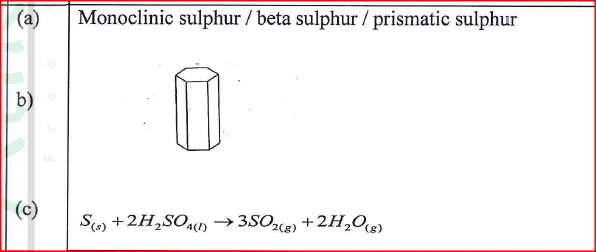
One of the allotrope s of sulphur is rhombic sulphur.
(a) Name the other allotrope of sulphur. (b) Draw a diagram to show the shape of the allotrope named in (a) above. (c) Write an equation for the reaction between concentrated sulphur(VI) acid and sulphur
The following are formulae of organic compounds. Use the formulae to answer the questions that follow:
(a) Select:
(i) two compounds which when reacted together produce a sweet smelling compound. (ii) an unsaturated hydrocarbon. (b) Name the compound selected in (a) (ii).
(a) Explain why it is not advisable to prepare a sample of carbon(IV) oxide using barium carbonate and dilute sulphuric(VI) acid.
(b) State a method that can be used to collect dry carbon(IV) oxide gas. Give a reason.
ANSWERS
(a)The reaction starts but soon stops.
This is because the insoluble barium sulphate produced forms a coating on the surface of the barium carbonate preventing further reaction and evolution of carbon(IV) oxide gas. (b) Downward delivery. Carbon(TV) oxide is denser than air.
(a) Define a soluble base.
(b) Aqueous solutions of 2M ethanoic acid and 2M nitric(V) acid were tested for electrical conductivity. Which solution is a better conductor of electricity? Explain.
ANSWERS
(a)A soluble base is a substance that dissociates in water to produce hydroxide ions as the only negative ions.
(b)Nitric(V) acid. This is because nitric(V) acid is a strong acid and dissociates completely in solution producing many H+ ions. |
Chemistry Topics
All
Archives
December 2024
|
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |


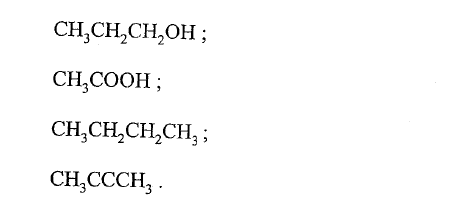


 RSS Feed
RSS Feed

