|
Ethene is prepared in the laboratory by dehydration of ethanol.
0 Comments
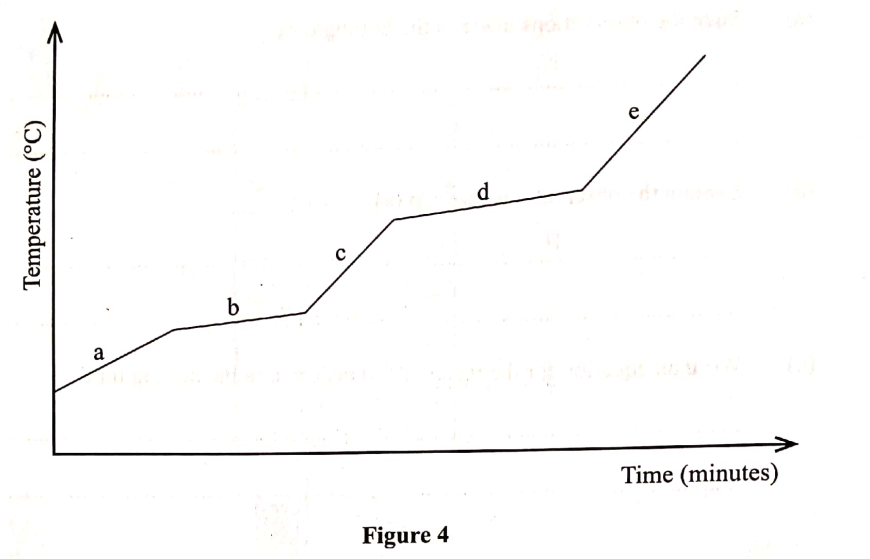
The graph in Figure 4 was obtained when a certain substance was heated and its temperature recorded at regular intervals.
(a) State the purity of the substance. (I mark)
(b) Explain the answer in (a). (2 marks)
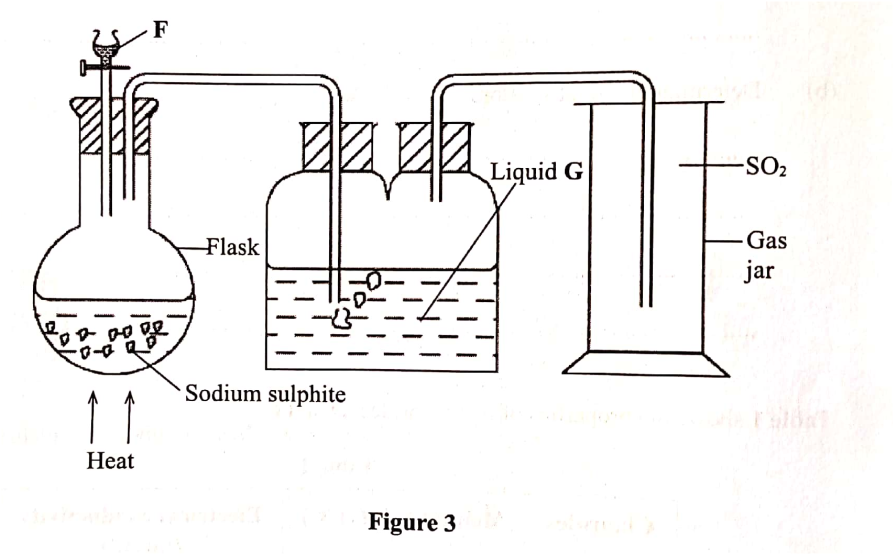
Sulphur(IV) oxide is prepared in the laboratory using the set-up in Figure 3. Study it and answer the questions that follow.
(a) Identify substance F. (1 mark)
(b) Write an equation for the reaction that takes place in the flask. (1 mark) (c) State the purpose of liquid G. (I mark)
Table 1 shows the properties of two chlorides, D and E.
(a) State the type of bond present in:
A solution contains 40.3 g of substance XOH per litre. 25M cm3 of this solution required 30.0cm3 of 0.3 M sulphuric(Vl) acid for complete neutralisation. (a) Calculate the number of moles of XOH that reacted. (½ mark) (b) Determine the relative atomic mass of X. (1½ marks)
A farmer intended to plant cabbages in his farm. He first tested the pH of the soil and found it to be 3.0. If cabbages do well in alkaline soils, explain the advice that would be given to the farmer in order to realise a high yield. (2 marks)
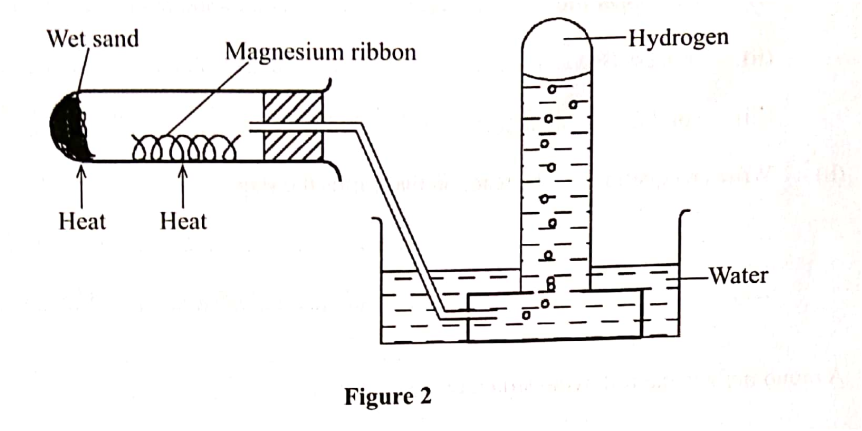
Hydrogen gas can be prepared by passing steam over heated magnesium ribbon as shown in Figure 2.
(a) Write an equation for the reaction that produces hydrogen gas. (1 mark)
(b) Explain why the delivery tube must be removed from beneath the water before heating is stopped. (1 mark) (c) Explain why sodium metal is not suitable for this experiment. (1 mark)
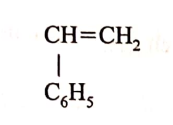
A monomer has the following structure.
(a) Draw the structure of its polymer that contains three monomers. (1 mark)
(b) A sample of the polymer formed from the monomer has a molecular mass of 4992. Determine the number of monomers that formed the polymer (C= 12; H= 1.0). (2 marks)
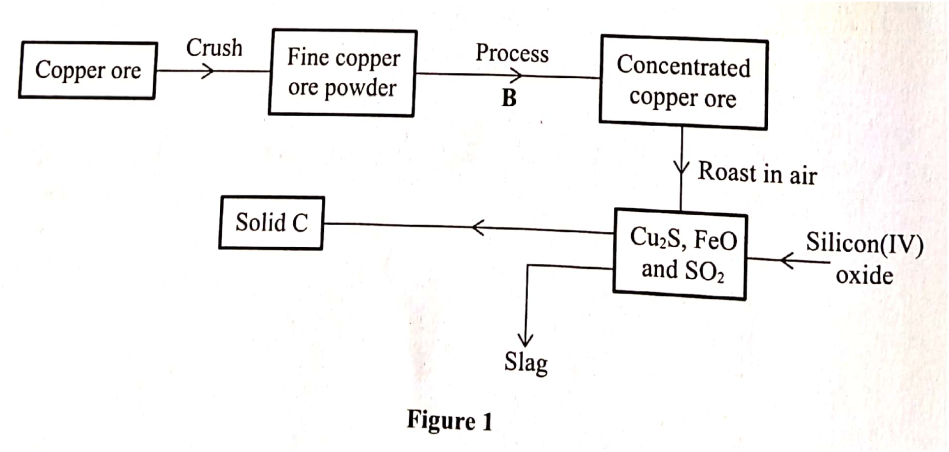
The flow chart in Figure 1 represents some stages in the extraction of copper metal. Study it and answer the questions that follow.
Identify:
Describe how an increase in concentration increases the rate of a reaction. (2 marks)
Expected Response
As the concentration increases the number of reactivity of particles per unit volume increases thus affecting collisions. This leads to increase in the rate of reaction
An atom of element A has mass number 39 and 19 protons.
|
Chemistry Topics
All
Archives
December 2024
|
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |















 RSS Feed
RSS Feed

