|
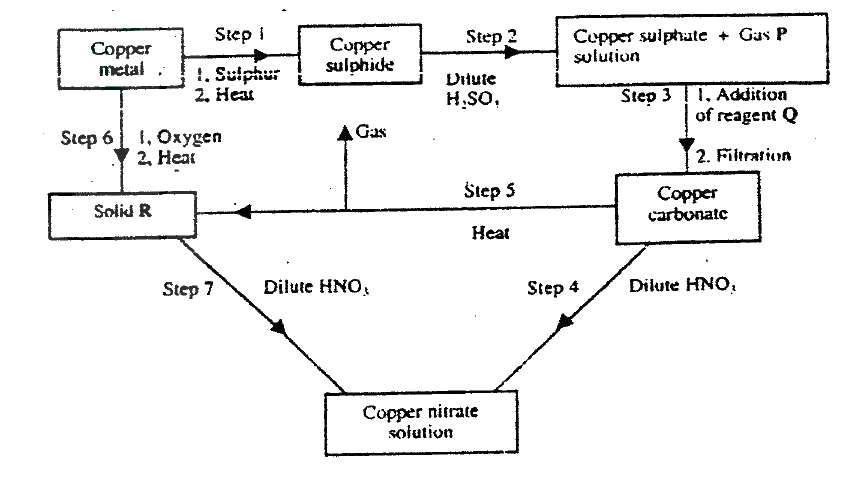
The flow chart below shows a sequence of chemical reactions starting with copper study it and answer the questions that follow.
(a) In step 1, excess 3M nitric acid was added to 0.5g of copper powder
(i) State two observations which were made when the reactions was in progress (ii) Explain why dilute hydrochloric acid cannot be used in step 1 (iii) I Write the equation for the reaction that took place in step 1 II Calculate the volume of 3M nitric that was needed to react completely with 0.5g of copper powder. (Cu = 63.5) (b) Give the names of the types of reactions that took place in steps 4 and 5 Step 4 Step 5 (c) Apart from the good conductivity of electricity, state two other properties that make it possible for copper to be extensively used in the electrical industry.
0 Comments
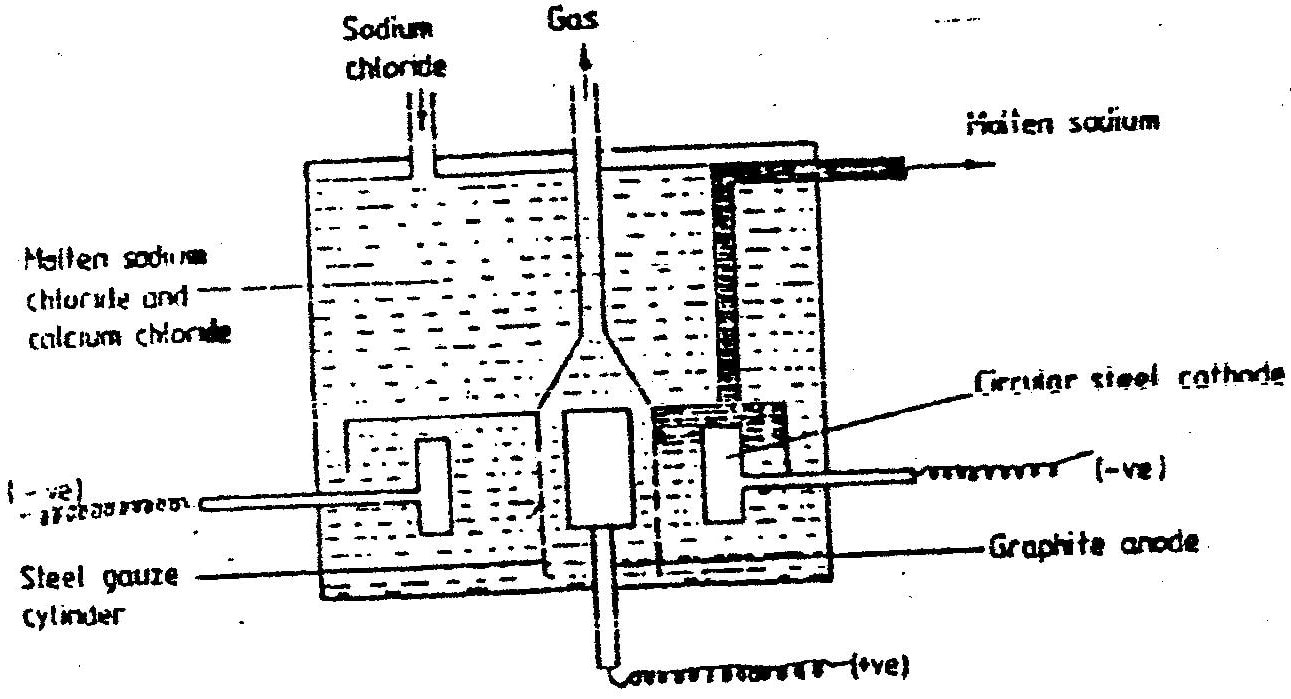
(a) The diagram below shows the extraction of sodium metal using the downs cell. Study it answer the questions that follow
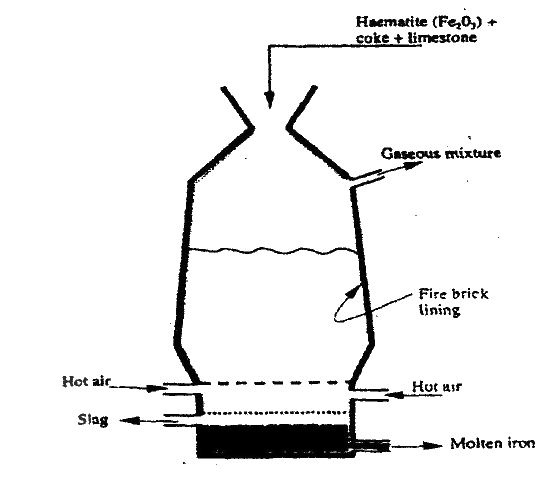
The extraction of iron from its ores takes place in the blast furnace. Study it and answer the questions that follow.
a) Name
(i) One of the substances in the slag (ii) Another iron ore material used in the blast furnance. (iii) One gas which is recycled. b) Describe the process which leacd to the formation of iron in the blast furnace c) State the purpose of limestone in the blast furnace. d) Give a reason why the melting point of the iron obtained from the blast furnace is 12000 C while tat of pure iron is 15350C (e) State two uses of steel
(a) Explain why the metals magnesium and aluminium are good conductors of electricity.
b) Other than cost, give two reasons why aluminium is used for making electric cables while magnesium is not
ANSWERS
a)They have delocalized valency electrons
b)Aluminium has three delocalized electrons. It is resistant to corrosion
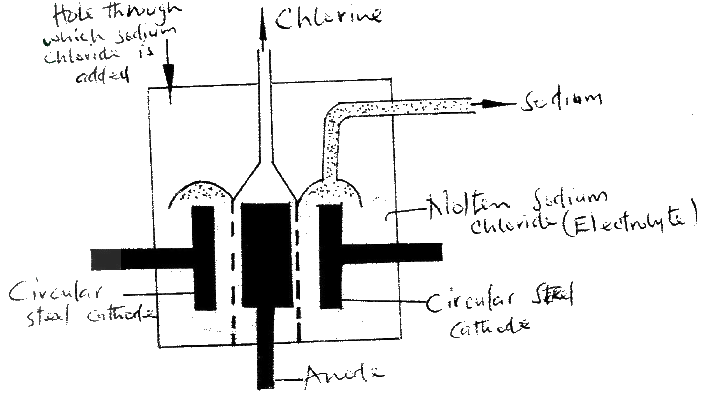
(a) Below is a simplified diagram of the Downs Cell used for the manufacture of sodium.
Study it and answer the questions that follow
(i) What material is the anode made of? Give a reason
(ii) What precaution is taken to prevent chlorine and sodium from re- combination? (iii) Write an ionic equation for the reaction in which chlorine gas is formed (b) In the Downs process, (used for manufacture of sodium), a certain salt is added to lower the melting point of sodium chloride from about 800°C to about 600°C. (i) Name the salt that is added (ii) State why it is necessary to lower the temperature (c) Explain why aqueous sodium chloride is not suitable as an electrolyte for the manufacture of sodium in the Downs process (d) Sodium metal reacts with air to form two oxide. Give the formulae of two oxides
In the industrial extraction of lead, the ore is first roasted in a furnace. The solid mixture obtained is then fed into another furnace together with coke, limestone and scarp iron. State the function of each of the following in this process:
(a) Coke (b) Limestone (c) Scrap iron
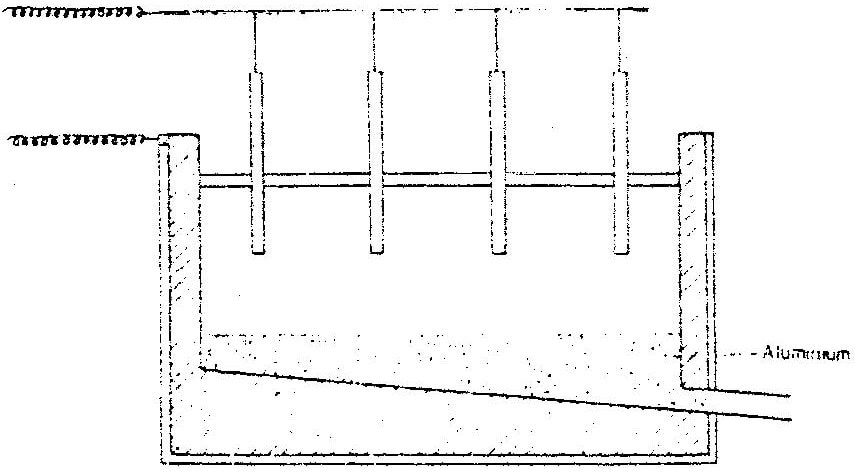
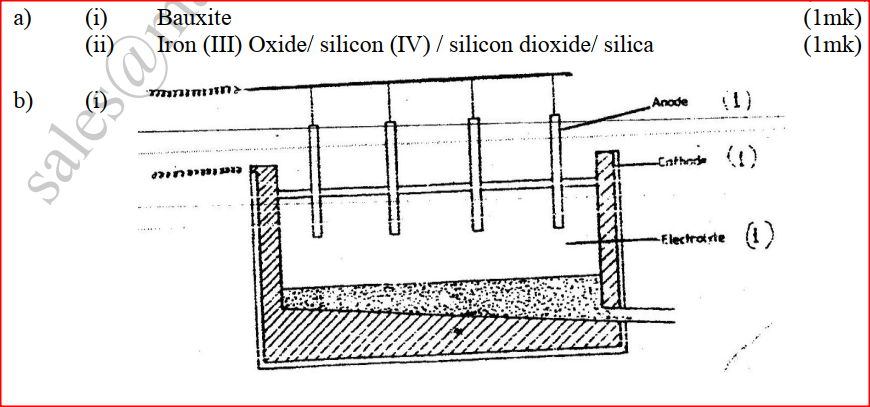
The extraction of aluminium from it s ore takes place in two stages, purification stage and electrolysis stage. The diagram below shows the set – up for the electrolysis stage
a)
i) Name the ore from which aluminium extracted. (1mk) ii) Name one impurity, which is removed at the purification stage.(1mk) b) i) Label on the diagram each of the following i) Anode ii) Cathode iii) Region containing the electrolyte. ii) The melting point aluminium oxide is 20540C, but electrolysis is carried out between 800 – 9000C. i) Why is the electrolysis not carried out at 20540C (1mk) ii) What is done lower the temperatures? (1mk) iii) The aluminium which is produced is tapped off as aliquid. What does this suggest about it smelting point? (1mk) c) A typical electrolysis cell uses current of 40,000 amperes.Calulate the mass (in kilograms) of aluminium produced in one hour) (3mks)
answers
The basic raw material for extraction of aluminium is bauxite
a) Name the method that is used to extract aluminium from bauxite b) Write the chemical formula of the major component of bauxite c) i) Name two major impurities in bauxite ii) Explain how the impurities in bauxite are removed d) Cryolite is used in the extraction of aluminium from bauxite. State its function f) Aluminium is a reactive metal yet utensils made of aluminium do not corrode easily. Explain this observation
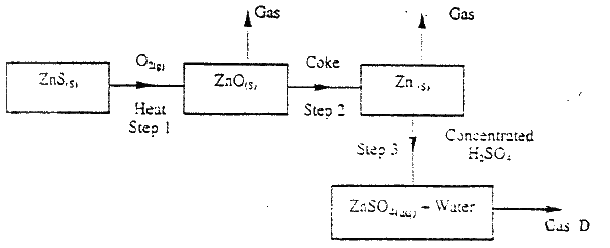
Brass is an alloy of zinc and copper. Give one use of brass
answers
(a) Name one ore from which copper metal is extracted
(b) The chart below shows a sequence of reactions starting with copper. Study it and answer the questions that follow
(ii) Write an equation for the reaction that takes place in step 5
(iii) State the observations made in steps 4 and 7 Step 4 Step 7 (c) Bronze is an alloy of copper and another metal (i) Name the other metal (ii) Give one use of Bronze
Iron is extracted from its ore by the blast furnace process
(a) Name one ore from which iron is extracted (b) One of the impurities in iron is removed in the form of calcium silicate. Write an equation for the reaction in which calcium silicate is produced |
Chemistry Topics
All
Archives
December 2024
|
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |
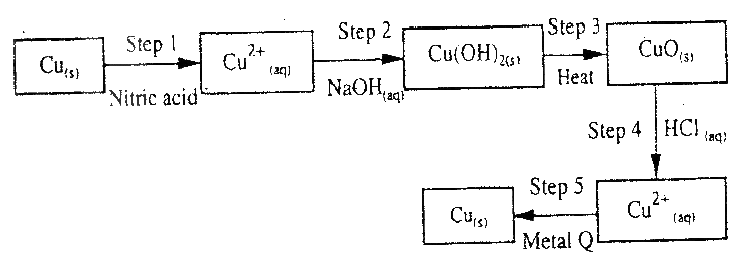










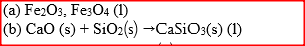

 RSS Feed
RSS Feed

