|
Describe two chemical tests that can be used to distinguish ethanol from ethanoic acid.
0 Comments
Give two uses of the polymer polystyrene.
ANSWERS
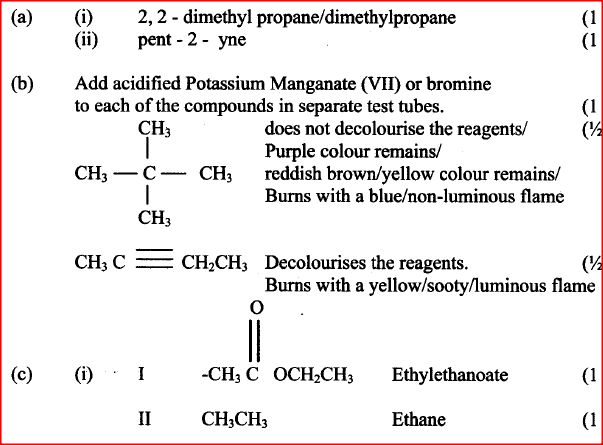
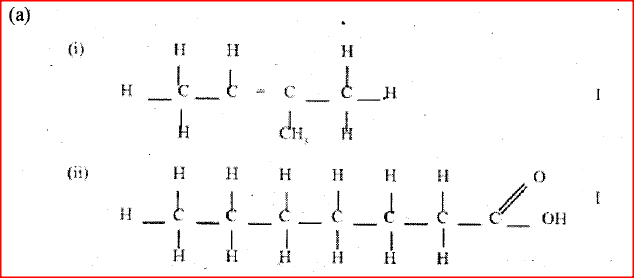
(a) Give the names of the following compounds
Name the type of bromination reaction that takes place in: (i) and (ii)
(d) The polymerization of tetra flouroathene (C2H4) is similar to that of ethane ( C2H4)
(e) State any two advantages that synthetic polymers have over natural polymers
Pentane and ethanol are miscible. Describe how water could be used to separate a mixture of pentane and ethanol
Expected Response
20. Add water to the mixture in a separating funnel. Ethanol dissolves while pentane does not. Allow the mixture to separate in two layers. Open the tap to drain the lower aqueous layer. Distil the water ethanol mixture to get ethanol.
a) When an organic compound Y is reacted with aqueous sodium carbonate, it produces carbon dioxide reacts with propanol to form a sweet smelling compound Z whose formula is.
c) The scheme below was used to prepare a cleaning agent. Study and answer the questions that follow.
i) What name is given to the type of cleaning agent prepared by the method shown in the scheme?
ii) Name one chemical substance added in step II iii) What is the purpose of adding the elements substance names in C(ii) above. iv) Explain how an aqueous solution of the cleansing removes oil from utensils during washing.
Bromine reacts with ethane as shown below
(a) What condition is necessary for this reaction to occur?
(b) Identify the bonds which are broken and those that are formed
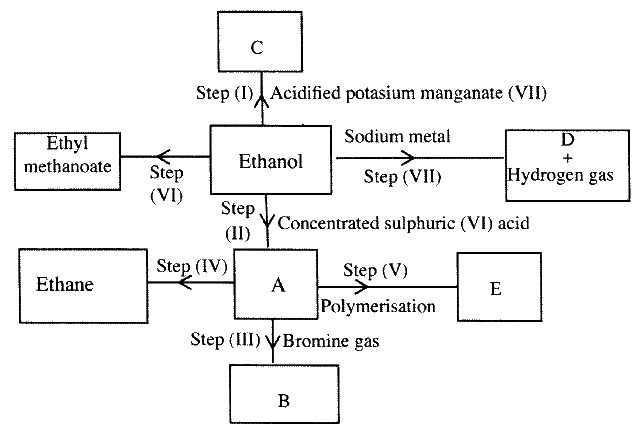
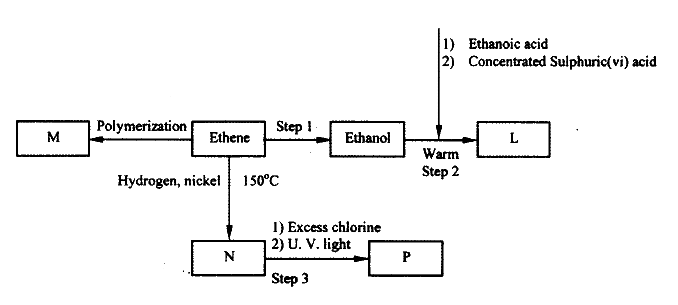
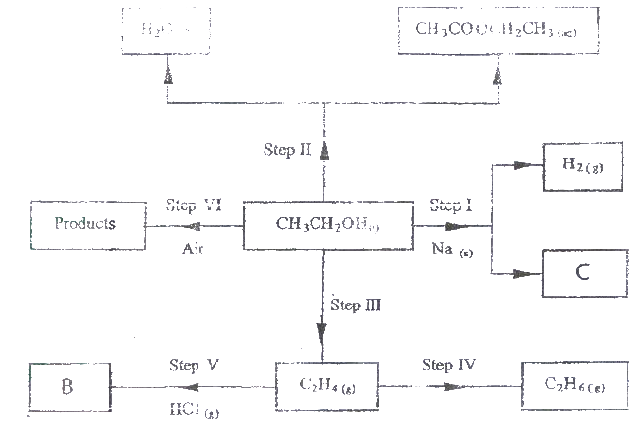
(a) Study the flowchart below and answer the questions that follows;
(i) I What observation will be made in Step I
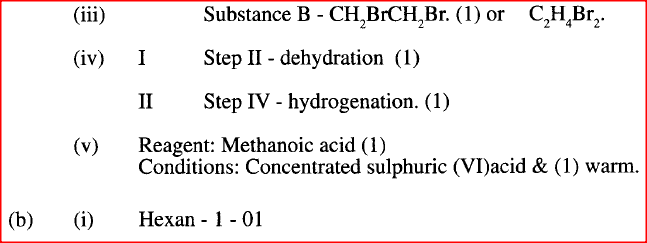
II Describe a chemical test that can be carried out to show the identity of compound C (ii) Give the names of the following I E II substance D (iii)Give the formula of substance B . (iv) Name the type of reaction that occurs in: I Step (II) II Step (IV) (v) Give the reagent and conditions necessary for Step (VI). Reagent: Conditions (b) (i) Name the following structure.
(ii) Draw the structure of an isomer of pentene.
ANSWERS
(a) (i) I The potassium permanganate is decolorized or changes from purple to colourless.
II C is a ethanoic acid (carboxylic acid) Add sodium carbonate, you will see effervescence, test gas evolved with lime water, it will form a white precipitate. (ii) I Polyethene II Substance D - sodium ethoxide
One of the fuels associated with crude oil is natural gas. Name the main constituent of natural gas and write an equation for its complete combustion
Name: Equation:
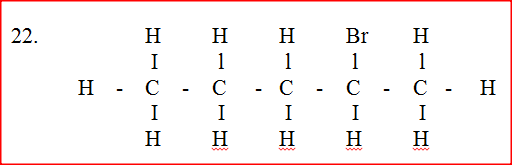
Compound L reacts with hydrogen bromide gas to give another compound whose structure is
(a) Give the structural formula and name of compound L
(b) Write an equation for the reaction, which takes place between ethane and excess chlorine gas
Two organic compounds P and Q decolourise acidified potassium manganate (VII) solution but only P reacts with sodium metal to give a colourless gas. Which homologous series does compound P belong? Give a reason.
ANSWERS
P is in alkanol R - OH. The alkanol reacts with sodium metal to produce the colourless gas.
(a) What name is given to the process by which alcohol is formed from a carbohydrate?
(b) Explain why the solubility of ethane in water is lower than that of ethanol.
ANSWERS
(a) Fermentation.
(b) Ethane remains in molecular form while ethanol forms hydrogen bonds with water.
(a).i) Give the name of the following compounds:
b). Describe a chemical test that can be carried out in order to distinguish between
c). Study the flow chart below and answer the questions that follows
i) Name the compounds:
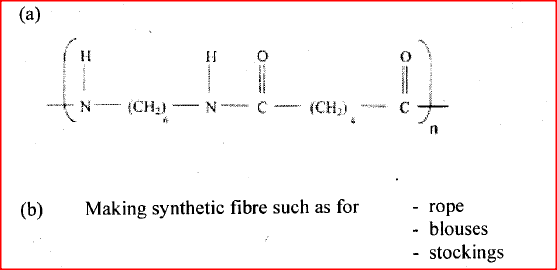
(I). L (II). N ii) Draw the structural formula of compound M showing two repeat units iii) Give the reagent and the conditions used in step I iv) State the type of reaction that take place in: (I). Step 2 (II). Step 3
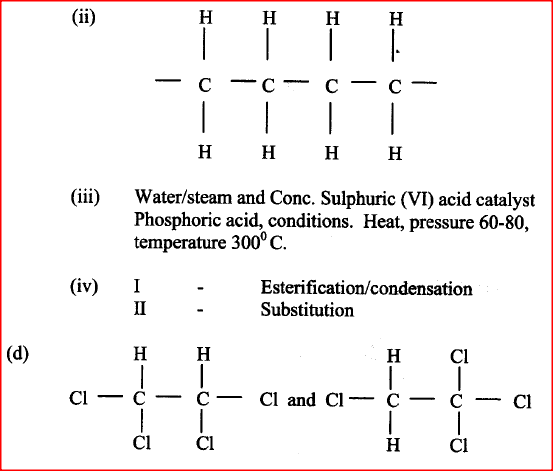
d). The molecular formula of compound P is C2H2Cl4. Draw the two structural formulae of compound P
The use of CFCs has been linked to depletion of the ozone layer.
a) What does CFC stand for? b) Explain the problem associated with the depletion of the ozone layer c) State another environment problem caused by CFCs
ANSWERS
(a)Chlorofluorocarbon
(b)When ozone is depleted, high energy UV radiations reach the earth, which may cause skin cancer to human beings. (c)Global warmings, /green house effect.
Some animal and vegetable oils are used to make margarine and soap. Give the reagents and conditions necessary for converting the oils into:
a) Margarine b) Soap
State the observation that would be made when a piece of sodium metal is placed in samples of:
Pentane: __________ Pentanol: __________
a) Draw the structures of the following compounds:
i) 2- methylbut -2 ene; ii) heptanoic acid b) Describe a physical test that can be used to distinguish between methanol and hexanol. c) Use the flow chart below to answer the questions that follow.
i) Name: (I) the type of reaction that occurs in step II; (II) Substance B. ii) Give the formula of substance C. iii) Give the reagent and the conditions necessary for the reaction in step (IV)
ANSWERS
(b)
(a) The following equations represents two different types of reactions
(i) nC4H8(g) → (C4H8)n(s) (ii) C2H6(g) + CI2(g) → C2H5CI(I) + HCI(g) State the type of reaction represented by: (i) (ii) (b) The fermentation of glucose produces ethanol as shown in the equation below C2H12O6(aq) → 2CH3CH2OH(aq) + 2CO2(g)
(c) The molecular formula of a hydrocarbon is C6H14. The hydrocarbon can be converted into two other hydrocarbons as shown by the equation below (i) Name and draw the possible structural formula of X Name Structural formula (ii) State and explain the observation that would be made if a few drops of bromide water were added to a sample of X. (iii) Write an equation for the complete combustion of C3H8
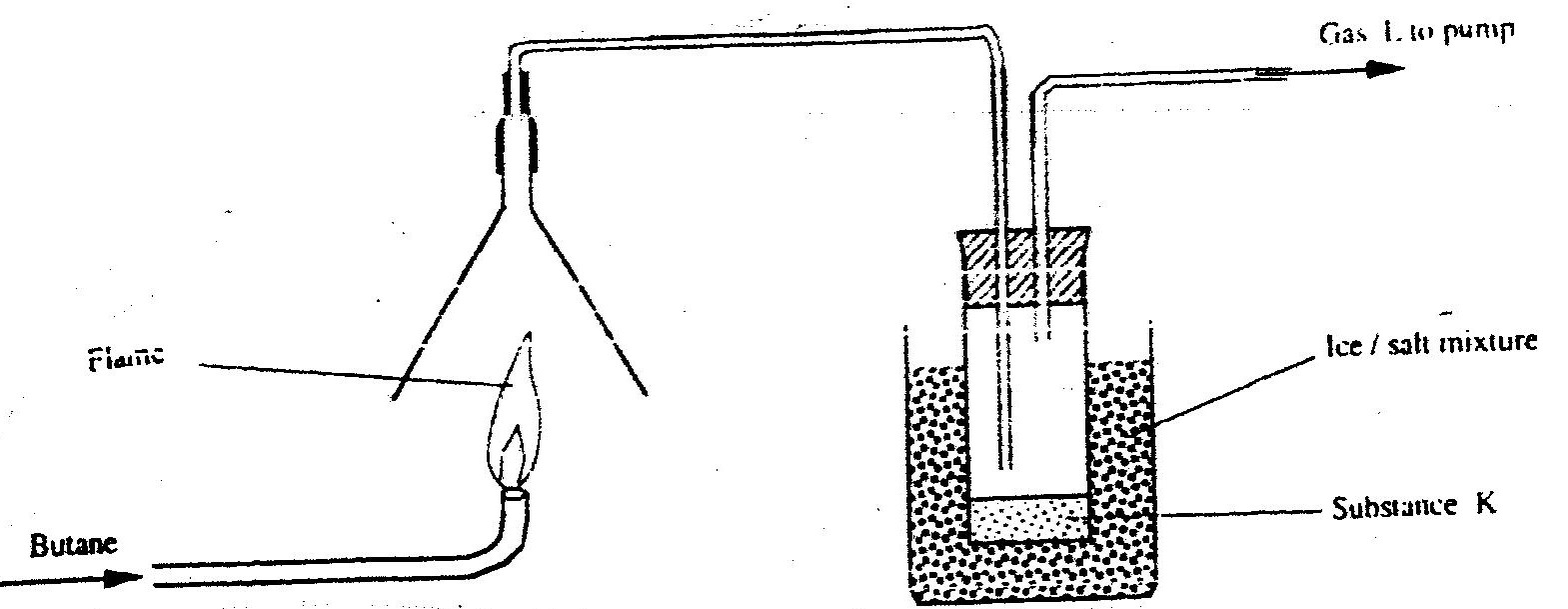
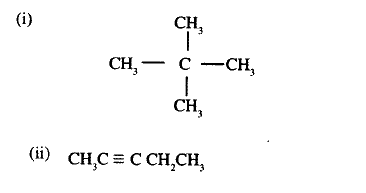
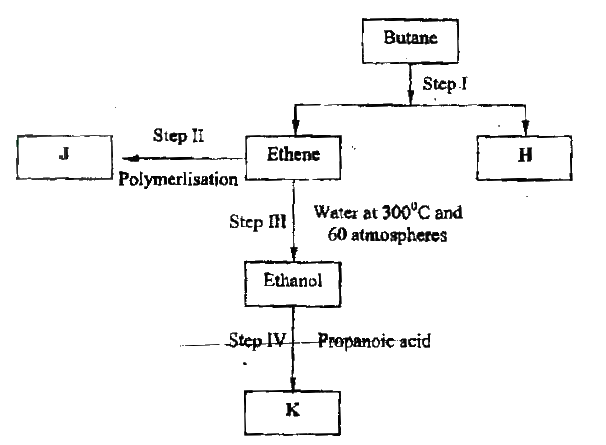
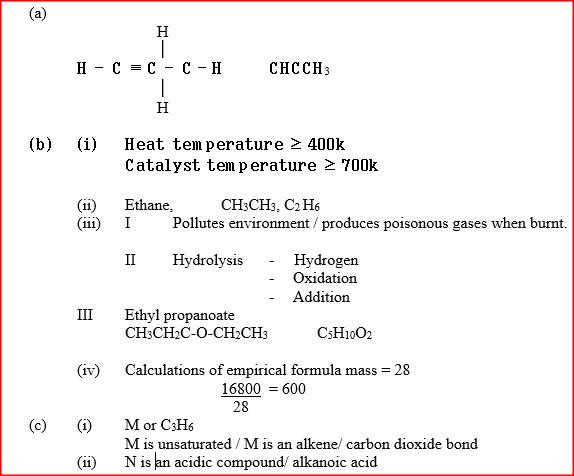
(a) Alkanes, alkenes and alkynes can be obtained from crude oil. Draw the structure of the second member of the alkyne homologous series.
(b) Study the flow chart below and answer the questions that follow
(i) State the conditions for the reaction in step 1 to occur
(ii) Identify substance II (iii) Give: I. One advantage of the continued use of substance such as J II The name of the process that takes place in step III III The name and the formula of substance K Name: Formula: (iv) The relative molecular mass of J is 16,800. Calculate the number of monomers that make up J. (c) The table below give the formula of four compounds L,M,N and P
Giving a reason in each case, select the letter which represents a compound that:
(i) Decolorizes bromine in the absence of UV light (ii) Gives effervescence when reacted with aqueous sodium carbonate
Draw the structural formula of:
Expected Response
a) Biogas is a mixture of mainly carbon (IV) oxide and methane.
(i) Give a reason why biogas can be used as a fuel. (ii) Other than fractional distillation, describe a method that can be used to determine the percentage of methane in biogas. b) A sample of biogas contains 35.2% by mass of methane. A biogas cylinder contains 5.0 kg of the gas. Calculate the; (i) Number of moles of methane in the cylinder. (Molar mass of methane=16) (ii) Total volume of carbon (IV) oxide produced by the combustion of methane in the cylinder (Molar gas Volume=24.0 dm-3+ at room temperature and pressure). c) Carbon (Iv) oxide, methane, nitrogen (I) oxide and trichlorofluoromethane are green-house gases. (i) State one effect of an increased level of these gases to the environment. (ii) Give one source from which each of the following gases is released to the environment; I Nitrogen (i) oxide II Trichlorofluoromethane.
ANSWERS
The structure of a detergent is
a) Write the molecular formula of the detergent.
b) What type of detergent is represented by the formula? c) When this type of detergent is used to wash linen in hard water, spots (marks) are left on the linen. Write the formula of the substance responsible for the spots
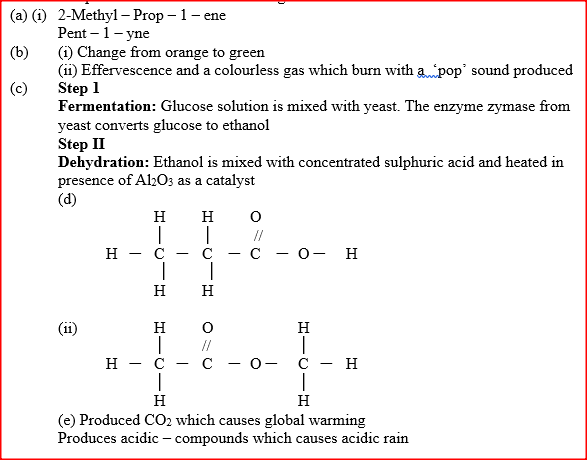
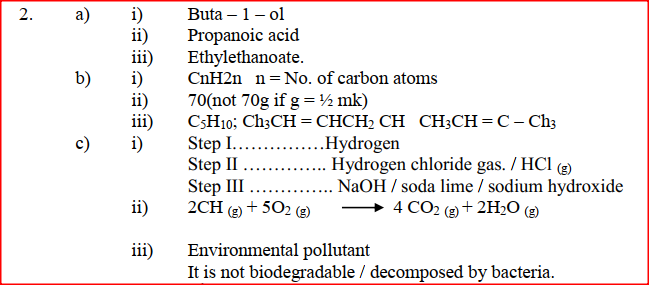
(a) Give the systematic names of the following compounds
(b) State the observations made when Propan – 1- ol reacts with:
(i) Acidified potassium dichromate (VI) Solution (ii) Sodium metal (c) Ethanol obtained from glucose can be converted to ethane as shown below
Name and describe the process that take place in steps I and IIStep I Step II
(d) Compounds A and B have the same molecular formula C3H6O2. Compound A liberates carbon (IV) oxide on addition of aqueous sodium carbonate while compound B does not. Compound B has a sweet smell. Draw the possible structures of: (i) Compound A (ii) Compound B (e) Give two reasons why the disposal of polymers such as polychloroethane by burning pollutes the environment.
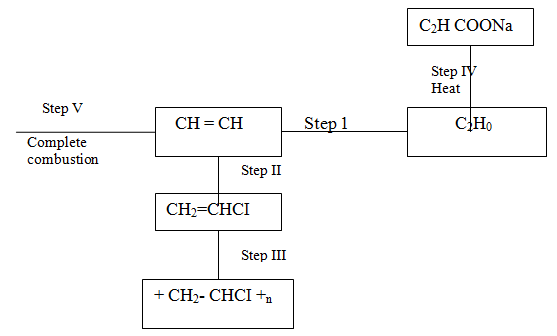
(a) Give the names of the following compounds
(b) Study the information in the table below and answer the questions that follow
(i) Name the reagents used in:
Step I ……………………. Step II …………………….. Step III …………………….. (ii) Write an equation for the complete combustion of CH = CH (iii) Explain one disadvantage of the continued use of items made from the compound formed in step III |
Chemistry Topics
All
Archives
December 2024
|
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |


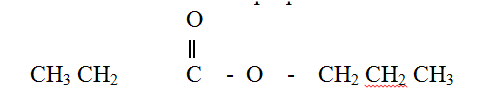


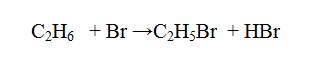

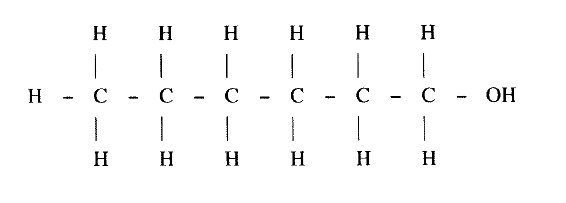
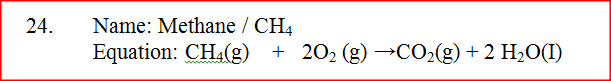
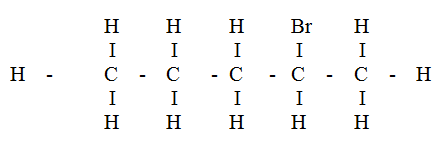
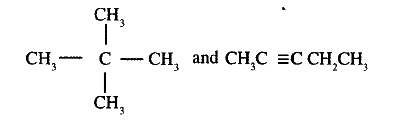



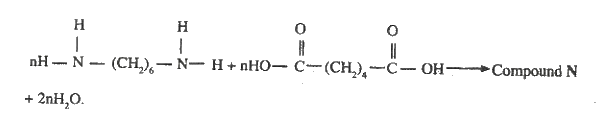

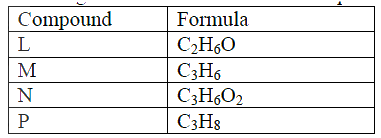




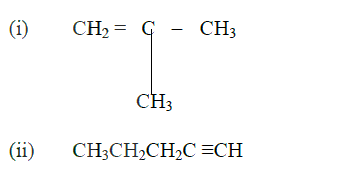
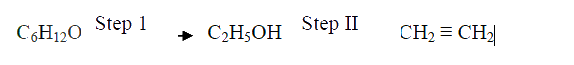
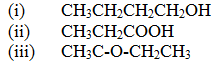


 RSS Feed
RSS Feed

