|
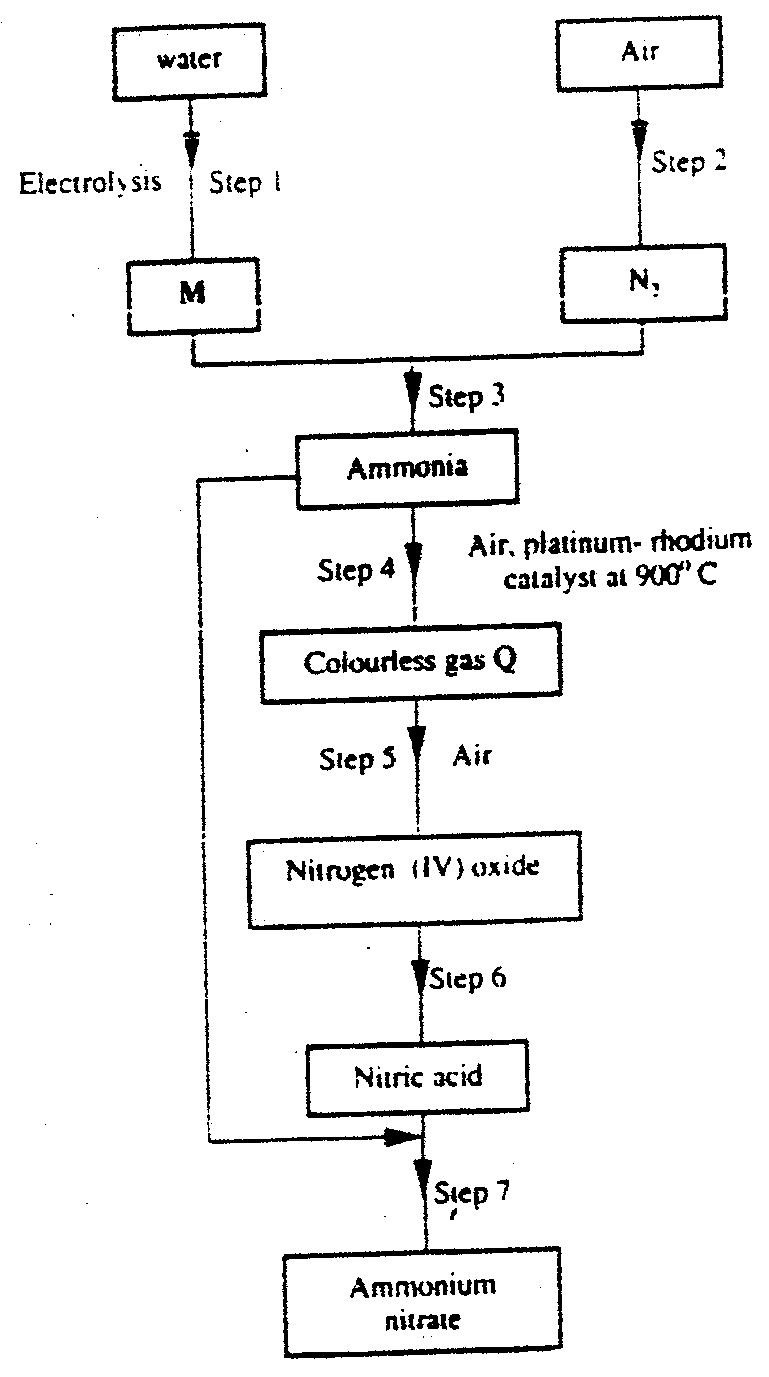
(a) Describe the process by which Nitrogen is obtained from air on a large scale.
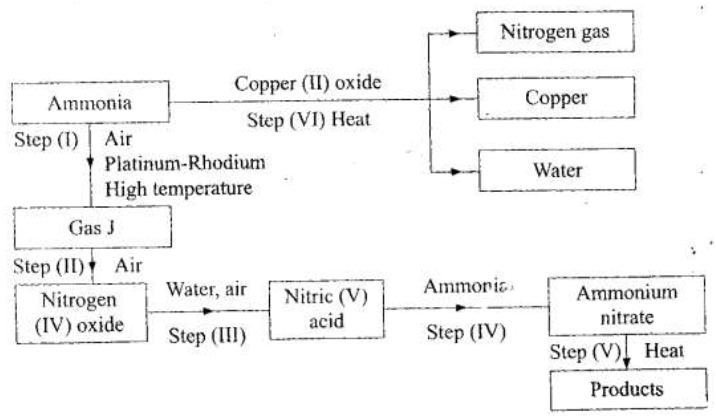
(b) Study the flow chart below and answer the questions that follow.
(i) Identify gas J.
(ii) Using oxidation numbers, show that ammonia is the reducing agent in step (VI) (iii) Write the equation for the reaction that occurs in step (V). (iv) Give one use of ammonium nitrate. (c) The table below shows the observations made when aqueous ammonia was added to cations of elements E, F and G until in excess.
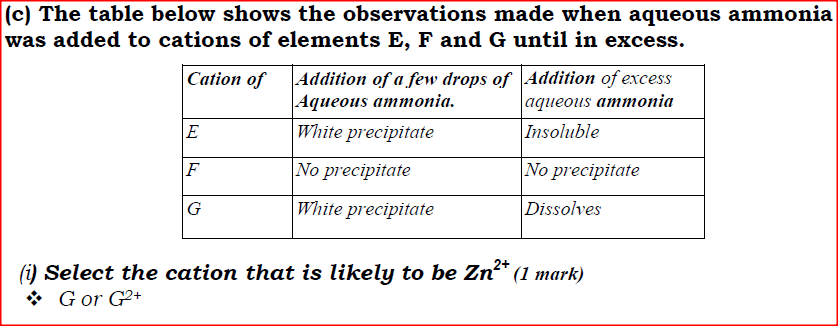
(i) Select the cation that is likely to be Zn2+
(ii) Given that the formula of the cation of element E is E 2+ , write the ionic equation for the reaction between E2+(aq) and aqueous ammonia.
0 Comments
Use the information in the table below to answer the questions that follow. The letters do not represent the actual symbols of the elements.
(a) Give reasons why the melting point of:
(i) S is higher than that of R; (ii) V is lower than that of U. (b) How does the reactivity of W with Chlorine compare with that of R with chlorine? Explain. (c) Write an equation for the reaction between T and excess oxygen. (d) When 1.15g of R were reacted with water, 600cm3 of gas was produced. Determine the relative atomic mass of R. (Molar gas volume = 24 000 cm3). (e) Give one use of element V.
(a) What is meant by rate of reaction. (1 mark)
(b) In the space provided, sketch the diagram of a set-up that can be used to determine the rate of reaction between manganese(IV) oxide and hydrogen peroxide. (3 marks)
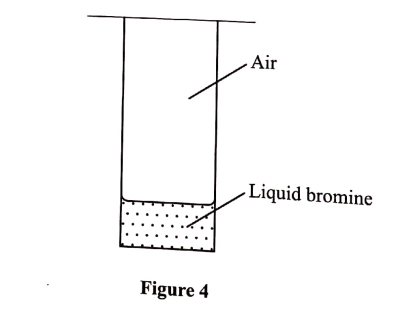
(e) A student placed a small amount of liquid bromine at the bottom of a sealed gas jar of air as shown in Figure 4.
(i) Describe what will be observed: (1 mark)
I. after two minutes . II. after 30 minutes
(ii) Use the Kinetic theory to explain the observations: (2 marks)
I. after 2 minutes . II. after 30 minutes
(d) Some plants have seeds that contain vegetable oil.
(a) What is meant by standard electrode potential of a an element? (1 mark)
(b) use the standard electrode potentials given below to answer the questions that follow.
(i) State whether acidified MnO4- can oxidise M2+. Give a reason. (2 marks) (ii) Select two half-cells which when combined will give the highest e.m.f. (1 mark) (iii) Write the cell representation for the cell formed in b (ii). (I mark) (iv) Calculate the E0 value for the cell formed in b (iii). (2 marks) (c) A mass of 1.24g of a divalent metal was deposited when a current of 6A was passed through a solution of the metal sulphate for 12 minutes. Determine the relative atomic mass of the metal.
(1 Faraday = 96,500 C mol-1 (3 marks)
(d) State two applications of electrolysis. (I mark)
(a) What ¡s meant by molar heat of neutralisation? (1 mark)
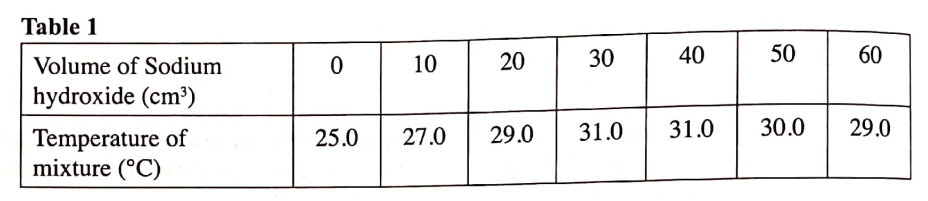
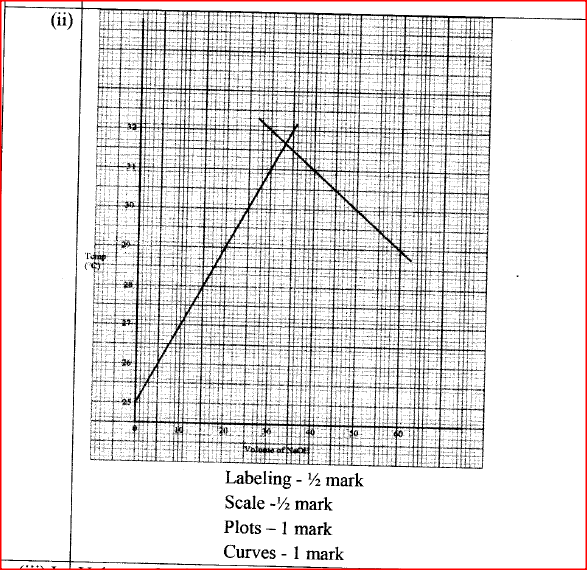
(b) In an experiment to determine the molar heat of neutralisation, 50 cm3 of 1M hydrochloric acid was neutralised by adding 10 cm3 portions of dilute sodium hydroxide. During the experiment, the data in Table 1 was obtained.
(iii) Determine from the graph the: I. volume of sodium hydroxide which completely neutralises 50 cm3 of 1M hydrochloric acid. (1 mark) II. change in temperature,△T, when complete neutralisation occurred. (1 mark) (iv) Calculate: I. the heat change, △H when complete neutralisation occurred. (Specific heat capacity = 4.2 Jg-1K-1, density of solution 1.0 gcm-3) (2 marks) II. molar heat of neutralisation of hydrochloric acid with sodium hydroxide. (1 mark) (v) How would the value of molar heat differ if 50 cm3 of 1M ethanoic acid was used instead of 1M hydrochloric acid? Give a reason. (2 marks)
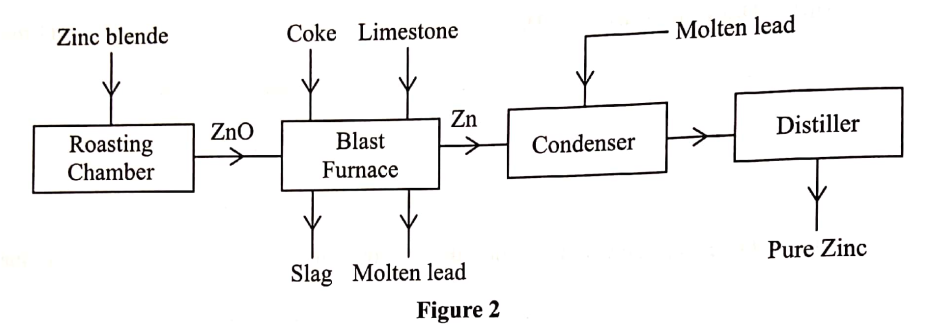
(a) Zinc occurs mainly as zinc blende. Name one other ore from which Zinc can be extracted. (1 mark)
(b) The flow chart in Figure 2 shows the various stages in the extraction of zinc metal. Study it and answer the questions that follow.
(c) Explain the observations made when zinc metal is added to hot sodium hydroxide. (2 marks)
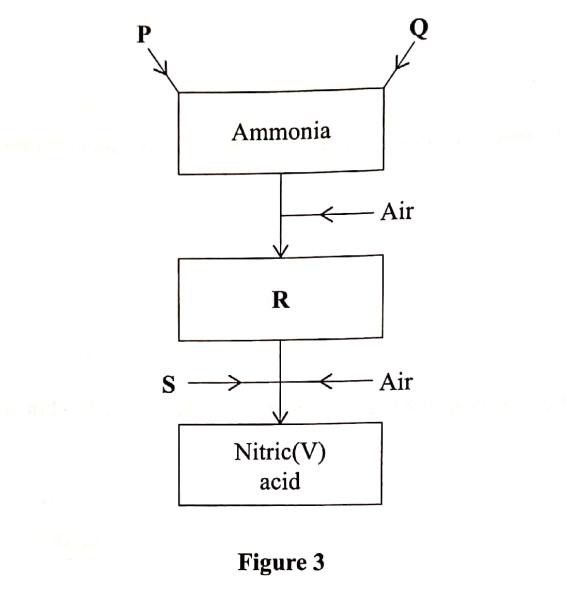
Figure 3 is a flow chart that shows the process that occurs in the manufacture of nitric (V) acid.
(a) Name substance P, Q, R and S.
To obtain substance R, ammonia is heated at 9000C in the presence of air and a catalyst.
The product is then cooled in air.
(e) When ammonia ¡s reacted with nitric(V) acid, it produces a nitrogenous fertiliser.
(a) Explain the following observations:
(b) A sample of water is suspected to contain aluminium ions (AI3+) . Describe a laboratory experiment that can be carried out to show that AI3+ ions are present in the water sample. (3 marks) (c) In an experiment to determine the number of moles of water of crystallisation of a hydrated compound, Na2S04. X H2O, 5g of the compound were heated strongly to a constant mass.
(b) When magnesium burns in air, it forms a white solid and a grey-green solid.
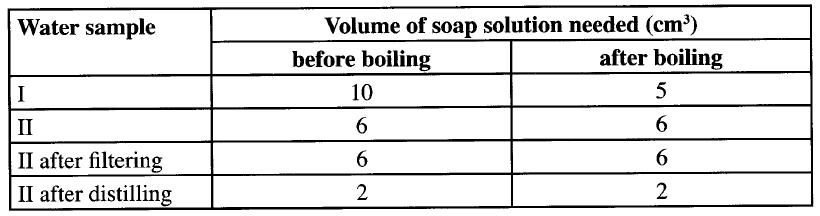
When a few drops of water are added to the mixture, a gas that turns red litmus paper blue is evolved. Identify the (i) white solid. (ii) gas evolved and state its use. (I) Name of gas (II) Use of the gas. ; (c) Two different samples of water (I and II) were tested with soap solution. Sample II was further subjected to two other processes before adding soap. 20 cm3 of each sample of water was shaken with soap solution in a boiling tube until a permanent lather was obtained. The results are shown in the table below
(i) Identify the water sample that had temporary hardness. Explain your answer.
(ii) Explain why the results for sample II are different after distilling but remain unchanged after filtering. (iii) State two disadvantages of using both water samples for domestic purposes.
(a) Other than concentration, state two factors that determine the rate of a reaction.
b) In an experiment to determine the rate of reaction, excess lambs of calcium carbonate were added to 2 M hydrochloric acid. The mass of calcium carbonate left was recorded after every 30 seconds. The results are shown in the table below
i) Write the equation for the reaction that took place
ii) On the grid provided, plot a graph of mass of calcium carbonate vertical axis Against time (iii) Determine the rate of reaction at the 105th second. (c) Why does the curve level off after some time? (d) On the same grid, sketch a curve for the same reaction using 4 M hydrochloric acid and label the curve R.
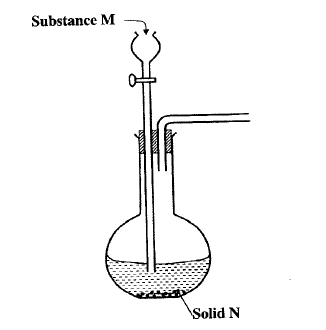
The set up below can be used to generate a gas without heating. This occurs when substance M reacts with solid N.
a i) Complete the table below giving the names of substance M and solid N if the gasses generated are chlorine and sulphur (IV) oxide.
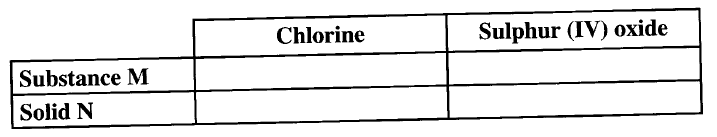
(ii) Complete the diagram above to show how a dry sample of sulphur (IV) oxide can be collected
(b) Describe two chemical methods that can be used to test the presence of sulphur (IV) oxide. (c) Other than the manufacture of sulphuric (VI) acid, state two uses of sulphur (IV) oxide.
ANSWERS
(b) Presence of SO2
- Use of acidified potassium dichromate (VI) which turns from orange to green. - Bubble gas through acidified potassium manganate (VII) which decolourises /changes i.e from purple to colourless. - Iron (III) sulphate solution - yellow/brown changes to green - Bromine water colour changes from yellow/brown / orange to colourless (c) - Fumigation - Bleaching agent - Preservative . - Disinfectant - Antioxidant
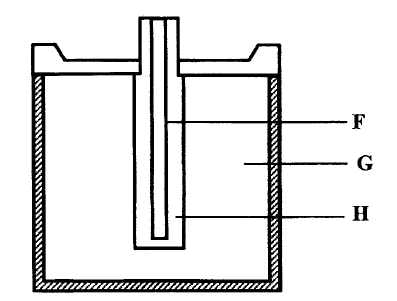
a) The diagram below represents a dry cell. Use it to answer the quest ions that follows.
i) Which of the letters represent
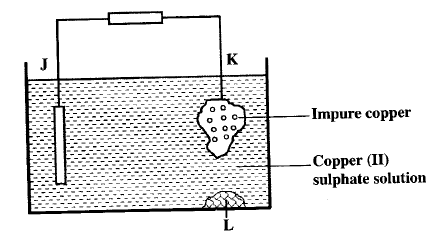
i) Carbon electrode? ii) The electrolyte? ii) One of the substances used in a dry cell is manganese (IV) oxide. State two roles of manganese (IV) oxide in the dry cells b) Below is simplified electrolytic cell used for purification of copper. Study it and answer the questions that follows.
i) Identify the cathode
ii) Write the equation for the reaction at the anode iii) What name is given to L? iv) A current of 0.6 A was passed Through the electrolyte for 2 hours. Determine the amount of copper deposited (Cu=63.5; 1 Faraday = 96,500 coulombs) v) St ate two uses of copper metal
ANSWERS
(a) (i) I F
II G (ii) - Manganese (IV) oxide oxidises hydrogen to water /depolariser . - It increases the surface area of the electrolyte (b) (i) Cathode J.
(v)Uses of copper metal - soldering bits / wires
- Electrical cables and alloys - coins, ornaments/lightening arrestors/ diodes/ - calorimeters.
(a) Name the method that can be used to obtain pure iron (III) chloride from a mixture of iron (III) chloride and sodium chloride.
(b) A student was provided with a mixture of sunflower flour, common salt and a red dye. The characteristics of the three substances in the mixture are given in the table below.
The student was provided with ethanol and any other materials needed.
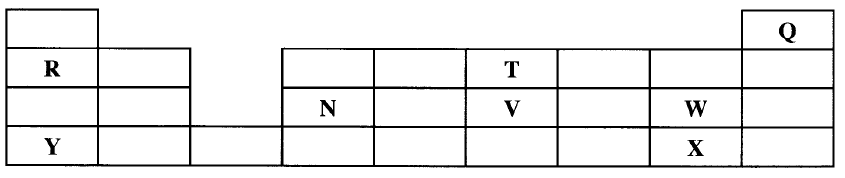
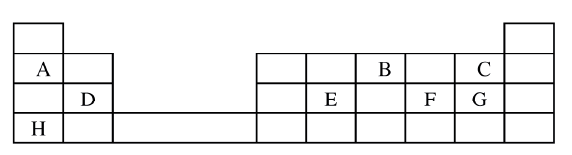
Described how the student can separate the mixture into its three components c) The diagram below show part of a periodic table. The letters do no represent the actual symbols of elements. Use the diagram to answer the questions that follow.
i) Explain why the oxidizing power of W is more than that of X
ii) How do the melting points of R and T compare? Explain iii) Sketch an element that could be used i) In weather ballons ii) For making a cooking pot d i) Classify the substances water, iodine, diamond and candle wax into elements and compounds
ii) Give one use of diamond
ANSWERS
(a)Sublimation
(b)Add ethanol to the mixture . Filter and evaporate filtrate to obtain red dye . Add water to the residue . Filter to obtain sunflower flour . Evaporate filtrate to obtain salt . OR Add H,O to mixture , filter , residue is sunflower , evaporate the water ; add ethanol to the residue filter . The filtrate is red dye. (3 marks) (c)(i) W accepts electrons more readily than X. W has small atomic radius/ W has less energy levels than X/ W has less screening effect than X/ W has greater effective nuclear attraction than X. W is more electro negative than X. (ii) T has a lower melting point than R because it exists in simple molecular form with weak Van der Waals forces while R has strong metallic bonds.
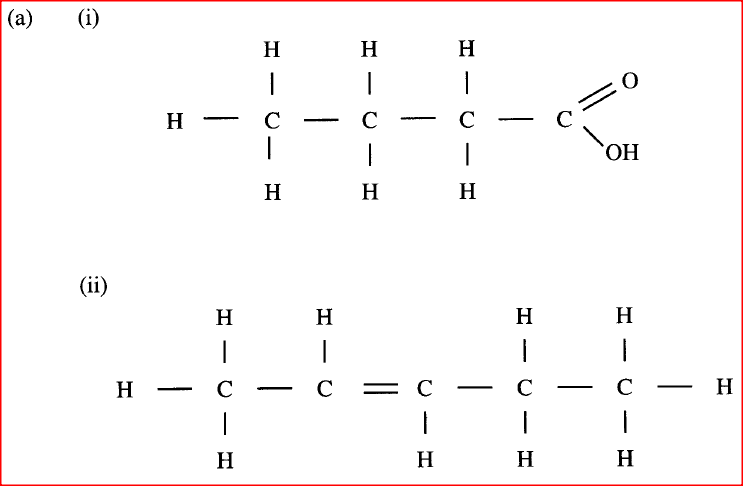
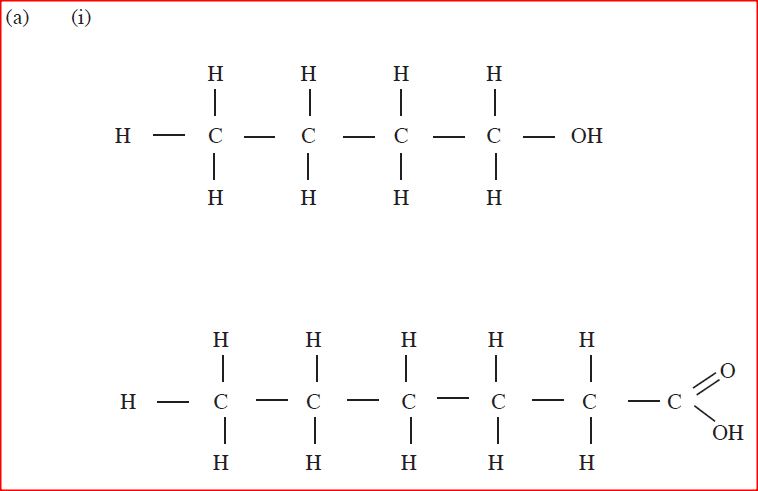
(a) Draw the structure of the folio wing compounds.
(i) Butanoic acid; (ii) Pent-2-ene. (b) Explain why propan-1-ol is soluble in water while prop-1-ene is not. (Relative molecular mass of propan-1-ol is 60 while that of prop-1-ene is 42). (c) What would be observed if a few drops of acidified potassium manganate (VII) Were added to oil obtained from nut seeds? Explain. (d) State one method that can be used to convert liquid oil from nut seeds into solid. (e) Describe how soap is manufactured from liquid oil from nut seeds (f) 0.44 g of an ester A reacts with 62.5 cm3 of 0.08 M potassium hydroxide giving an alcohol Band substance C. Given that one mole of the ester reacts with one mole of the alkali, calculate the relative molecular mass of the ester.
(a) (i) Carbon (IV) oxide is present in soft drinks. State two roles of carbon
(IV) oxide in soft drinks. (ii) Explain the observation made when a bottle containing a soft drink is opened. (iii) Carbon (IV) oxide dissolves slightly in water to give an acidic solution. Give the formula of the acid. (b) Zinc oxide can be obtained by heating zinc nitrate. A student heated 5.76 g of zinc nitrate. (i) Write an equation for the reaction that occurred. (ii) Calculate the total volume of gases produced. (Molar gas volume is 24 dm3; Zn = 65.4; O = 16.0; N = 14.0). (iii) Identify the element that is reduced when zinc nitrate is heated. Give a reason.
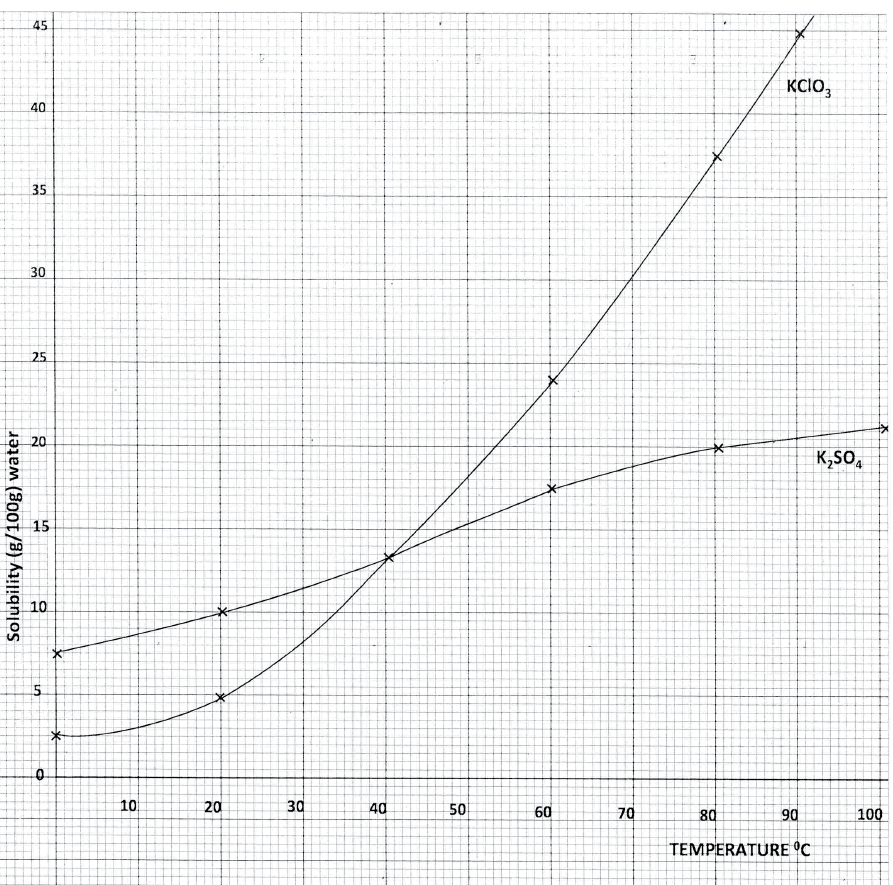
a) Dissolving of potassium nitrate in water is an endothermic process. Explain the effect of increase in temperature on the solubility of potassium nitrate
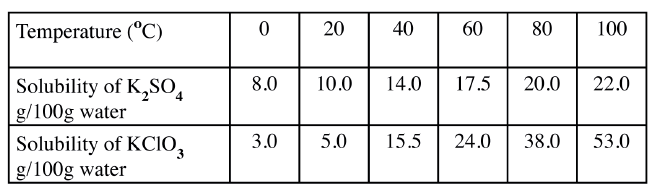
b) The table below shows the solubilities of potassium sulphate and potassium chlorate (V) at different temperatures.
i) Draw the solubility curves for both salts on the same axis. (Temperature on the X-axis
ii)A solution of potassium sulphate contains 20g of the salt dissolved in 100 g of water at 100°C. This solution is allowed to cool to 25°C I) at what temperature will crystals first appears? II) What mass of crystals will be present at 25°C? iii) Which of the two salts is more soluble at 30°C? iv) Determine the concentration of potassium sulphate in moles per litre when the solubility of the two salts are the same (K= 39.0, O=16.0 ; S=32.0) v) 100 g of water at 100°C contains 19g of potassium sulphate and 19 g of potassium chlorate (V). Describe how a solid sample of potassium sulphate at 60°C can be obtained
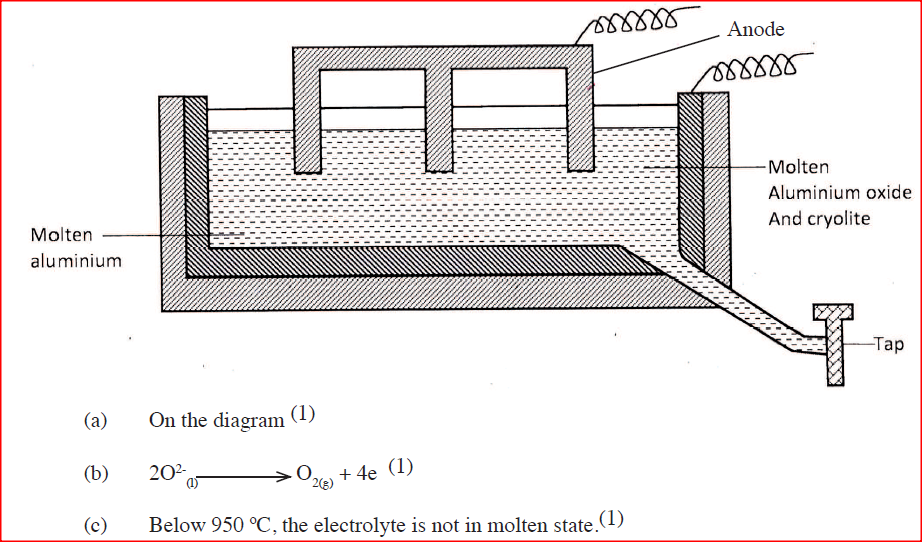
The diagram below represents a set up of an electrolytic cell that can be used in the production of aluminium
(a) One the diagram, label the anode
(b) Write the equation for the reaction at the anode (c) Give a reason why the electrolytic process is not carried out below 950°C (d) Give a reason why the production of aluminium is not carried out using reduction process (e) Give two reasons why only the aluminium ions are discharged (f) State two properties of duralumin that makes it suitable for use in aircraft industry (g)Name two environmental effects caused by extraction of aluminium
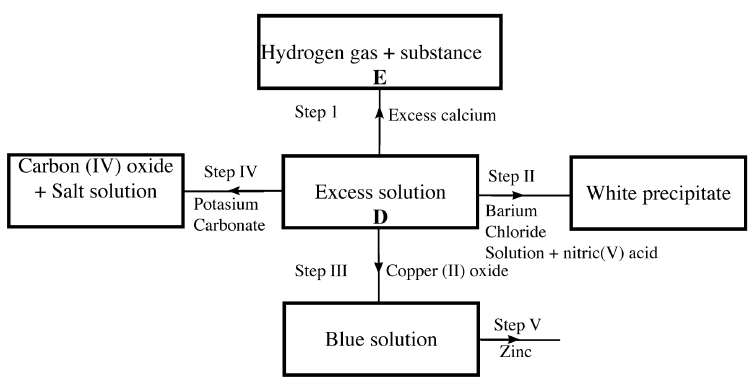
(a) The scheme below shows some of the reaction of solution D. Study it and answer the questions that follow
(i) Give a possible caution present in solution D
(ii) Write an ionic equation for the reaction in Step II (iii) What observations would be made in Step V? Give a reason (iv) Explain why the total volume of hydrogen gas produced in step 1 was found to be very low although calcium and solution D were in excess. (v) State one use of substance E. (b)Starting with solid sodium chloride, describe how a pure sample of lead (II) Chloride can be prepared in the laboratory (c) (i) State a property of anhydrous calcium chloride which makes it suitable for use as a drying agent for chlorine gas. (ii) Name another substance that can be used to dry chlorine gas
(a) Other than temperature, state two factors that determine the rate of a chemical reaction.
(b) A solution of hydrogen peroxide was allowed to decompose and the oxygen gas given off collected. After 5 minute, substance G was added to the solution of hydrogen peroxide. The total volume of oxygen evolved was plotted against time as shown in the graph below
(i)Describe the procedure of determining the rate of the reaction at minute 12.
(ii)How does the production of oxygen in region AB compare with that in region BC? Explain (iii)Write an equation to show the decomposition of hydrogen peroxide. (c) Sulphur (IV) oxide react with oxygen to form Suplhur (VI) oxide as shown in the equation below
(ii)Name one catalyst used for the reaction.
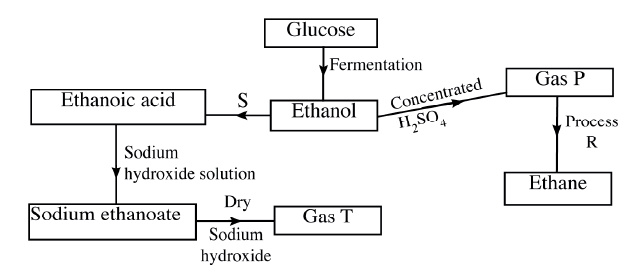
(a) Draw the structures of the following.
(i)Butan -1-ol (ii)Hexanoic acid. (b) Study the flow chart below and answer the questions that follow
(i) State the conditions necessary for fermentation of glucose to take place
(ii) State one reagent that can be used to carry out process S. (iii) Identify gases P: T: (iv) How is sodium hydroxide kept dry during the reaction (v) Give one commercial use of process R. (c) When one mole of ethanol is completely burnt in air, 1370kJ of heat energy is released. Given that 1 lire of ethanol is 780 g , calculate the amount of heat energy released when 1 litre of ethanol is completely burnt (C = 12.0; H=1.0; 0=16.0) (d) State two uses of ethanol other than as an alcoholic drink.
(a) The grid below represents part of the periodic table . Study it and answer the questions that follow. The letters are not the actual symbols of the elements
i) Select the most reactive metal. Explain
ii) Select an element that can form an ion with a charge of 3- iii) Select an alkaline earth metal iv) Which group 1 element has the highest first ionization energy? Explain v) Element A combines with chlorine to form a chloride of A. State the most likely pH value of a solution of a chloride of A. Explain (b) (i) Explain why molten calcium chloride and magnesium chloride conduct electricity while carbon tetrachloride and silicon tetrachloride do not. (ii) Under the same conditions , gaseous neon was found to diffuse faster than gaseous fluorine. Explain this observation. (F=19.0;Ne=20.0)
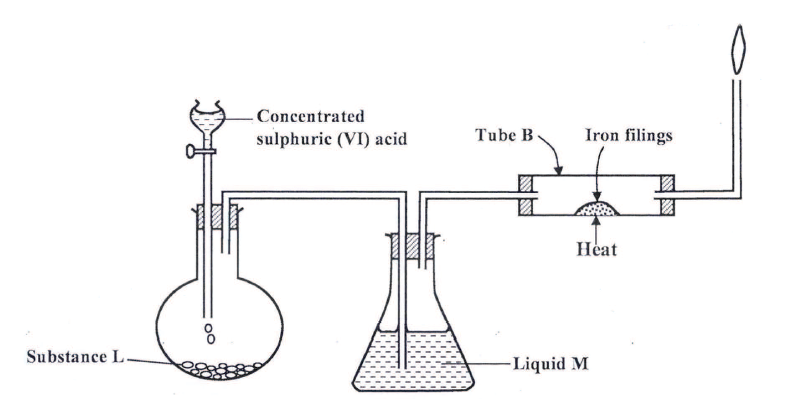
(a) The set up below was used to prepare dry hydrogen chloride gas, and investigate its effect on heated iron filings.
(i). Name substance L
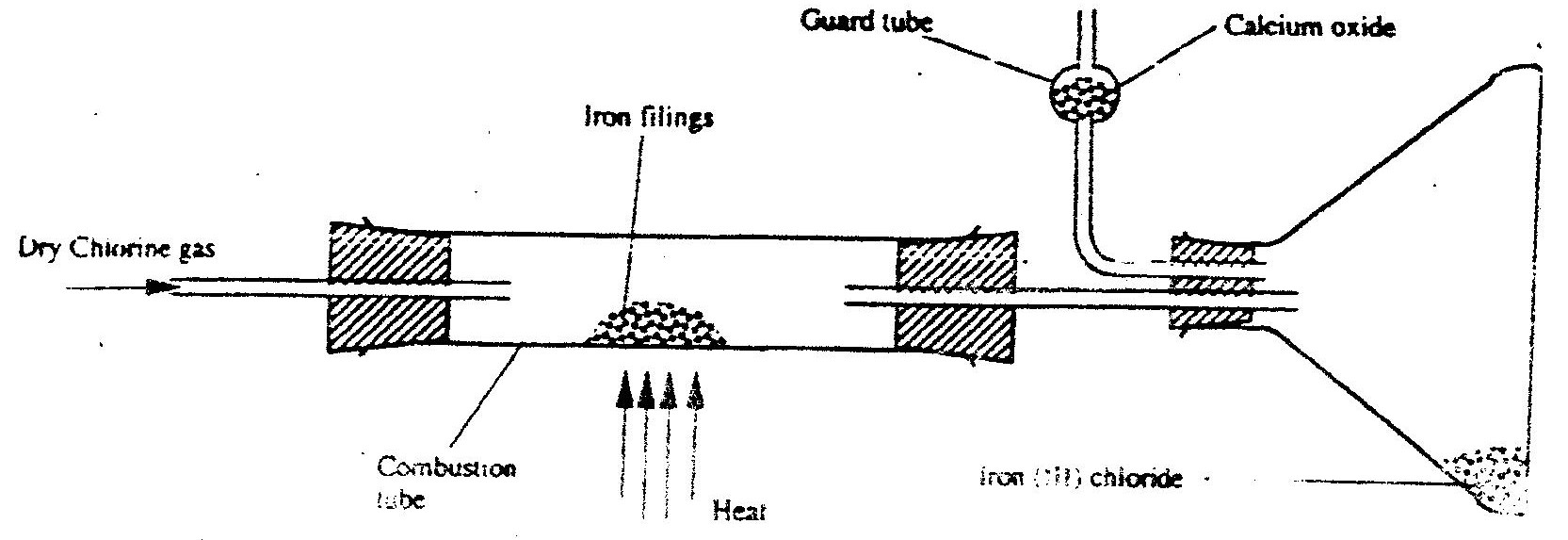
(ii) Name liquid M (iii) What will be observed in tube B? (iv) Write an equation for the reaction that occurs in tube B. (v) Why is the gas from tube B burnt? (b) (i) Explain the following observations: I) A white precipitate is formed when hydrogen chloride gas is passed through aqueous silver nitrate. II) Hydrogen chloride gas fumes in ammonia gas. (ii) State two uses of hydrogen chloride gas (c) The diagram below is a representation of an industrial process for the manufacture of a bleaching powder.
(i) Name substance Q.
(ii)When the bleaching powder is added to water during washing, a lot of soap is used. Explain
ANSWERS
(a) (i) Sodium chloride / potassium chloride /rock salt.
(ii) Concentrated sulphuric (VI) acid (iii) Grey solid turns green
(i) I The gas reacts with silver nitrate to form insoluble silver chloride
II Both gases form ammonium chloride which is white. (ii) To make hydrochloric acid. Manufacture of ammonium chloride. Manufacture of PVC. Making chloroethene /vinyl chloride
a) Study the information in the table below and answer the questions that follow.
(The letters do not represent the actual symbols of the elements).
c) Neutralization is one of the methods of preparing salts. i) What is meant by neutralization? ii) Describe how you would prepare crystals of sodium nitrate starting with 200cm3 of 2M sodium hydroxide. iii) Write an equation for the reaction that takes place when a solid sample of sodium nitrate is heated.
(Fe - = 56.0,Cl = 35.5 and Molar gas volume at 298K is 24,000cm3
C) When hydrogen sulphide gas was passed through a solution of iron (III) chloride, the following observation were made:
a) Fraction distillation of liquid air usually produces nitrogen and oxygen as the major products.
(Boiling points nitrogen = - 1960C, oxygen = -1830C)
b) Study the flow chart below and answer the questions that follow.
|
Chemistry Topics
All
Archives
December 2024
|
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |






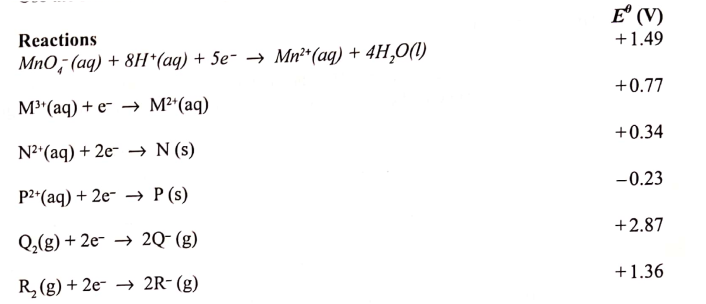




























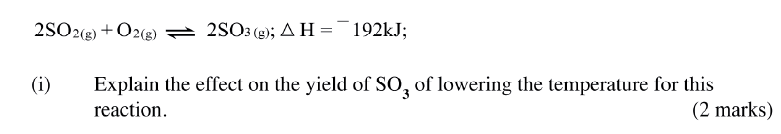












 RSS Feed
RSS Feed

