|
When 15 cm3 of a gaseous hydrocarbon, P, was burnt in 100cm3 of oxygen, the resulting gaseous mixture occupied 70cm3 at room temperature and pressure.When the gaseous mixture was passed through potassium hydroxide solution, its volume decreased to 25cm3
a) What volume of oxygen was used during the reaction? b) Determine the molecular formula of the hydrocarbon
0 Comments
A weighed sample of crystalline sodium carbonate (Na2CO3. H2O) was heated in a crucible until there was no further change in mass. Calculate the number of moles (n) of the water of crystallization
Describe how a solid sample of potassium sulphate can be prepared starting with 200cm3 of 2M potassium hydroxide.
Describe how the percentage by mass of copper in copper carbonate can be determined.
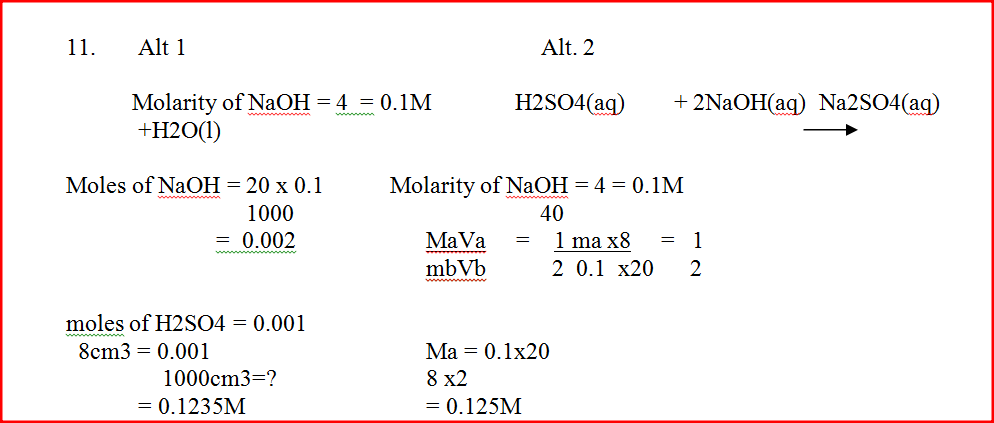
20.0cm3 of a solution containing 4 gm per litre of sodium hydroxide was neutralized by 8.0cm3 of dilute sulphuric acid. Calculate the concentration of sulphuric acid in moles per litre (Na = 23.0, O = 16.0, H = 1.0)
The empirical formula of A is CH2Br. Given that 0.470g of A occupies a volume of 56cm3 at 546K and 1 atmospheric pressure, determine its molecular formula.(H = 1.0, C = 12.0, Br = 80.0, molar gas volume at STP = 22.4 dm3).
10cm3 of concentrated sulphuric (VI) acid was diluted to 100cm3. 10cm3 of the Resulting solution was neutralised by 36cm3 of 0.1M sodium hydroxide solution. Determine the mass of sulphuric (VI) acid that was in the concentrated acid (S = 32.0; H = 1.0; O = 16.0).
The data given below was recorded when Metal M was completely burnt in air. M is not the actual symbol of the metal. (RA.M; M = 56, O ==16)
Mass of empty crucible and lid = I0.240g Mass of crucible, ;lid and metal M = 10.352g Mass crucible, lid and metal oxide = 10.400g (a) Determine the mass of: (i) Metal M (ii) oxygen. (b) Determine the empirical formula of the metal oxide.
(a) State the Gay Lussac's Law.
(b) 10cm3 of a gaseous hydrocarbon, C2HX required 30cm3 of oxygen for complete combustion. If steam and 20cm3 of carbon (IV) oxide were produced, what is the value of X?
50kg of ammonium sulphate (NH4)2SO4and 30kg of urea CO(NH2)2 fertilizers were applied in two equal sizes of plots A and B to enrich their nitrogen content.Show by working, which plot was more enriched with nitrogen. (N =14;S = 32; O = 16; C = 12; H = 1)
When lead(II) nitrate is heated, one of the products is a brown gas.
(a) Write the equation of the reaction that occurs . (b) If 0.290dm3 of the brown gas was produced, calculate the mass of the lead(II) nitrate that was heated. (R .F.M of lead (II) nitrate = 331; Molar gas volume = 24dm3 ).
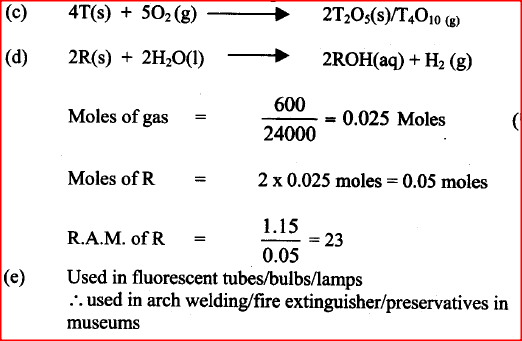
Use the information in the table below to answer the questions that follow. The letters do not represent that actual symbols of the elements.
a) Give the reasons why the meling point of:
i) S is higher than that of R ii) V is lower than that of U b) How does the reactivity of W with chlorine compare with that of R with chlorine? Explain, c) Write an equation for the reaction between T and excess oxygen
d) When 1.15g of R were reacted with water, 600cm3 of gass was produced.
Determine the relative atomic mass of R. (Molar gas volume = 24000cm3) e) Give one use of element V
ANSWERS
(a)(i) Metallic bonds in S are stronger than in R.
(ii) V is monoatomic (independent) hence weaker van der waals forces while U is diatomic hence stronger van der waals forces V has less van der waals forces while U has more van der waal forces (b) reacts more vigorously/more reactive/reacts faster - reactivity of group 1 elements increases down the group/Ionisation energy is less than that of R - easier to lose outer electron in W than in R W is more electropositive than R
Analysis of a compound showed that it had the following composition: 69.42% carbon, 4.13% hydrogen and the rest oxygen.
a) Determine the empirical formula of the compound. (C = 12.0, H = 1.0, O = 16.0) b) If the mass of one mole of the compound is 242, determine its molecular formula
The pressure of nitrogen gas contained in a 1dm3 cylinder at -196°C was 107 Pascals.
Calculate the: a) Volume of the gas at 25°C and 105 Pascals. b) Mass of nitrogen gas(Molar volume of gas is 24dm3, N = 14.0)
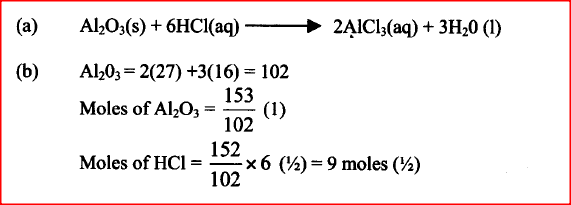
Aluminium oxide reacts with both acids and bases.
a) Write an equation for the reaction between aluminium oxide and hydrochloric acid. b) Using the equation in (a) above, calculate the number of moles of hydrochloric acid that would react completely with 153.0g of aluminium oxide. (AL = 27.0, 0= 16.0) D gm of potassium hydroxide were dissolved in distilled water to make 100cm3 of solution. 50cm3 of the solution- required 50cm3 of 2 M nitric acid for complete neutralization. Calculate the mass D, of potassium hydroxide (KOH)(aq) + HNO3(aq) →KNO3 (aq) + H2O (I) Relative formula mass of KOH = 56
4.76g of liquid ammonia and 4.76g of liquid nitrogen were each allowed warming up and hanging into gas at warm temperature and pressure. Relative atomic masses: H=1.0 and N=14.0. Using the data given above, explain which gas occupied the greater volume.
(a) An ore is suspected to contain mainly iron. Describe a method that can be used to confirm the presence of iron in the ore
(b) Excess carbon monoxide gas was passed over a heated sample of an oxide of iron as shown in the diagram below. Study the diagram and the data below it to answer the questions that follow.
Mass of empty dish = 10.98g
Mass of empty dish 4 oxide of iron = 13.30g Mass of empty dish 4 residue = 12.66g
(c) Corrosive is a destructive process in which iron which is converted into hydrated iron (III) oxide State:
(d) Explain why it is not advisable to wash vehicles using seawater
In an experiment to determine the percentage of magnesium hydroxide in an anti-acid, a solution containing 0.50 g of the anti-acid was neutralized by 23.0 cm3 of 0.010m hydrochloric acid (Relative formula mass of magnesium hydroxide =58)
a) Mass of magnesium hydroxide in the anti-acid; b) Percentage of magnesium hydroxide in the anti-acid
On complete combustion of a sample of hydrocarbon, 3.52 gm of carbon dioxide and 1.44 gm of water were formed. Determine the molecular formula of the hydrocarbon.
(Relative molecular masses of hydrocarbon = 56, carbon dioxide 44, water = 18 and relative atomic masses H = 1.0 and c = 12.0)
Phosphoric acid is manufactured from calcium phosphate according to the following equation.
Ca3(PO4)2(s) + 3H2SO4(l) → 2H3PO4(aq) + 3 CaSO4(s) Calculate the mass in (Kg) of phosphoric acid that would be obtained if 155 Kg of calcium phosphate reacted completely with the acid (Ca=40, P=31, S=32, O=16, H=1)
When a hydrated sample of calcium sulphate CaSO4 .XH2O was heated until all the water was lost, the following data recorded;
Mass of crucible = 30.296 g Mass of crucible +hydrated salt = 33.111 g Mass of crucible + anhydrous salt = 32.781 g Determine the empirical formula of the hydrated salt (Relative formula mass of CaSO4 =136, H2O =18).
6.84g of aluminium sulphate were dissolve in 150cm3 of water. Calculate the molar concentration of the sulphate ions in the solution. (Relative formula mass of aluminium sulphate is 342)
15.0cm3 of ethanoic acid (CH3COOH) was dissolved in water to make 500cm3 of solution. Calculate the concentration of the solution in moles per litre. (C=12.0;H=1.0;O=16.0; density of ethanoic acid is 1.05 g/cm3)
90cm3 of 0.01M calcium hydroxide were added to a sample of water containing 0.001 moles of calcium hydrogen carbonate. a) Write an equation for the reaction which took place b) Calculate the number of moles of calcium ions in 90cm3 of 0.01M calcium hydroxide. c) What would be observed if soap solution was added drop wise to a sample of the water after the addition of calcium hydroxide? Give a reason. |
Chemistry Topics
All
Archives
December 2024
|
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |


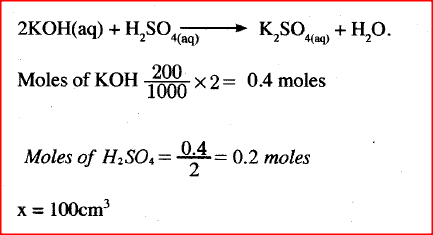




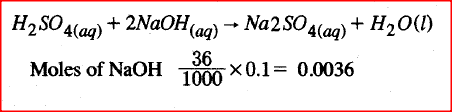
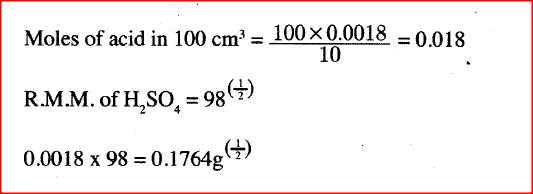








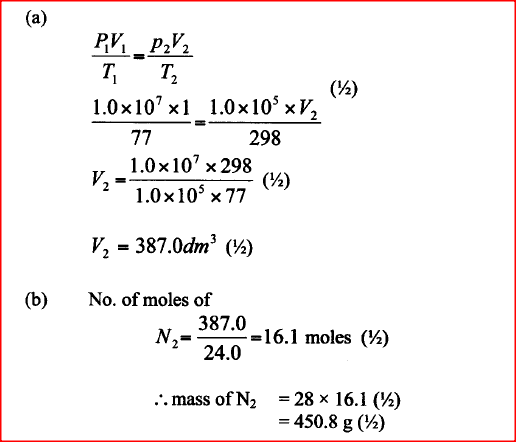









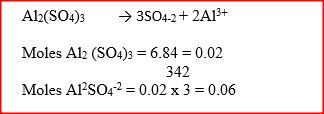



 RSS Feed
RSS Feed

