|
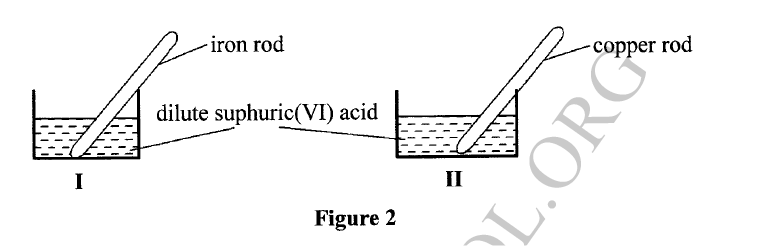
(a) A student used Figure 2 to investigate the action of dilute sulphuric(VI) acid on some metals. Beaker I and II contained equal volumes of dilute sulphuric(VI) acid. To beaker I, a clean iron rod was dipped and to beaker II, a clean copper rod was dipped.
(i) Why was it necessary to clean the metal rods?
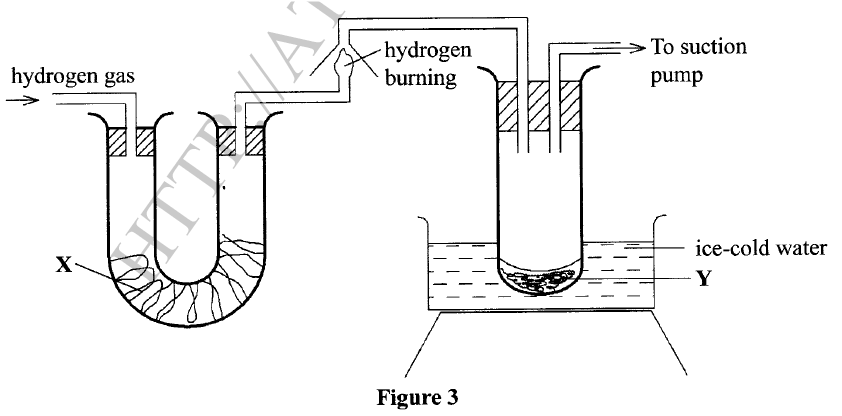
(ii) Describe the observations made in each beaker. Beaker I: Beaker II: (iii) Explain the observations in (a) (ii). (b) Figure 3 shows the apparatus used to burn hydrogen in air. Use it to answer the questions that follow.
(i)State the role of substance X.
(ii) Give the name of the substance that could be used as X. (iii) State the role of the suction pump. (iv) Name the product Y formed. (v) Give a simple physical test to prove the identity of Y. vi) State the difference between 'dry' and 'anhydrous'.
ANSWERS
(a) (i) To remove oxide layer on the metal.
(ii) Beaker I : . Bubbles of a colourless gas / effervescence ; . Solution turns green; . the size of iron rod decreases Beaker II: . The solution remained colourless. . No bubbles/effervescence
OR
Iron is more reactive than hydrogen hence it reacts with sulphuric(VI) acid to produce hydrogen gas and iron(III) sulphate which is green. Beaker II: Copper is below hydrogen hence no reaction will take place. (b) (i) To dry hydrogen gas. (ii) Calcium oxide /anhydrous calcium chloride /silica gel. Related Chemistry Questions and Answers on Water and Hydrogen Form 1 Level
0 Comments
Leave a Reply. |
Chemistry Topics
All
Archives
December 2024
|
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |



 RSS Feed
RSS Feed

