|
(a) The table below shows the standard reduction potentials for four half- cells.
Study it and answer the questions that follow. (Letters are not the actual symbols of the elements)
(i) Identify the strongest reducing agent
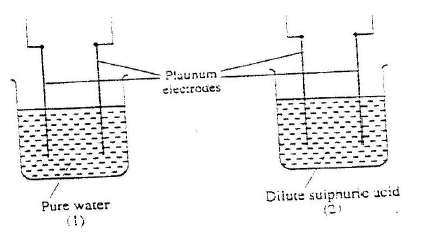
(ii) Write the equation for the reaction which takes place when solid G is added to a solution containing H2+ ions (iii) Calculate the Eθ value for the reaction in (ii) above (b) The diagram below shows the apparatus that can be used to electrolyze acidified water to obtain hydrogen and oxygen gases. Study it and answer the questions that follow
(i) Identify the electrode at which oxidation takes place
(ii) Give a reason why it is necessary to acidify the water (iii) Explain why hydrochloric acid is not used to acidify the water (c) During electrolysis of aqueous copper (II) sulphate, 144750 coulombs of electricity were used. Calculate the mass of copper metal that was obtained Cu = 64 ; 1 Faraday = 96500 coulombs)
0 Comments
When a current of 0.82A was passed for 5 hours through an aqueous solution of metal Z, 2.65 g of the metal were deposited. Determine the charge on the ions of metal Z. ( 1 Faraday = 96500 Coulombs:
Relative atomic mass of Z = 52
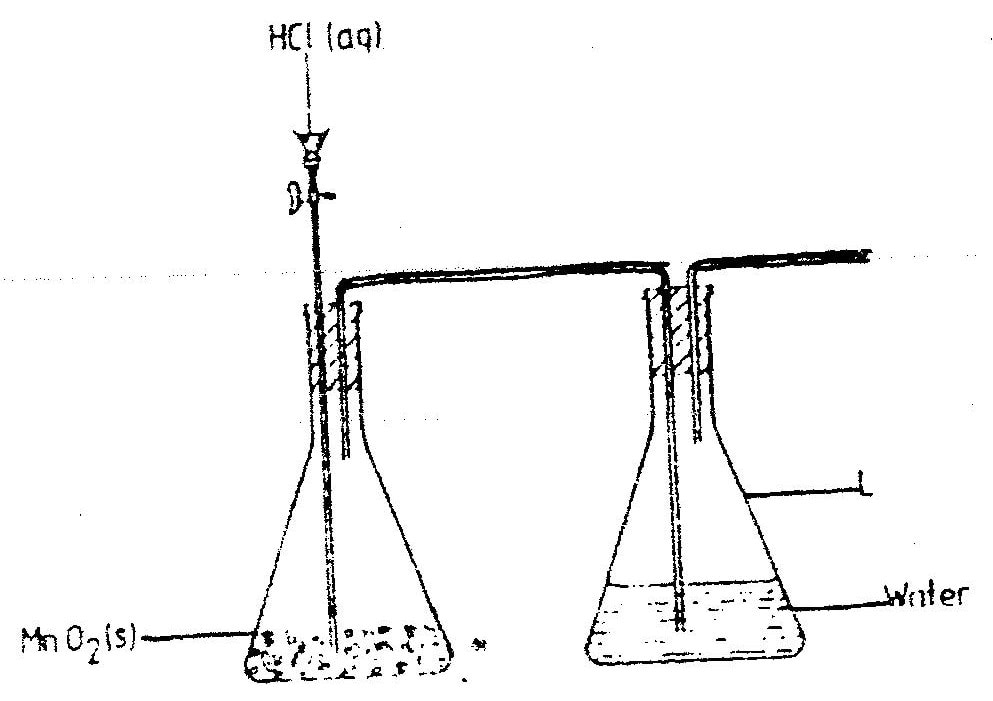
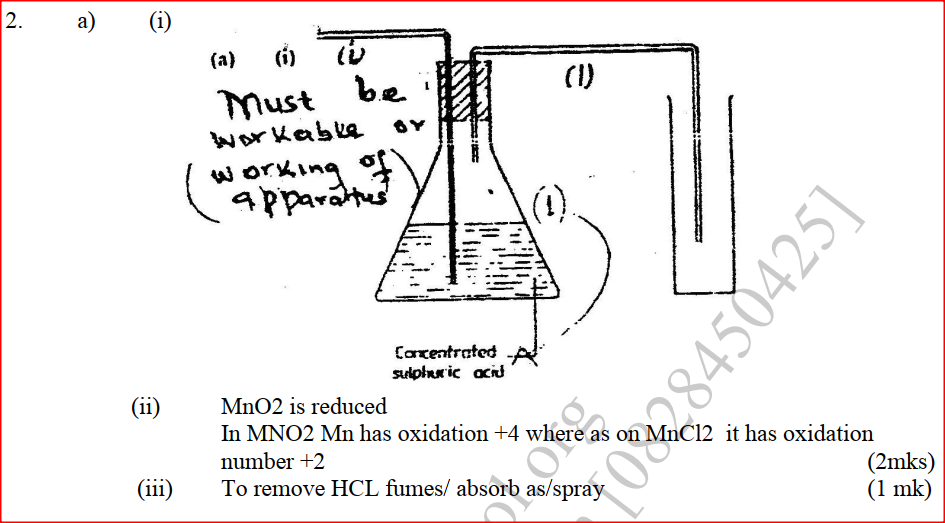
a) The diagram below shows incomplete set – up of the laboratory and preparation collection of chlorine gas. Study it and answer the questions that follow.
i) Complete the set – up to show how dry chloride gas may be collected.
ii) The equation for the redox reaction that takes place is
Explain, using oxidation numbers, which species is reduced (2mks)
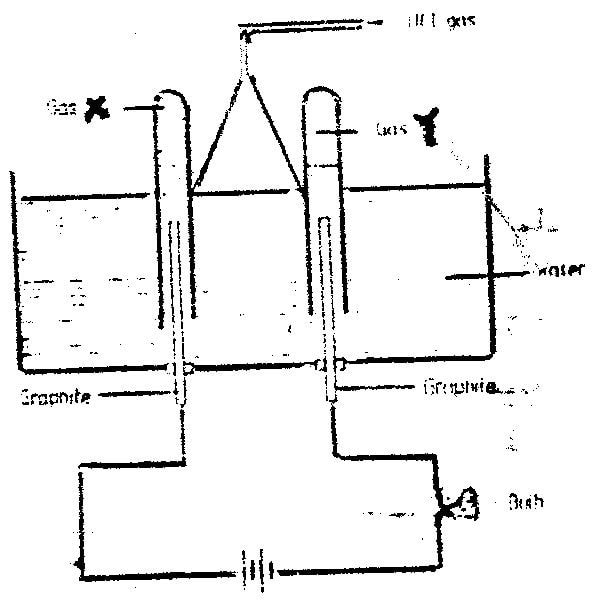
iii) What is the purpose of water in flask L? (1mk) b) Study the diagram below and answer the questions that follow.
When some hydrogen chlorides gas is allowed into water and the mixture stirred, the bulb lights and gases X and Y are formed.
i) Name: Gas X Gas Y ii) Explain why the bulb does not light before the hydrogen chloride gas is let into water. (2mks) iii) Explain using equations why the volume of gas X is less than that of gas (2mks)
(a) Brine usually contains soluble calcium and magnesium salts. Explain how sodium carbonate is used to purify brine.
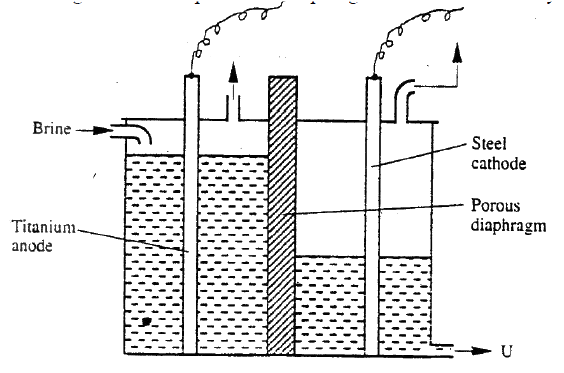
b) The diagram below represents a diaphragm cell used to electrolysed pure brine
i) Write the equations for the reactions that take place at
I Cathode II Anode ii) Name: I Product at U II Another material that can be used instead of titanium III The impurity present in the product at U iii) State two functions of the diagram c) Give one industrial use of the product at U.
ANSWERS
answers
Study the set – up below and answer the question that follows.
State and explain the observations that would be made when the circuit is completed.
Explain why it is not advisable to use aqueous chloride solution as the salt bridge in the electrochemical cell formed between half cells, Pb2-(aq)/pb(s) E0 = 0.13V and CU2 + (aq) + (aq)/CU2+(aq)/Cu2(s) E0=0.34V (2mks)
expected response
Sodium chloride will remove Pb from the insoluble pbC12. This affects the value of the cell voltage.
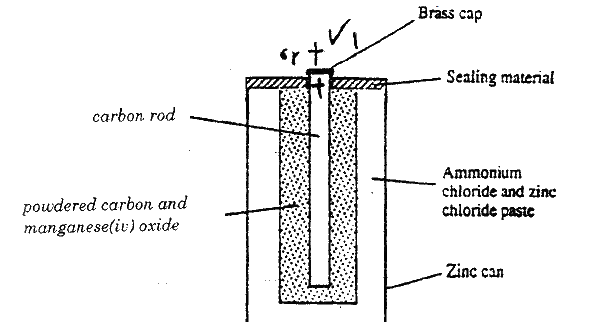
a) The diagram below is a cross- section of a dry cell. Study it and answer the questions that follow.
i) On the diagram, show with a (+) sign the positive terminal
ii) Write the equation for the reaction in which electrons are produced
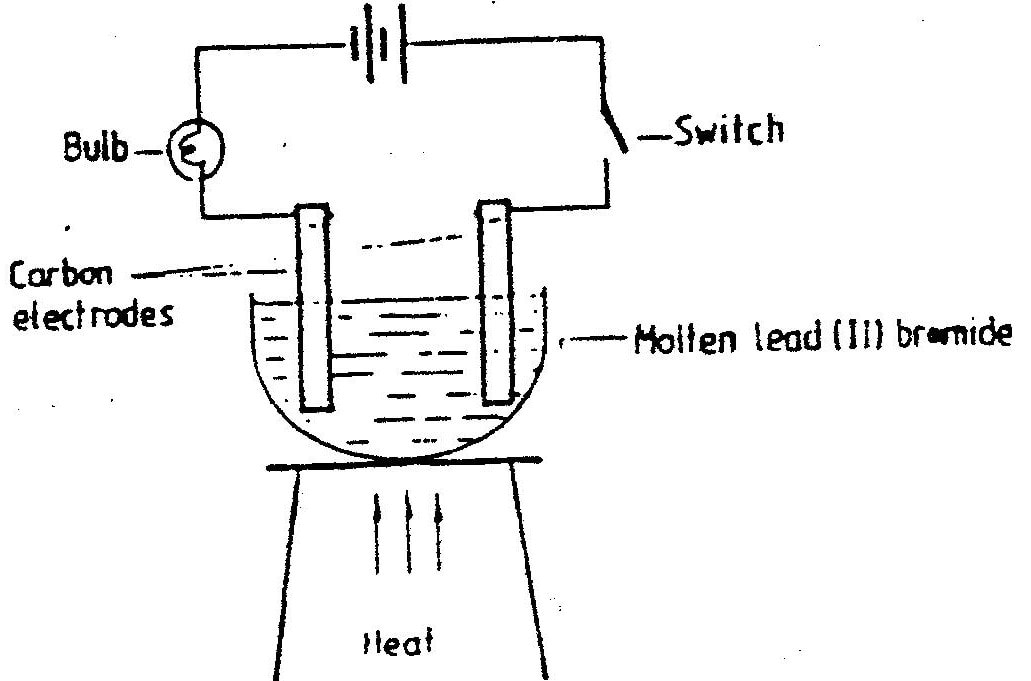
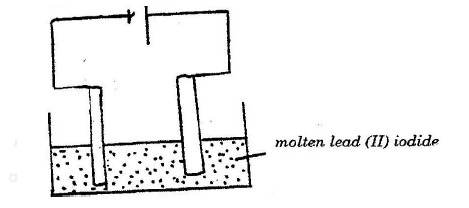
iii) The zinc can is lined with ammonium chloride and zinc chloride paste. iv) Give one advantage and one disadvantage of dry cells. b) The set – up below was used to (electrolyse roller lead (II) ionide.
i) State the observation that was made at the anode during the electrolysis.
Give a reason for your answer. ii) A current of 0.5 A was passed for two hours. Calculate the mass of lead that was deposited (Pb = 1F = 9,500C)
During purification of copper by electrolysis, 1.48g of copper were deposited when current was passed through aqueous copper(II) sulphate for 2 1/2 hours. Calculate (CU = 63.5, 1 Faraday = 96,500C).
The table below gives standard electrode potentials for the metals represented by the Letters D, E, F and G. study it and answer the questions that follow.
(a) Which metal can be displaced from a solution of its salts by all the other metals in the table? Give a reason
(b) Metals F and G were connected to form a cell as shown in the diagram below
(i) Write the equation for the reactions that occur at electrodes
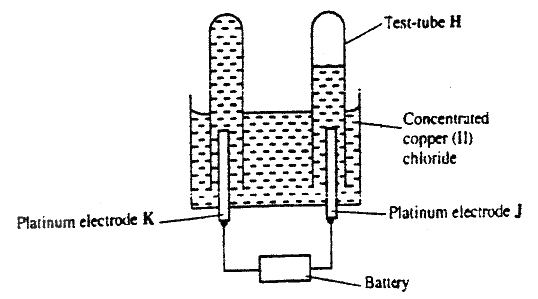
F G (ii) On the diagram, indicate with an arrow the direction in which electrons would flow on the diagram above (ii) What is the function of the salt bridge? (c) An electric current was passed through a concentrated solution of copper (II) chloride as shown in the diagram below
(i) Explain the observation that would be made on the electrolyte as the experiment progresses
(ii) After sometime, test- tube H was found to contain a mixture of two gases. Explain this observation (iii) Which of the electrodes is the anode? Explain
Study the flow chart below and answer the questions follow
(a) Identify substance
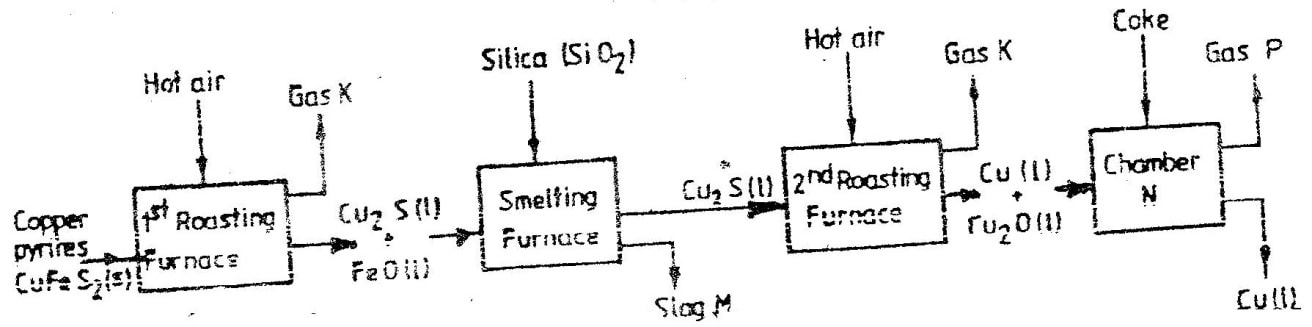
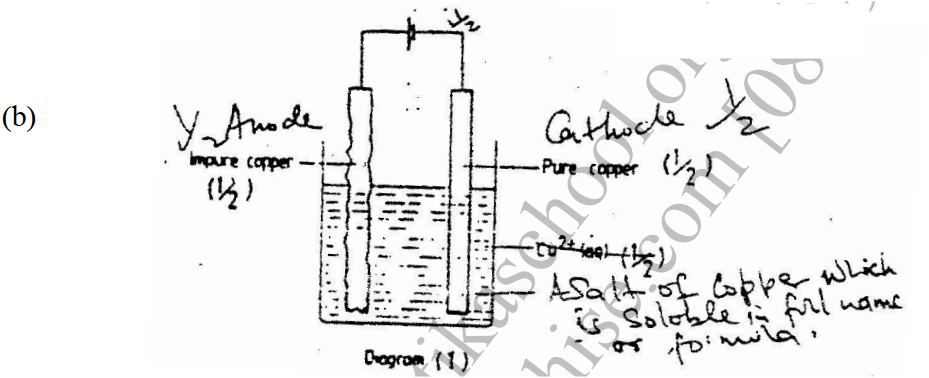
(i) A (ii) B (b) Name process C (c) Give one use of PVC (d) Write an equation for the reaction in which chlorine gas is produced (e) State and explain the observation that would be made if chlorine gas was bubbled into an aqueous solution of sodium iodide (f) In the preparation of a bleaching agent (Sodium hypochlorite), Excess chlorine gas was bubbled into 15 litres of cold 2 m sodium hydroxide (i) Write an equation for the reaction between chlorine gas and cold dilute sodium Hydroxide (ii) Calculate the: Number of moles of sodium hydroxide used Mass in kilograms of the sodium hypochlorite produced = 1. 1175 The flow chart below outlines some of the process involved during extraction of copper from copper pyrites. Study it and answer the questions that follow. (i) Name gas K (1mk) (b) The copper obtained from chamber N is not pure. Draw a labeled diagram to show the set up you would use to refine the copper by electrolysis. (3mks) (c) Given that the mass of copper obtained from above extraction was 210kg, determine the percentage purity of the ore(copper pyrites) if 810kg of it was fed to the 1st roasting furnace. (Cu = 63.5, Fe = 56.0, s=32.0) (3mks) (d) Give two effects that this process could have on the environment (2mks) Explain the following observation. A chloride dissolves in water to form an electrolyte while the same chloride dissolves in methylbenzene to for a non – electrolyte. (1mk)
Expected Response
The chloride form ions in water which conduct electric current. NO ions are formed in methylbenzene /chloride exists in methylbenzene as molecules. (2mks)
|
Chemistry Topics
All
Archives
December 2024
|
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |
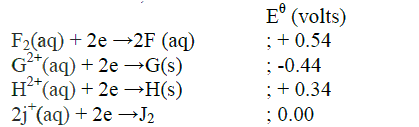

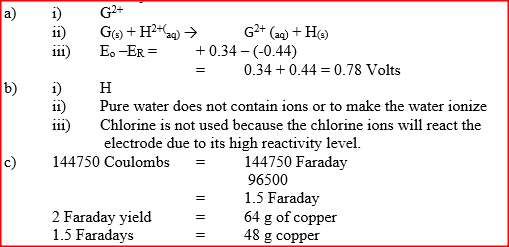
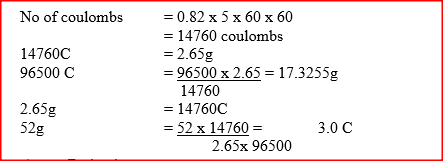
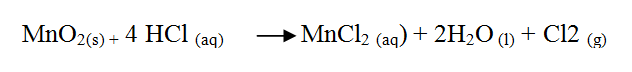





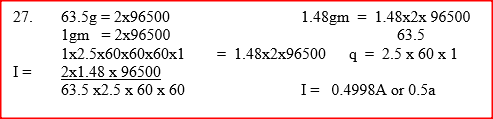










 RSS Feed
RSS Feed

