KCSE CHEMISTRY QUESTIONS AND ANSWERS PER TOPIC
|
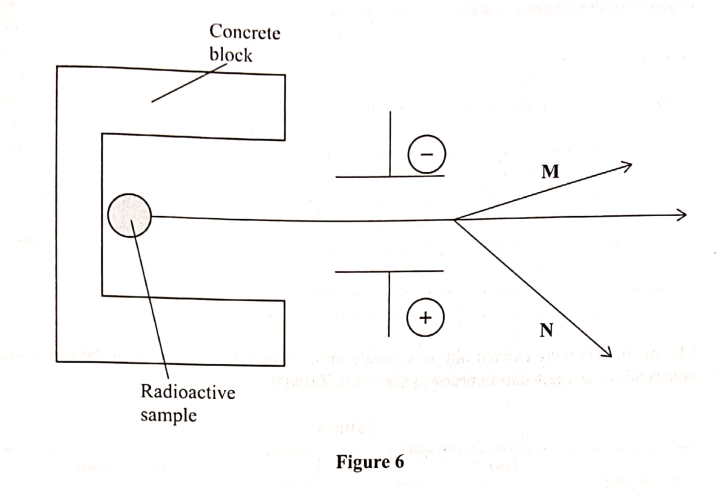
The diagram in Figure 6 Shows radiations emitted by a radioactive sample.
0 Comments
(b) When magnesium burns in air, it forms a white solid and a grey-green solid.
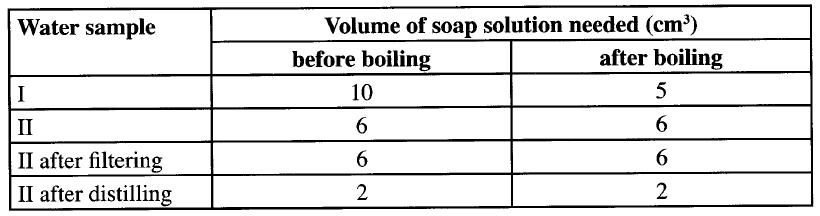
When a few drops of water are added to the mixture, a gas that turns red litmus paper blue is evolved. Identify the (i) white solid. (ii) gas evolved and state its use. (I) Name of gas (II) Use of the gas. ; (c) Two different samples of water (I and II) were tested with soap solution. Sample II was further subjected to two other processes before adding soap. 20 cm3 of each sample of water was shaken with soap solution in a boiling tube until a permanent lather was obtained. The results are shown in the table below
(i) Identify the water sample that had temporary hardness. Explain your answer.
(ii) Explain why the results for sample II are different after distilling but remain unchanged after filtering. (iii) State two disadvantages of using both water samples for domestic purposes.
(a) Other than concentration, state two factors that determine the rate of a reaction.
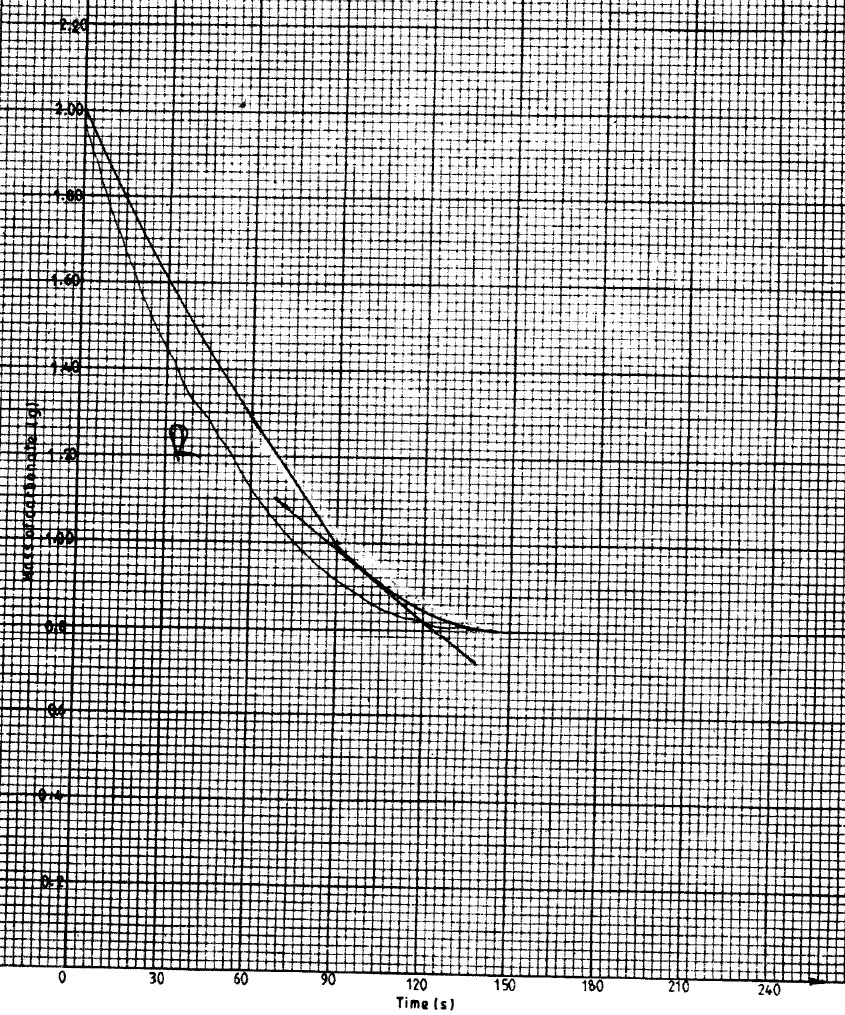
b) In an experiment to determine the rate of reaction, excess lambs of calcium carbonate were added to 2 M hydrochloric acid. The mass of calcium carbonate left was recorded after every 30 seconds. The results are shown in the table below
i) Write the equation for the reaction that took place
ii) On the grid provided, plot a graph of mass of calcium carbonate vertical axis Against time (iii) Determine the rate of reaction at the 105th second. (c) Why does the curve level off after some time? (d) On the same grid, sketch a curve for the same reaction using 4 M hydrochloric acid and label the curve R.
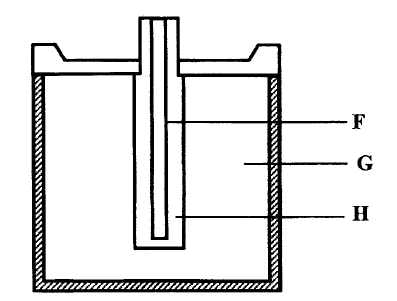
a) The diagram below represents a dry cell. Use it to answer the quest ions that follows.
i) Which of the letters represent
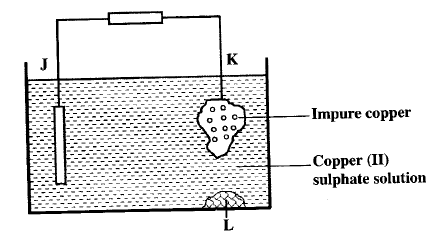
i) Carbon electrode? ii) The electrolyte? ii) One of the substances used in a dry cell is manganese (IV) oxide. State two roles of manganese (IV) oxide in the dry cells b) Below is simplified electrolytic cell used for purification of copper. Study it and answer the questions that follows.
i) Identify the cathode
ii) Write the equation for the reaction at the anode iii) What name is given to L? iv) A current of 0.6 A was passed Through the electrolyte for 2 hours. Determine the amount of copper deposited (Cu=63.5; 1 Faraday = 96,500 coulombs) v) St ate two uses of copper metal
ANSWERS
(a) (i) I F
II G (ii) - Manganese (IV) oxide oxidises hydrogen to water /depolariser . - It increases the surface area of the electrolyte (b) (i) Cathode J.
(v)Uses of copper metal - soldering bits / wires
- Electrical cables and alloys - coins, ornaments/lightening arrestors/ diodes/ - calorimeters.
(a) Name the method that can be used to obtain pure iron (III) chloride from a mixture of iron (III) chloride and sodium chloride.
(b) A student was provided with a mixture of sunflower flour, common salt and a red dye. The characteristics of the three substances in the mixture are given in the table below.
The student was provided with ethanol and any other materials needed.
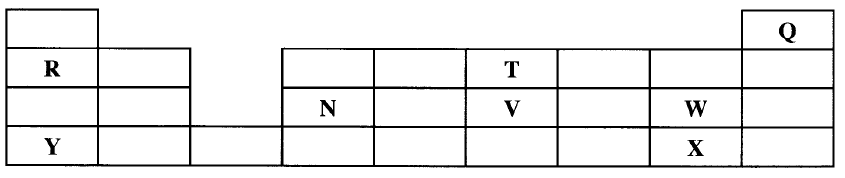
Described how the student can separate the mixture into its three components c) The diagram below show part of a periodic table. The letters do no represent the actual symbols of elements. Use the diagram to answer the questions that follow.
i) Explain why the oxidizing power of W is more than that of X
ii) How do the melting points of R and T compare? Explain iii) Sketch an element that could be used i) In weather ballons ii) For making a cooking pot d i) Classify the substances water, iodine, diamond and candle wax into elements and compounds
ii) Give one use of diamond
ANSWERS
(a)Sublimation
(b)Add ethanol to the mixture . Filter and evaporate filtrate to obtain red dye . Add water to the residue . Filter to obtain sunflower flour . Evaporate filtrate to obtain salt . OR Add H,O to mixture , filter , residue is sunflower , evaporate the water ; add ethanol to the residue filter . The filtrate is red dye. (3 marks) (c)(i) W accepts electrons more readily than X. W has small atomic radius/ W has less energy levels than X/ W has less screening effect than X/ W has greater effective nuclear attraction than X. W is more electro negative than X. (ii) T has a lower melting point than R because it exists in simple molecular form with weak Van der Waals forces while R has strong metallic bonds.
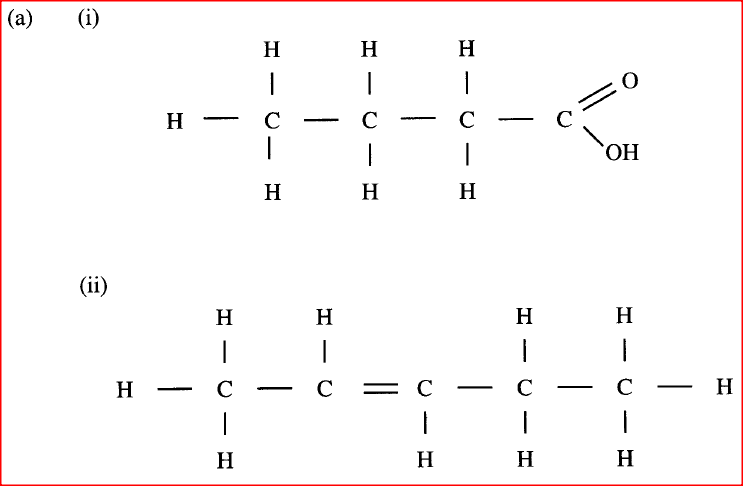
(a) Draw the structure of the folio wing compounds.
(i) Butanoic acid; (ii) Pent-2-ene. (b) Explain why propan-1-ol is soluble in water while prop-1-ene is not. (Relative molecular mass of propan-1-ol is 60 while that of prop-1-ene is 42). (c) What would be observed if a few drops of acidified potassium manganate (VII) Were added to oil obtained from nut seeds? Explain. (d) State one method that can be used to convert liquid oil from nut seeds into solid. (e) Describe how soap is manufactured from liquid oil from nut seeds (f) 0.44 g of an ester A reacts with 62.5 cm3 of 0.08 M potassium hydroxide giving an alcohol Band substance C. Given that one mole of the ester reacts with one mole of the alkali, calculate the relative molecular mass of the ester.
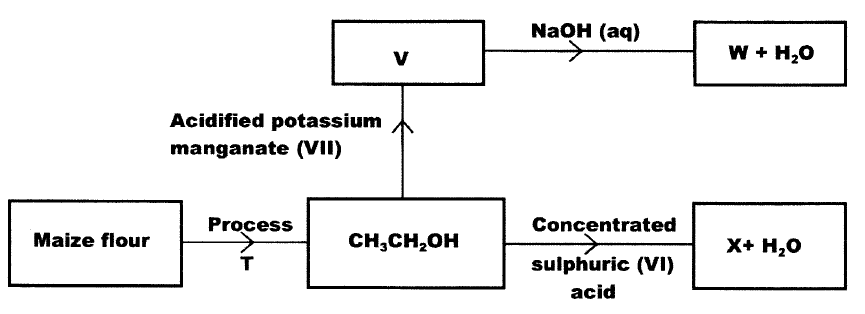
Study the flow chart below and use it to answer the questions that follows
a) Name process T
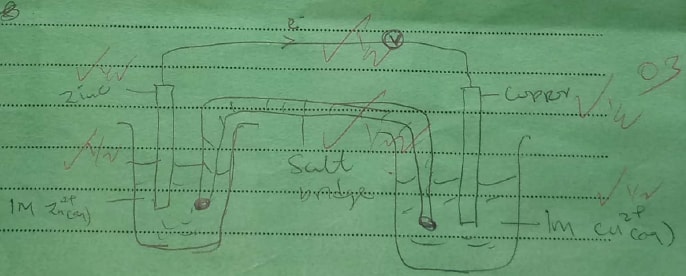
b) Give the formula of W. c) State two uses of X Given that the E° of Cu(s) /Cu2+(aq) is +0.34 V and that of Zn (s)/Zn2+(aq) is —0.76 V, draw a labelled diagram of zinc and copper electrochemical cell. (3 marks)
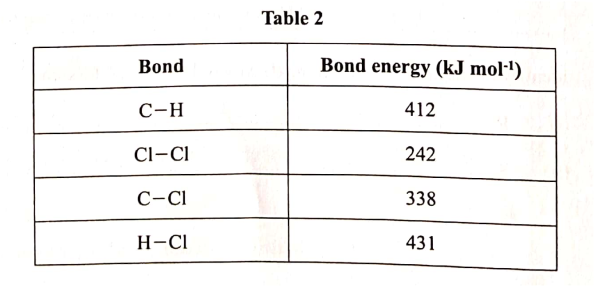
Use the information in Table 2 to answer the questions that follow.
(a) State what is meant by heat of reaction. (1 mark)
(b) Calculate the heat change when one mole of methane reacts completely with excess chlorine in the presence of UV light. (2 marks)
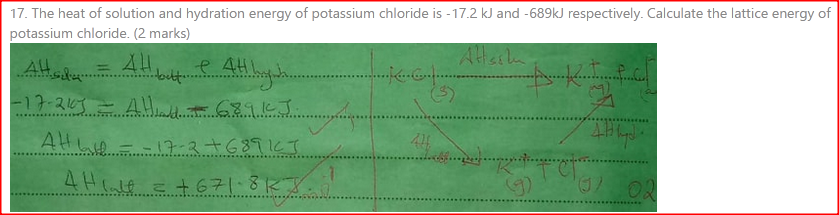
The heat of solution and hydration energy of potassium chloride is —17.2kJ and —689kJ respectively. Calculate the lattice energy of potassium chloride. (2 marks)
A radioactive substance weighing M kg took 1900 years for the original mass to reduce to 15 kg. Given that the half life of the radioactive substance is 380 years;
(a) Determine the original mass of the radioactive substance. (b) State two uses of radioactivity in medicine. .
Study the standard electrode potentials in the table below and answer the questions that follow.
(a) Which of the metals is the strongest reducing agent?
(b) What observations will be made if a silver coin was dropped into an aqueous solution of copper (II) sulphate? Explain.
ANSWERS
(a) Calcium
(b) No observable change silver is below copper in the reactivity series so it cannot displace it.
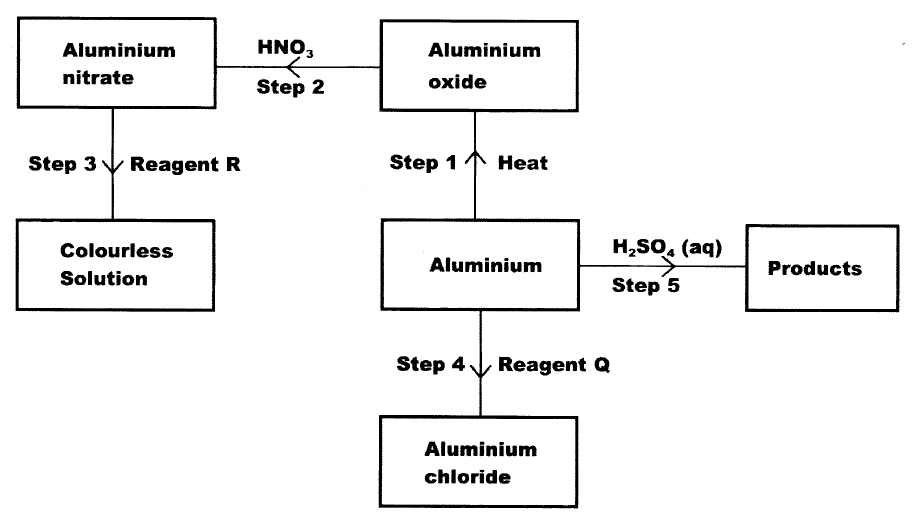
The flow chart below shows various reactions of aluminium metal. Study it and answer the questions that follow
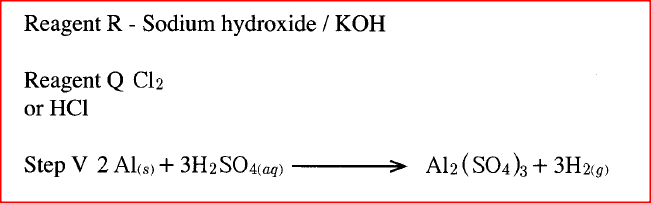
a) i) Other than water, name another reagent that could be R
ii) Write the formula of reagent Q b) Write an equation or the reaction in step 5.
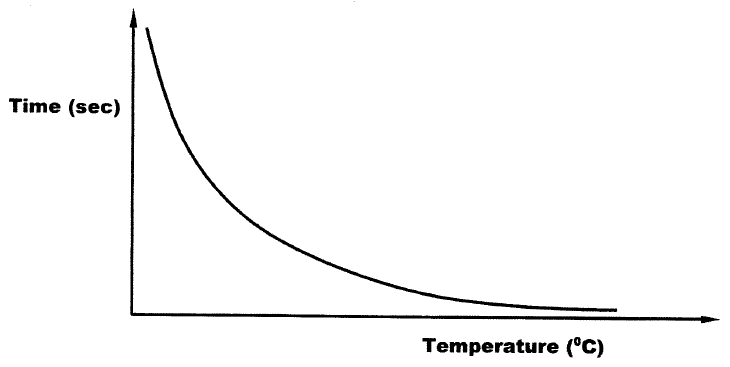
The curve shown below shows the variation of time against temperature for the reaction between sodium thiosulphate and hydrochloric acid.
a) Write the equation for the reaction between sodium thiosulphate and dilute hydrochloric acid
b) Explain the shape of the curve
ANSWERS
Dilute sulphuric (VI) acid was electrolysed using platinum electrodes.
Name the product formed at the anode and give a reason for your answer.
When solid A was heated strongly, it gave off water and a solid residue. When water was added to the solid residue, the original solid A, was formed
(a) What name is given to the process described? (b) Give one example of solid A
ANSWERS
(a)Type of reaction: Reversible reaction/temporary reaction.
(b)Copper (II) Sulphate salt (Crystals) Copper (II) Chloride hydrated. Any other hydrated salts e.g. Cobalt (II) Chloride
Describe how sodium carbonate is used to remove water hardness
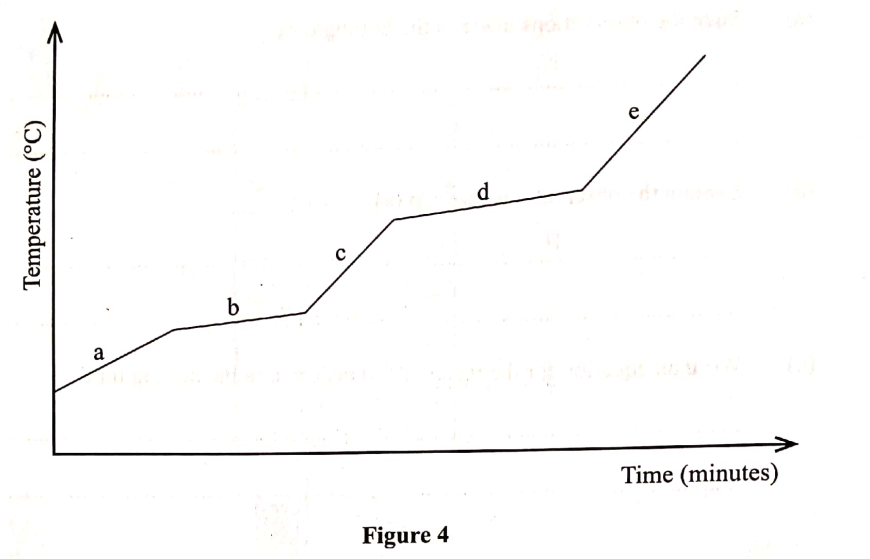
The graph in Figure 4 was obtained when a certain substance was heated and its temperature recorded at regular intervals.
(a) State the purity of the substance. (I mark)
(b) Explain the answer in (a). (2 marks)
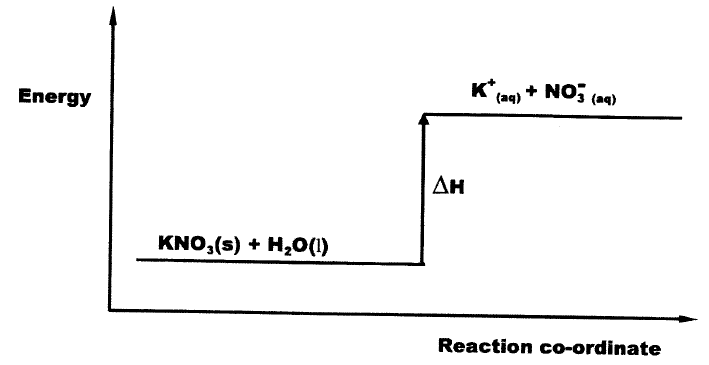
(a) What is meant by lattice energy?
b) Study the energy level diagram below and answer the question that follows
What type of reaction is represented by the diagram?
ANSWERS
(a) Enthalpy change, when one mole of crystal lattice is broken into its ions in gaseous state.
(b) Endothermic reaction (process)
(a) Name the raw material from which sodium is extracted.
(b) Give a reason why sodium is extracted using electrolysis. (c) Give two uses of sodium metal.
ANSWERS
(a)Brine (NaCl)
(b) Sodium is very reactive (use electrolysis) More reactive than carbon. (c) Uses Sodium lamps, coolant in nuclear reactors Sodium cyanide, sodium amalgam Na202 , Extraction of titanium,
(a) Give the name of the first member of the alkene homologous series.
(b) Describe a chemical test that can be used to distinguish butanol from butanoic acid
a) Dissolving of potassium nitrate in water is an endothermic process. Explain the effect of increase in temperature on the solubility of potassium nitrate
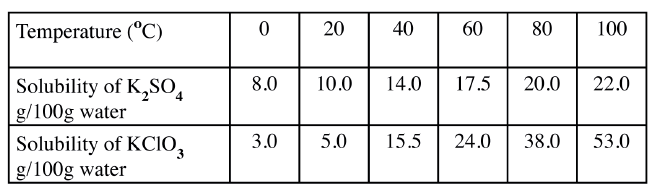
b) The table below shows the solubilities of potassium sulphate and potassium chlorate (V) at different temperatures.
i) Draw the solubility curves for both salts on the same axis. (Temperature on the X-axis
ii)A solution of potassium sulphate contains 20g of the salt dissolved in 100 g of water at 100°C. This solution is allowed to cool to 25°C I) at what temperature will crystals first appears? II) What mass of crystals will be present at 25°C? iii) Which of the two salts is more soluble at 30°C? iv) Determine the concentration of potassium sulphate in moles per litre when the solubility of the two salts are the same (K= 39.0, O=16.0 ; S=32.0) v) 100 g of water at 100°C contains 19g of potassium sulphate and 19 g of potassium chlorate (V). Describe how a solid sample of potassium sulphate at 60°C can be obtained
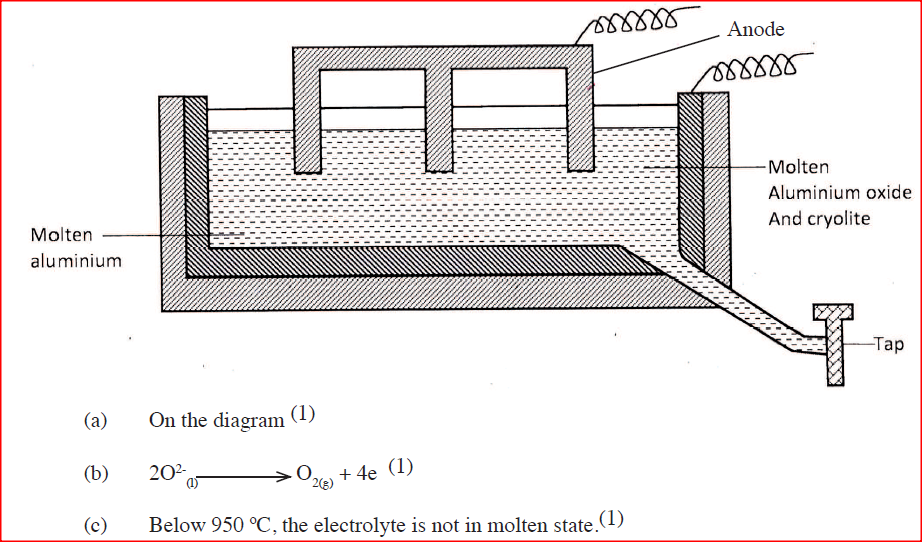
The diagram below represents a set up of an electrolytic cell that can be used in the production of aluminium
(a) One the diagram, label the anode
(b) Write the equation for the reaction at the anode (c) Give a reason why the electrolytic process is not carried out below 950°C (d) Give a reason why the production of aluminium is not carried out using reduction process (e) Give two reasons why only the aluminium ions are discharged (f) State two properties of duralumin that makes it suitable for use in aircraft industry (g)Name two environmental effects caused by extraction of aluminium
(a) Other than temperature, state two factors that determine the rate of a chemical reaction.
(b) A solution of hydrogen peroxide was allowed to decompose and the oxygen gas given off collected. After 5 minute, substance G was added to the solution of hydrogen peroxide. The total volume of oxygen evolved was plotted against time as shown in the graph below
(i)Describe the procedure of determining the rate of the reaction at minute 12.
(ii)How does the production of oxygen in region AB compare with that in region BC? Explain (iii)Write an equation to show the decomposition of hydrogen peroxide. (c) Sulphur (IV) oxide react with oxygen to form Suplhur (VI) oxide as shown in the equation below
(ii)Name one catalyst used for the reaction.
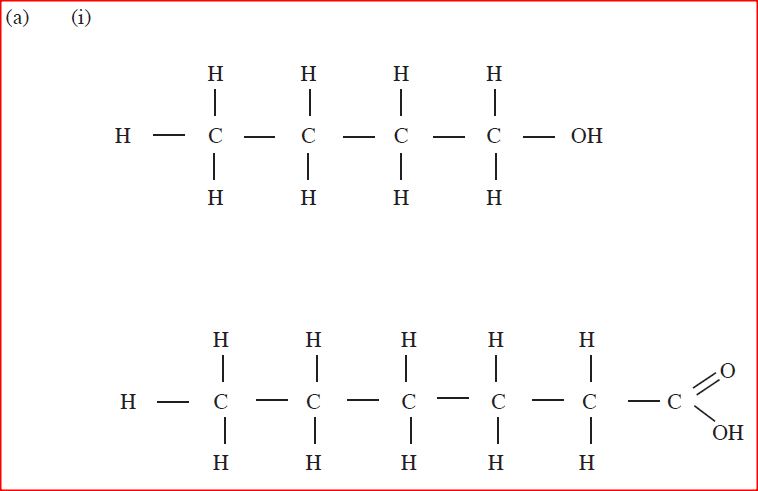
(a) Draw the structures of the following.
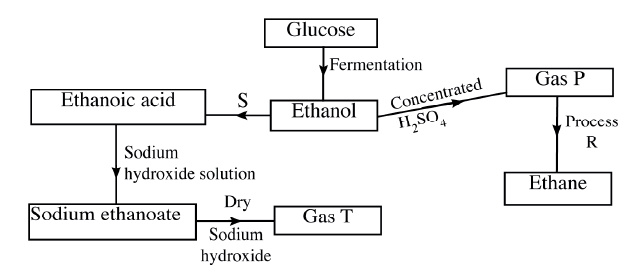
(i)Butan -1-ol (ii)Hexanoic acid. (b) Study the flow chart below and answer the questions that follow
(i) State the conditions necessary for fermentation of glucose to take place
(ii) State one reagent that can be used to carry out process S. (iii) Identify gases P: T: (iv) How is sodium hydroxide kept dry during the reaction (v) Give one commercial use of process R. (c) When one mole of ethanol is completely burnt in air, 1370kJ of heat energy is released. Given that 1 lire of ethanol is 780 g , calculate the amount of heat energy released when 1 litre of ethanol is completely burnt (C = 12.0; H=1.0; 0=16.0) (d) State two uses of ethanol other than as an alcoholic drink. |
Chemistry Topics
All
Archives
December 2024
|
We Would Love to Have You Visit Soon! |
Hours24 HR Service
|
Telephone0728 450425
|
|
8-4-4 materialsLevels
Subjects
|
cbc materialsE.C.D.E
Lower Primary
Upper Primary
Lower Secondary
Upper Secondary
|
teacher support
Other Blogs
|














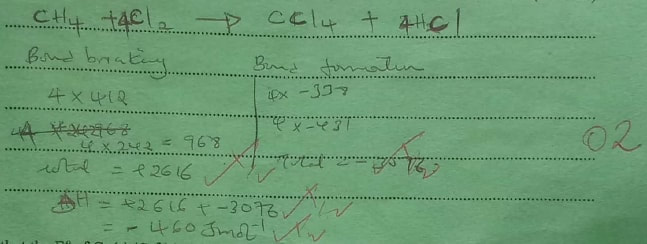


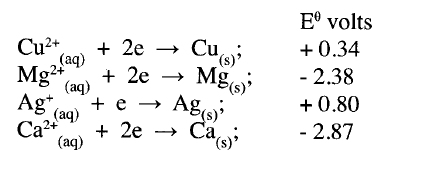
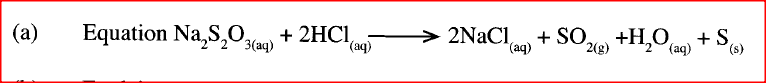






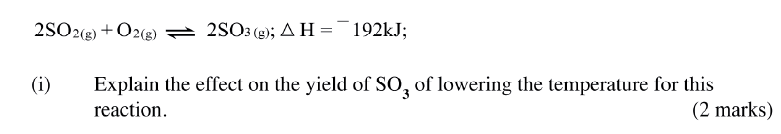
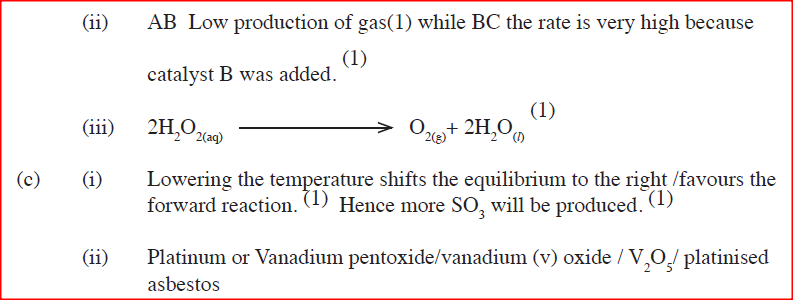


 RSS Feed
RSS Feed