|
Name the process which takes place when:
a) Solid carbon (IV) oxide (dry ice) changes directly into gas b) A red litmus paper turns white when dropped into chlorine water. c) Propane gas molecules are converted into a giant molecule
ANSWERS
a)sublimation
b)Bleaching. c)Polymerization
0 Comments
ANSWERS
When 94.5g of hydrated barium hydroxide, ba(OH)2. nH2O were heated to constant mass, 51.3g of anhydrous barium hydroxide were obtained. Determine the empirical formula of the hydrated barium hydroxide.
A group of compounds called chlorofluorocarbons have a wide range of uses but they also have harmful effects on the environment.
State one: a) Use of chlorofluorocarbons b) Harmful effect of chlorofluorocarbons on the environments.
ANSWERS
a)Refrigeration
b) - They deplete the ozone layer. - They cause green house effect.
In an experiment to study the properties of concentrated sulphuric acid, a mixture of the acid and wood charcoal was heated in a boiling tube.
(a) write the equation of the reaction that took place in the boiling tube. (b) Using oxidation numbers, show that reduction and oxidation reactions took place in the boiling tube
The atomic numbers of elements C and D are 19 and 9 respectively. State and explain the electrical conductivity of the compound CD in:
(a) Solid state (b) aqueous state.
ANSWERS
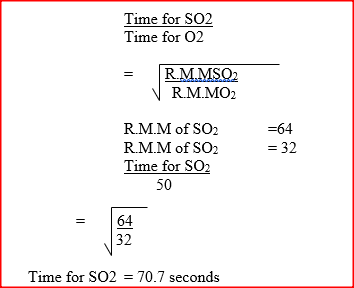
60cm3 of oxygen gas diffused through a porous partition in 50 seconds. How long would it take 60cm3 of sulphur (IV) oxide gas to diffuse through the same partition under the same conditions? (S= 32.0, 0 = 16.0)
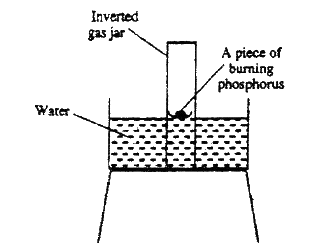
The diagram below represent a set-up that was used to show that part of air is used during burning.
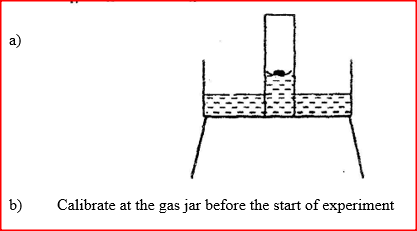
a) Given that phosphorus used was was in excess, draw a diagram of the set-up at the end of the experiment (when there was no further observable change).
b) Suggest one modification that should be made on the apparatus if the percentage of the air used is to be determined.
(a) What is meant by isomerism?
(b) Draw and name two isomers of butane. |
Chemistry Topics
All
Archives
December 2024
|
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |






 RSS Feed
RSS Feed

