KCSE CHEMISTRY QUESTIONS AND ANSWERS PER TOPIC
|
a) Write an equation for the reaction
b) Using the bond energies given below, calculate the enthalpy change for the reaction in (a) above
0 Comments
The table below gives the solubility’s of substances T and U at 10°C and 40°C.
When an aqueous mixture containing 55g of T and 12g of U at 80°c was cooled to 10°c, crystals formed.
a) Identify the crystals formed b) Determined the mass of the crystals formed c) Name the method used to obtain the crystals
Aqueous hydrogen chloride reacts with potassium manganate (VII) to produce chlorine gas, while a solution of hydrogen chloride in methylbenzene has no effect
on potassium manganate (VII). Explain this observation
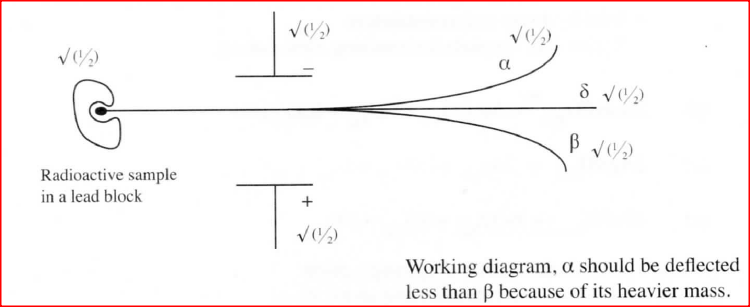
Draw a labeled diagram to illustrate how alpha, beta and gamma radiations can be distinguished from each other.
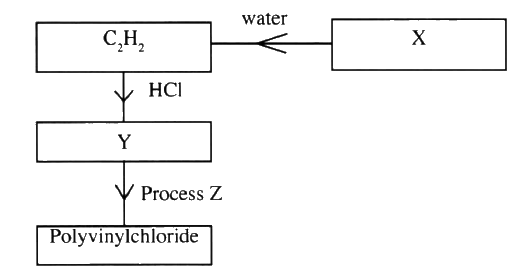
Study the flowchart below and answer the questions that follow:
(a) Identify:
(i) X (ii) Y (b) State two uses of polyvinylchloride
(a) A student electroplated a spoon with copper metal .Write an equation for the process that took place at the cathode.
(b) Calculate the time in minutes required to deposit 1.184g of copper if a current of 2 amperes was used. (1 Faraday = 96500 coulombs, Cu=63.5).
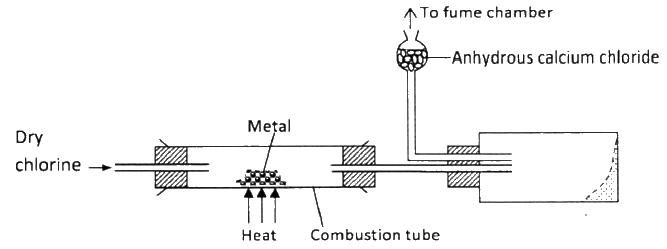
The diagram below illustrates a method of preparing salts by direct synthesis
(a) This method can be used to prepare either aluminum chloride or iron (III) chloride.
Explain why it cannot be used to prepare sodium chloride. (b) Describe how a sample of sodium chloride can be prepared in the laboratory by direct synthesis.
ANSWERS
(a) It does not sublime.
(b) Cut a piece of Sodium metal, place it on a deflagrating spoon. heat it briefly then lower it into a gas jar of chlorine. It will continue burning forming Sodium Chloride.
In terms of structure and bonding, explain the following observations:
(a) The melting point of aluminum is higher than that of sodium (b) Melting point of chlorine is lower than that of sulphur
ANSWERS
(a) Aluminium has a stronger metallic bond because it has more delocalised electronsthan sodium.
(b) Sulphur has a ringed structure of S8 molecules whiles chlorine is diatomic. The forces in sulphur are stronger than chlorine.
Describe an experiment procedure that can be used to extract oil from nut seeds
ANSWERS
Write equations to show the effect of heat on each of the following:
(a) Sodium hydrogen carbonate (b) Silver nitrate (c) Anhydrous iron (II) sulphate
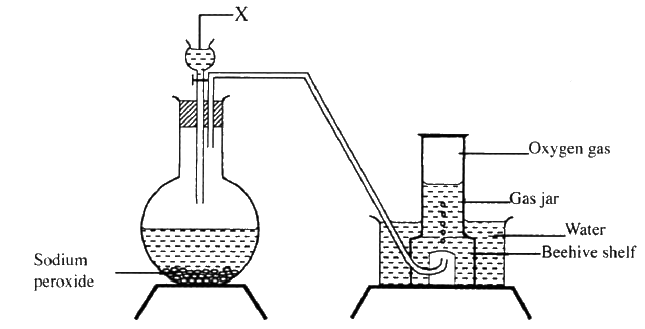
The set up below can be used to prepare oxygen gas. Study it and answer the questions that follow.
(a) Identify X
(b) What property of oxygen makes it possible for it to be collected as shown in the above set up? (c) State two uses of oxygen |
Chemistry Topics
All
Archives
December 2024
|
We Would Love to Have You Visit Soon! |
Hours24 HR Service
|
Telephone0728 450425
|
|
8-4-4 materialsLevels
Subjects
|
cbc materialsE.C.D.E
Lower Primary
Upper Primary
Lower Secondary
Upper Secondary
|
teacher support
Other Blogs
|
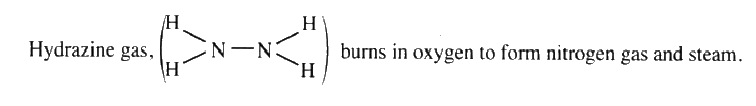

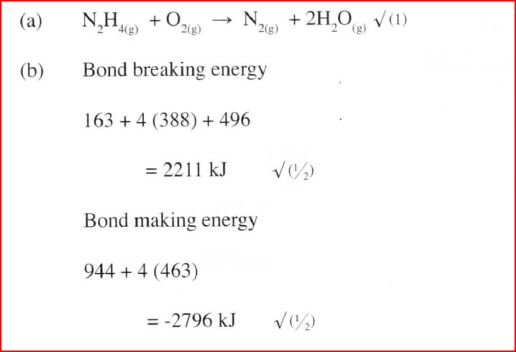








 RSS Feed
RSS Feed