|
(a) When excess calcium metal was added to 50 cm3 of 2 M aqueous copper (II) nitrate in a beaker, a brown solid and bubbles of gas were observed.
(i) "Write two equations for the reactions which occurred in the beaker. (ii) Explain why it is not advisable to use sodium metal for this reaction.
(b) Calculate the mass of calcium metal which reacted with copper (II) nitrate solution. (Relative atomic mass of Ca = 40)
(c) The resulting mixture in (a) above was filtered and sodium hydroxide added to the filtrate dropwise until in excess. What observations were made? (d) (i) Starting with calcium oxide, describe how a solid sample of calcium carbonate can be prepared. (ii) Name one use of calcium carbonate
ANSWERS
(c)A white precipitate is formed which is insoluble in excess.
(d) (I) Add dilute nitric (V) acid to calcium oxide to form the soluble salt calcium nitrate. Add sodium carbonate (another soluble salt) to form insoluble. Calcium Carbonate and sodium nitrate . Filter out the calcium carbonate, wash it with distilled water to remove traces of sodium nitrate and dry between filter papers (ii) Manufacture of cement Manufacture of sodium carbonate.
0 Comments
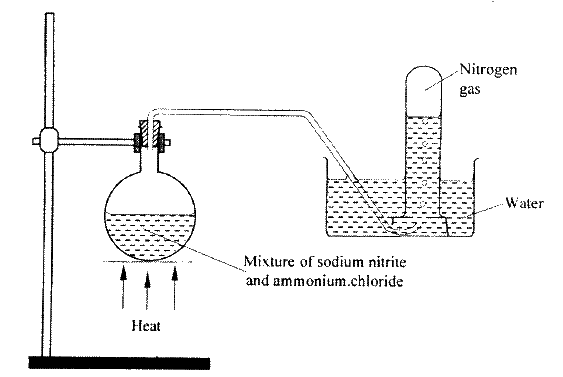
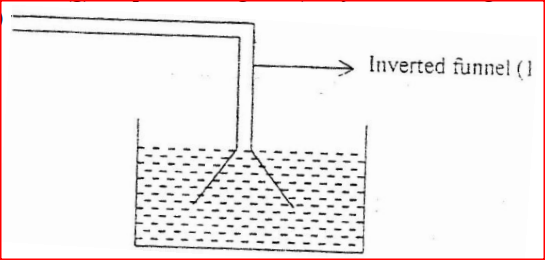
The set up below shows how nitrogen gas is prepared in a laboratory.
(a) Describe how nitrogen gas us formed in the flask.
(b). Nitrogen is inert, state one use of the gas based on this property.
ANSWERS
(a) Oxygen is used up
(b) Lighting of bulbs
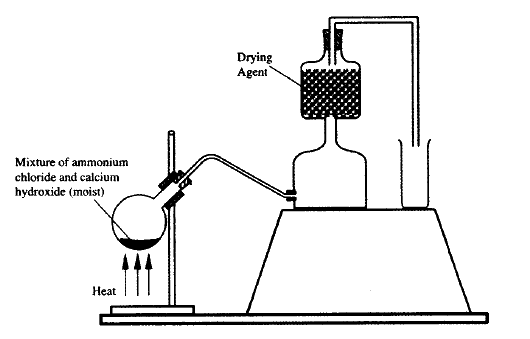
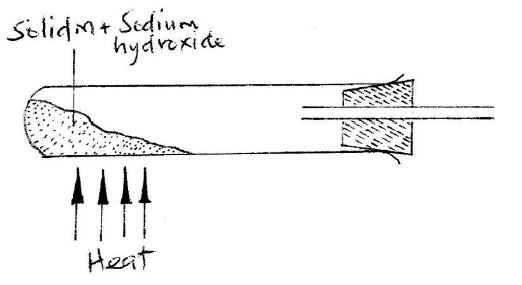
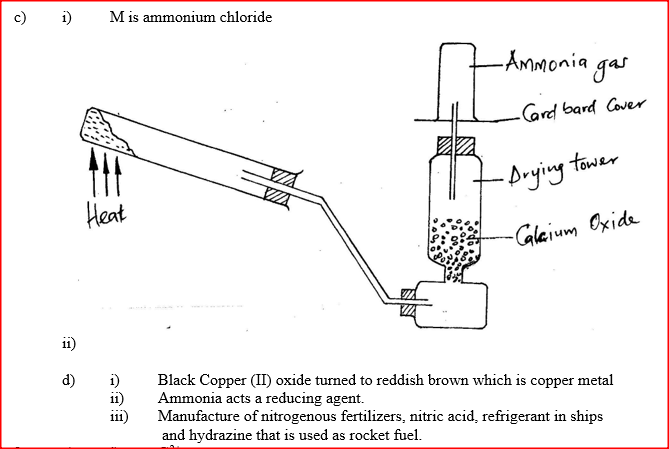
a) A student set up the apparatus as shown in the diagram below to prepare and collect dry ammonia gas.
i) Identify two mistakes in the set up and give a reason for each mistake.
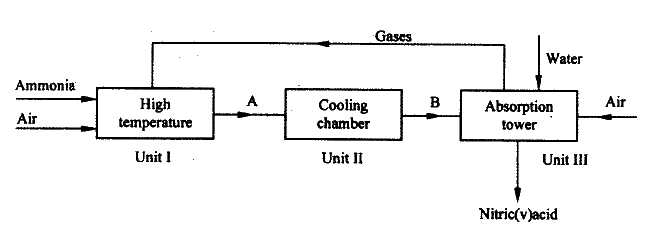
I. Mistake Reason II. Mistake Reason ii) Name a suitable drying agent for ammonia iii) Write an equation for the reaction that occurred when a mixture of ammonium chloride and calcium hydrogen was heated. iv) Describe one chemical test for ammonia gas d) Ammonia gas is used to manufacture nitric (V) acid, as shown below.
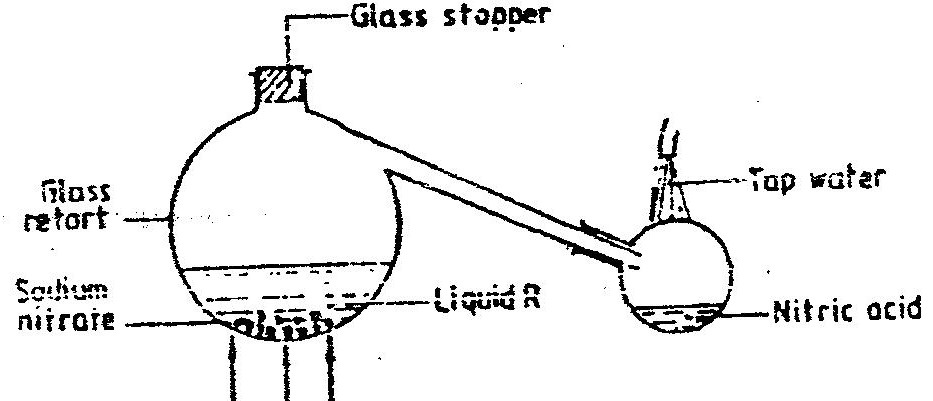
i) This process require the use of a catalyst. In which unit is the catalyst used
ii) Identify compound A and B A B iii) Using oxidation number, explain why the conversion of ammonia to nitric(V) acid is called catalytic oxidation of ammonia iv) Ammonia and nitric(V) acid are used in the manufacture of ammonium nitrate fertilizer. Calculate the amount of nitric (V) acid required to manufacture 1000kg ammonium nitrate using excess ammonia.(N=14.0, H=1.0, O=16.0)
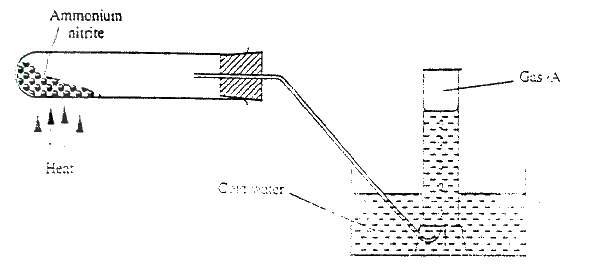
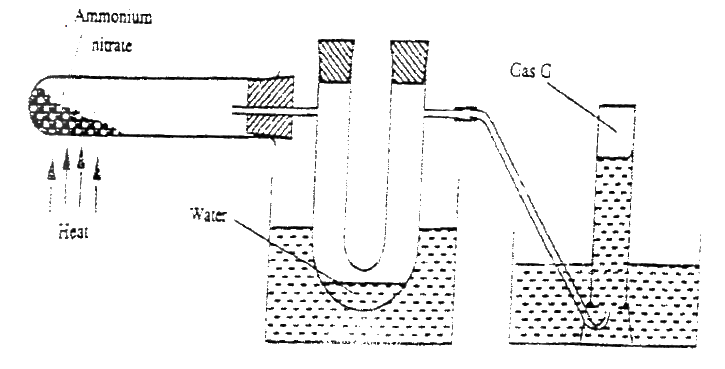
In an experiment to prepare nitrogen (I) oxide, ammonium nitrate was gently heated in a flask.
a) Write the equation for the reaction that took place in the flask. b) State and explain how the gas was collected. c) A sample of the gas was tested with damp blue and red litmus papers. What observations were made?
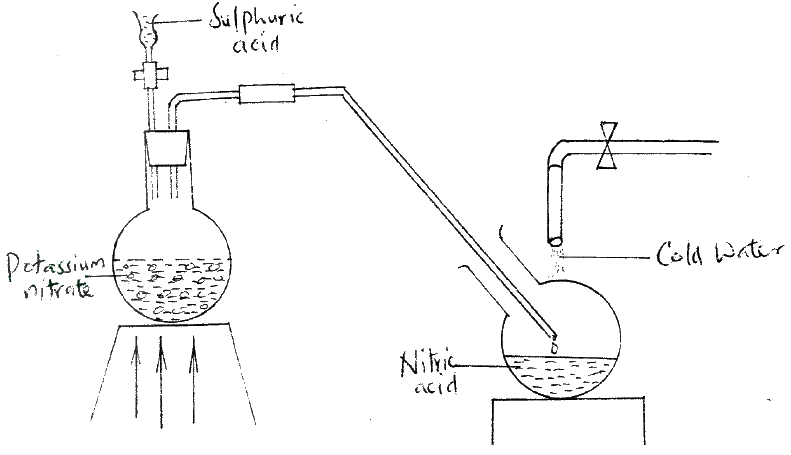
(a) A few drops of freshly prepared iron (II) sulphate solution were added to potassium nitrate solution in a test – tube. Concentrated sulphuric acid was then carefully added to the mixture. State the observation that were made
(b) Write an equation for the reaction that occurs when solid potassium nitrate is strongly heated
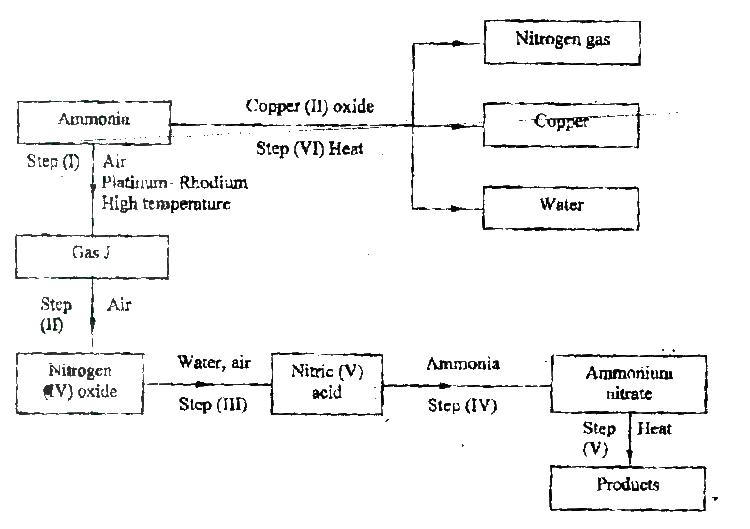
The flow chart below shows the industrial preparation of ammonia and the process used in the manufacture of some ammonium compounds. Study and answer the questions that follow
(a) Give the name of the
(c) Explain why it necessary to compress nitrogen and hydrogen in this process (d) Write an equation for the reaction which takes place in step 6 (e) Name the catalyst and the reagents used in step 3 Catalyst Reagent (f) Name compound Z1 (g) Give one commercial use of compound Z2
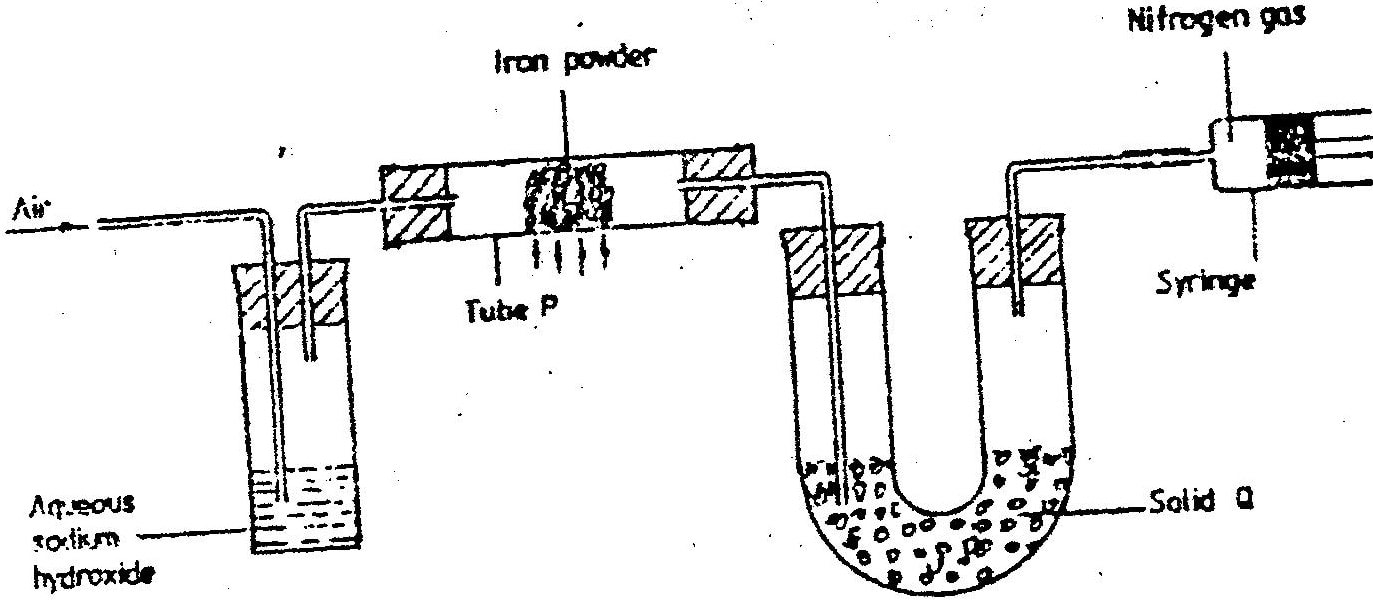
(a) Describe the process by which Nitrogen is obtained from air on a large scale.
(b) Study the flow chart below and answer the questions that follow.
(i) Identify gas J.
(ii) Using oxidation numbers, show that ammonia is the reducing agent in step (VI) (iii) Write the equation for the reaction that occurs in step (V). (iv) Give one use of ammonium nitrate. c) The table below shows the observations made when aqueous ammonia was added to cations of elements F2F and G until in excess.
(i) Select the cation that is likely to be Zn2+
(ii) Given that the formula of the cation of element E is E 2+, write the ionic equation for the reaction between E2+ (aq) and aqueous ammonia.
When magnesium was burnt in air, a solid mixture was formed. On addition of water to the mixture a gas which turned moist red litmus paper blue was evolved. Explain these observations.
(a) The diagram below represents a set up that was used to obtain dry nitrogen from air. Study it and answer the questions that follow
Related Chemistry Questions and Answers on nitrogen and its compounds form 3 level
In an experiment, a few drops of concentrated nitric acid were added to aqueous iron(II) sulphate in a test – tube. Excess sodium hydroxide solution was then added to the mixture.
a) State the observations that were made when: i) Concentrated nitric acid was added to aqueous iron (II) sulphate ii) Excess sodium hydroxide was added to the mixture. b) Write and ionic equation for the reaction which occurred in (a) (ii) above
The first step in the industrial manufacture of nitric acid is the catalytic oxidation of ammonia gas.
(a) What is the name of the catalyst used? (b) Write the equation for the catalytic oxidation of ammonia gas. (c) Nitric acid is used to make ammonium nitrate, state two uses of ammonium nitrate.
When dilute nitric acid was added to a sample of solid C, a colourless gas that formed a white a white precipitate with lime water was produced. When another sample of solid C was heated strongly in a dry test – tube, there was no observable change.
(a) Name one raw material which sodium hydroxide is manufactured
(b) Sodium hydroxide pellets were accidentally mixed with sodium chloride 17.6 g of the mixture were dissolved in water to make one litre of solution. 100 cm3 of the mixture were dissolved in water to make one litre solution. 100cm3 of the solution was neutralized by 40cm3 of 0.M sulphuric acid (i) Write an equation for the reaction that took place (ii) Calculate the: (i) Number of moles of the substance that reacted with sulphuric acid (ii) Number of moles of the substances that would react with sulphuric acid in the one litre of solution (iii) Mass of the unreacted substances in one litre of solution (H = 1,0 ; Na = 23.0 ; Cl= 35.5 ; 0= 16.0) (c) The diagram below shows an incomplete set-up used to prepare and collect ammonia gas
(i) Name solid M
(ii) Complete the diagram to show how a dry sample of ammonia gas can be collected (d) In an experiment, excess ammonia gas passed over heated copper (II) oxide on a combustion tube. (i) State the observation that was made in the combustion tube at the end of the experiment (ii) What property of ammonia is shown in the above reaction (iii) Name one use of ammonia
When a few drops of aqueous ammonia were added to copper (II) nitrate solution, a light tube precipitate was formed. On addition of more aqueous ammonia, a deep blue solution was formed.
Identify the substance responsible for the: a. Light blue precipitate b. Deep blue solution
ANSWERS
ANSWERS
State one use of sodium hydrogen carbonate.
ANSWERS
The flow chart below shows some reactions starting with lead (II) nitrate. Study it and answer the questions that follow.
(i) State the condition necessary in step 1.
(ii) Identify: I Reagent K II Gas Q III Acidic products S and R (iii) Write: I The formula of the complex ion formed instep 3. II The equation of the reaction in step 4 b) The use of materials made of lead in roofing and in water pipes is being discouraged State: (i) Two reasons why these materials have been used in the past. (ii) One reason why their use is being discouraged c) (i) The reaction between lead (II) nitrate and concentrated sulphuric acid starts but stops immediately. Explain
Explain why a high temperature is required for nitrogen of react with oxygen
answer
Nitrogen forms many compounds in which its oxidation state varies.
a) What is meant by oxidation state? b) What is the oxidation state of nitrogen in Mg3N2
ANSWERS
answers
answers
answers
answer
Nitrogen (II) oxide and nitrogen (IV) oxide are some of the gases released from car exhaust pipes. State how these gases affect the environment
answer
|
Chemistry Topics
All
Archives
December 2024
|
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |



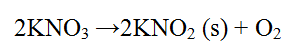
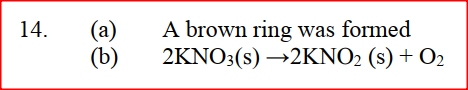













 RSS Feed
RSS Feed

