|
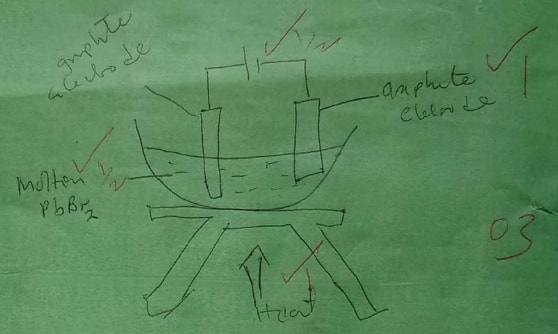
Draw in the space provided a labelled diagram of the set-up of the apparatus that can be used to electrolyse molten Lead (II) bromide. (3 marks)
Related Chemistry Questions and Answers on Effects of Electric Current on Substances Form 2 Level
0 Comments
Aluminium is both malleable and ductile.
(a)What is meant by? (i) Malleable: (ii)Ductile (b)State One use of aluminium based on: (i)malleability (ii)ductility
ANSWERS
(a) (i) Can be hammered into sheets.
(ii)Can be drawn into wires. (b)(i) Making of sufurias/ motor vehicle parts/ aeroplane parts,window / door flames, cups, plates, packaging materials, pans, making sheets/ roof. (ii)electricity cables/ wires.
Starting with sodium metal, describe how a sample of crystals of sodium hydrogen carbonate may be prepared.
ANSWERS
React sodium with water to get sodium hydroxide. Bubble into this solution excess carbon (IV) oxide to get sodium hydrogen carbonate
Starting with copper turnings. describe how a sample of copper(II) sulphate crystals can be prepared in the laboratory. (3 marks)
A student investigated the effect of an electric current by passing it through some substances. The student used inert electrodes, and connected a bulb to the circuit. The table below shows the substances used and their
states.
(a) In which experiment did the bulb not light?
(b) Explain your answer in (a) above.
ANSWERS
(a) 1 and 3
(b)In 1 ions K2CO3 are held rigidly within the crystal cannot move (no mobile ions) In 3 sugar exist as molecule hence no mobile ions.
(b) When magnesium burns in air, it forms a white solid and a grey-green solid.
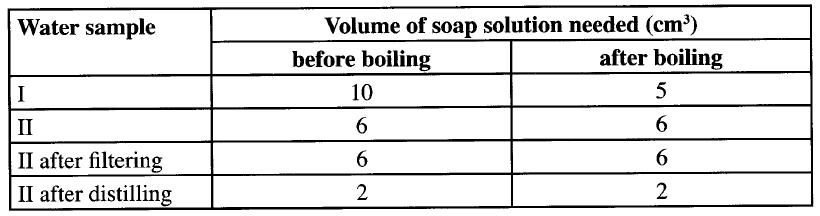
When a few drops of water are added to the mixture, a gas that turns red litmus paper blue is evolved. Identify the (i) white solid. (ii) gas evolved and state its use. (I) Name of gas (II) Use of the gas. ; (c) Two different samples of water (I and II) were tested with soap solution. Sample II was further subjected to two other processes before adding soap. 20 cm3 of each sample of water was shaken with soap solution in a boiling tube until a permanent lather was obtained. The results are shown in the table below
(i) Identify the water sample that had temporary hardness. Explain your answer.
(ii) Explain why the results for sample II are different after distilling but remain unchanged after filtering. (iii) State two disadvantages of using both water samples for domestic purposes.
(a) Name the method that can be used to obtain pure iron (III) chloride from a mixture of iron (III) chloride and sodium chloride.
(b) A student was provided with a mixture of sunflower flour, common salt and a red dye. The characteristics of the three substances in the mixture are given in the table below.
The student was provided with ethanol and any other materials needed.
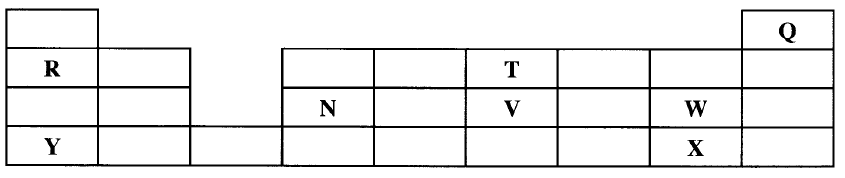
Described how the student can separate the mixture into its three components c) The diagram below show part of a periodic table. The letters do no represent the actual symbols of elements. Use the diagram to answer the questions that follow.
i) Explain why the oxidizing power of W is more than that of X
ii) How do the melting points of R and T compare? Explain iii) Sketch an element that could be used i) In weather ballons ii) For making a cooking pot d i) Classify the substances water, iodine, diamond and candle wax into elements and compounds
ii) Give one use of diamond
ANSWERS
(a)Sublimation
(b)Add ethanol to the mixture . Filter and evaporate filtrate to obtain red dye . Add water to the residue . Filter to obtain sunflower flour . Evaporate filtrate to obtain salt . OR Add H,O to mixture , filter , residue is sunflower , evaporate the water ; add ethanol to the residue filter . The filtrate is red dye. (3 marks) (c)(i) W accepts electrons more readily than X. W has small atomic radius/ W has less energy levels than X/ W has less screening effect than X/ W has greater effective nuclear attraction than X. W is more electro negative than X. (ii) T has a lower melting point than R because it exists in simple molecular form with weak Van der Waals forces while R has strong metallic bonds.
(a) (i) Carbon (IV) oxide is present in soft drinks. State two roles of carbon
(IV) oxide in soft drinks. (ii) Explain the observation made when a bottle containing a soft drink is opened. (iii) Carbon (IV) oxide dissolves slightly in water to give an acidic solution. Give the formula of the acid. (b) Zinc oxide can be obtained by heating zinc nitrate. A student heated 5.76 g of zinc nitrate. (i) Write an equation for the reaction that occurred. (ii) Calculate the total volume of gases produced. (Molar gas volume is 24 dm3; Zn = 65.4; O = 16.0; N = 14.0). (iii) Identify the element that is reduced when zinc nitrate is heated. Give a reason.
An electric current was passed through several substances and the results obtained recorded in the table below.
Which of these substance is likely to be
a)Magnesium b) Hexane c) lead (II) bromide ?
ANSWERS
(a) Describe how carbon (IV) oxide can be distinguished from Carbon II Oxide using calcium hydroxide solution.
(b) What is the role of carbon (IV) oxide in fire extinguishing?
ANSWERS
(a) Bubble the gases in calcium hydroxide solution. Carbon (II) Oxide does not react while Carbon (IV) Oxide forms a white precipitate.
(b) Carbon (IV) Oxide cuts the supply of oxygen
Starting with barium nitrate solution, describe how a pure sample of barium carbonate can be prepared in the laboratory.
ANSWERS
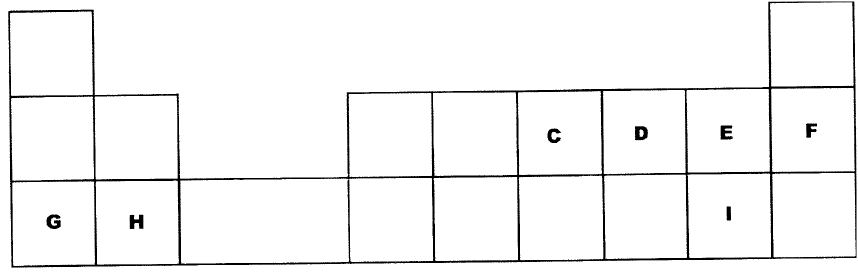
The table below is part of the periodic table. The letters are not the actual symbols of the elements. Study it and answer the questions that follows.
a) Select an element which is stored in paraffin in the laboratory
(b) How do the ionic radii of E and I compare? Explain
ANSWERS
(a) Element stored under paraffin G
(b) E is smaller than I. E has two energy levels while I has 3 energy levels.
Describe how samples of lead (II) sulphate, ammonium chloride and sodium chloride can be obtained from a mixture of the three.
ANSWERS
During laboratory preparation of carbon(IV) oxide gas, dilute hydrochloric acid was added to substance L in a conical flask.
Study the information in Table 3 and use it to answer the questions that follow.
A crystal of iodine, heated gently in a test tube gave off a purple vapour.
(a) Write the formula of the substance responsible for the purple vapour. (b) What type of bond is broken when the iodine crystal is heated gently? (c) State one use of iodine.
ANSWERS
(a)Formula of Iodine I2
(b)Weak Van der Waals (c)Antiseptic
The table below shows behaviour of metals R, X, Y and Z. Study it and answer the questions that follow:
a) Arrange the metals in the order of reactivity starting with the most reactive
b) Name a metal which is likely to be i X ii Y
ANSWERS
(a) Reactivity series starting with the most reactive
X R Z Y (b) X could be potassium Y could be copper
The atomic number of an element, T is 15.
(a) Write the electronic configuration of the ion T (b) Write the formula of an oxide of T.
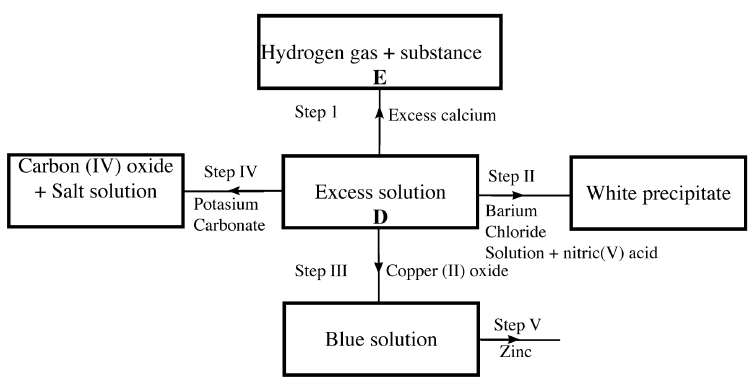
(a) The scheme below shows some of the reaction of solution D. Study it and answer the questions that follow
(i) Give a possible caution present in solution D
(ii) Write an ionic equation for the reaction in Step II (iii) What observations would be made in Step V? Give a reason (iv) Explain why the total volume of hydrogen gas produced in step 1 was found to be very low although calcium and solution D were in excess. (v) State one use of substance E. (b)Starting with solid sodium chloride, describe how a pure sample of lead (II) Chloride can be prepared in the laboratory (c) (i) State a property of anhydrous calcium chloride which makes it suitable for use as a drying agent for chlorine gas. (ii) Name another substance that can be used to dry chlorine gas
(a) The grid below represents part of the periodic table . Study it and answer the questions that follow. The letters are not the actual symbols of the elements
i) Select the most reactive metal. Explain
ii) Select an element that can form an ion with a charge of 3- iii) Select an alkaline earth metal iv) Which group 1 element has the highest first ionization energy? Explain v) Element A combines with chlorine to form a chloride of A. State the most likely pH value of a solution of a chloride of A. Explain (b) (i) Explain why molten calcium chloride and magnesium chloride conduct electricity while carbon tetrachloride and silicon tetrachloride do not. (ii) Under the same conditions , gaseous neon was found to diffuse faster than gaseous fluorine. Explain this observation. (F=19.0;Ne=20.0)
Table 1 shows the properties of two chlorides, D and E.
(a) State the type of bond present in:
a)What is meant by the term radical?
b) The table below contains atoms that form common radicals. Complete the table to show radicals formed from various atoms.
Given that the atomic number of Y is 13 and that of Z is 9:
a)Write the electronic arrangement of Y and Z; b) Draw the dot (.) and cross(x) diagram for the compound formed by Y and Z
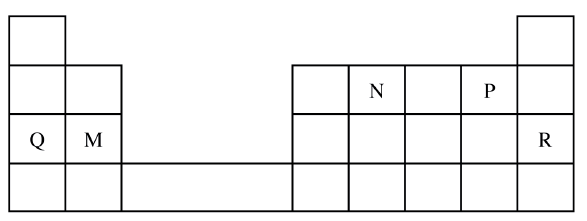
Use the part of the periodic table given below to answer the questions that follow (Letters are not the actual symbols of the elements)
a) Identify the element that forms giant covalent structures
b) Identify one element that does not form compounds c) Write the formula for the nitride of M
Explain how condition of electricity takes place in the following.
(a) Iron metal (b) Molten lead (II) iodide
ANSWERS
(a) delocalised electrons.
(b) Ions in the melt. |
Chemistry Topics
All
Archives
December 2024
|
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |
























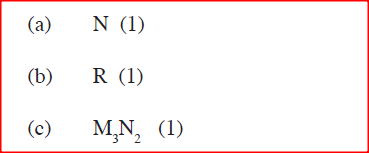

 RSS Feed
RSS Feed

