a) Identify the solids
|
| Molten M | Conduct electric current and is not decomposed |
| Molten N | Conduct electric current and a gas is formed at one of the electrodes |
Suggest the type of bonding present in;
a) Substance M (1mk)
Metallic bond
b) Substance N (1mk)
Ionic bond
Distinguish between empirical and molecular formula of a compound.
ANSWERS
- Empirical formula shows the simplest whole number ratio of the moles of atoms of different elements in a compound while the molecular formula shows the actual number of atoms of various elements present in one molecule of the compound.
(NH4)2HPO4 is a fertilizer used by farmers to boost their crop production.
(a) Calculate the mass of phosphorus in a 20 kg packet 14.0; H = 1.0; P = 31.0; 0 16.0)
(b) State one advantage of this fertilizer, (NH4)2HPO4 over urea CO(NH2)2
(a) Calculate the mass of phosphorus in a 20 kg packet 14.0; H = 1.0; P = 31.0; 0 16.0)
(b) State one advantage of this fertilizer, (NH4)2HPO4 over urea CO(NH2)2
Explain why commercial indicators are preferred to flower exacts as acid-base indicators.
ANSWERS
- The composition of commercial indicators remains constant. hence gives consistent results.
- The composition of flower extracts change with time giving inconsistent results.
Explain why a solution of sodium chloride Conducts electricity while that of sugar does not.
ANSWERS
- A solution of sodium chloride contains ions which are free to conduct electricity while that of sugar contains molecules hence cannot conduct electricity.
(a) Name two ores of iron.
(b) Describe how the amount of iron in a sample of iron(III) oxide can be determined.
(b) Describe how the amount of iron in a sample of iron(III) oxide can be determined.
ANSWERS
(a) Haematite;
Magnetite;
Siderite.
(b) Weigh the iron (III) oxide together with a crucible;
Heat the Iron(III) oxide and coke to a constant mass;
Cool and re-weigh residue and crucible
The difference in mass is weight of the iron.
Magnetite;
Siderite.
(b) Weigh the iron (III) oxide together with a crucible;
Heat the Iron(III) oxide and coke to a constant mass;
Cool and re-weigh residue and crucible
The difference in mass is weight of the iron.
Explain why it is important to put off a non-luminous flame immediately after use.
ANSWERS
- A non -luminous flame is invisible, hence it should be put off to avoid being accidentally burned.
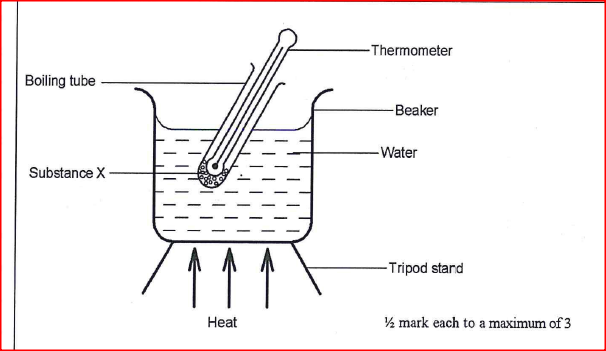
You are provided with the following: thermometer, boiling tube, beaker, Bunsen burner, pure substance X whose boiling point is about 80°C, water and any other apparatus that may be required. Draw a labelled diagram of the set-up that can be used to determine the melting point of X.
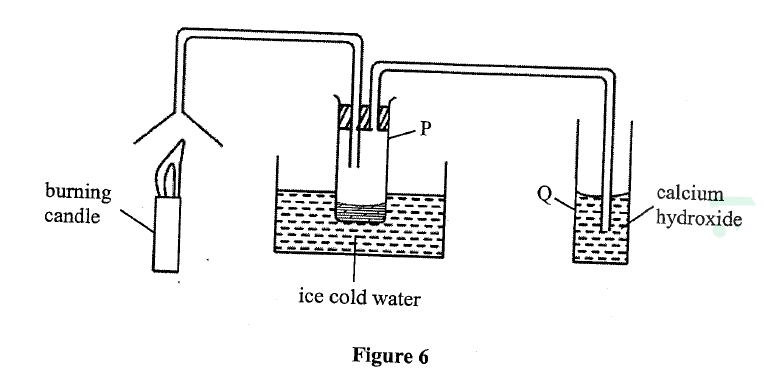
Study the set-up in Figure 6 and answer the questions that follow.
(a) Name the substance that was collected in tube p.
(b) Write an equation for the reaction which occurs in tube Q in the first few minutes of the experiment.
(c) Give a suitable Conclusion for the experiment in the set-up.
(b) Write an equation for the reaction which occurs in tube Q in the first few minutes of the experiment.
(c) Give a suitable Conclusion for the experiment in the set-up.
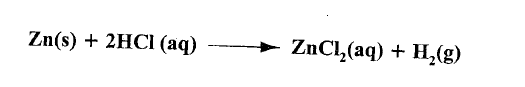
(a) Zinc reacts with hydrochloric acid according to the following equation.
Identify the reducing agent. Give a reason for the answer.
(b) Iron sheets are dipped in molten zinc to prevent rusting. Name this process.
(b) Iron sheets are dipped in molten zinc to prevent rusting. Name this process.
(a) Give the symbols of the o charged particles emitted by a radioactive isotope.
mass number
atomic number:
atomic number:
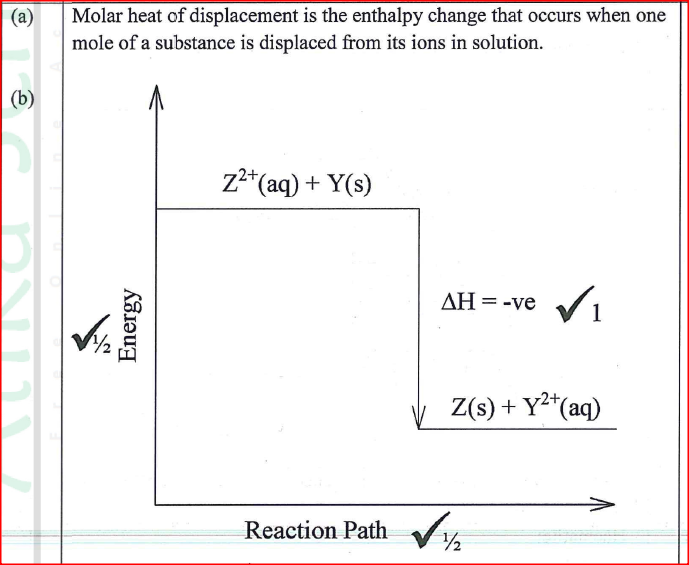
(a) Define molar heat of displacement.
(b)The following ionic equation represents the reaction between metal Y and an aqueous solution of Z2+.
(b)The following ionic equation represents the reaction between metal Y and an aqueous solution of Z2+.
Draw an energy level diagram to represent the reaction
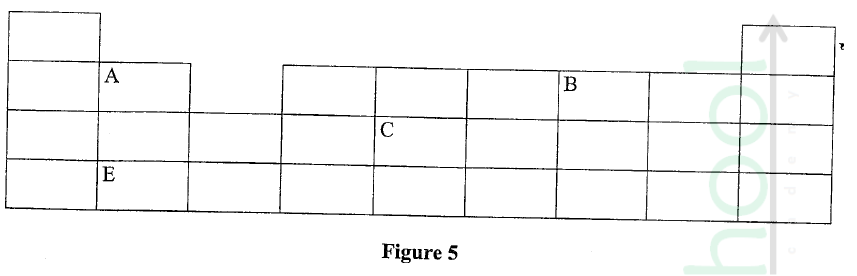
Figure 5 represents a grid that is part of the periodic table. Study it and answer the questions that follow. The letters are not the actual symbols of the elements.
(a) Write the electron arrangement of element C.
(b) On the grid provided, show with a tick (✓) the position of element D whose atomic number is 18.
(c) Element E is more reactive than A. Explain.
(b) On the grid provided, show with a tick (✓) the position of element D whose atomic number is 18.
(c) Element E is more reactive than A. Explain.
ANSWERS
(a) 2.8.4
(b) period 3, group 8
(c) E has a bigger atomic radius than A / the valence electrons of element E are further from the nucleus, hence loosely held by the positive nucleus and requires less energy to be removed during reaction.
OR
A has a smaller atomic radius than E / the valence electrons of element
A are closer to the nucleus, hence strongly held by the positive nucleus and requires more energy to be removed during a reaction.
(b) period 3, group 8
(c) E has a bigger atomic radius than A / the valence electrons of element E are further from the nucleus, hence loosely held by the positive nucleus and requires less energy to be removed during reaction.
OR
A has a smaller atomic radius than E / the valence electrons of element
A are closer to the nucleus, hence strongly held by the positive nucleus and requires more energy to be removed during a reaction.
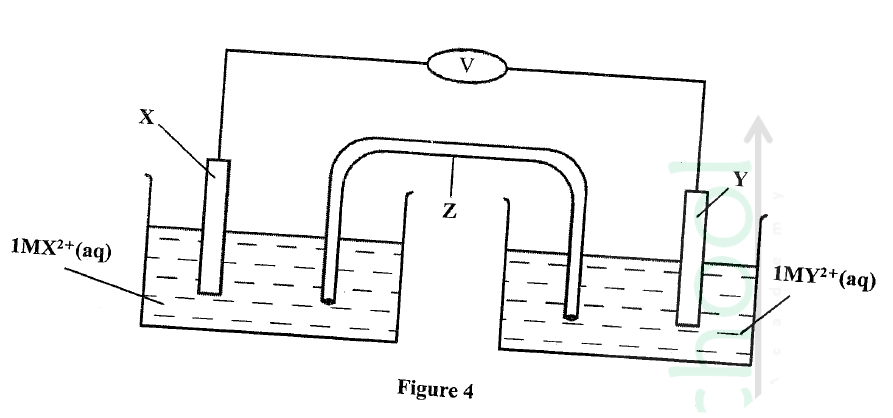
Metal X and Y have standard electrode Potentials of —0.13 V and —0.76V respectively .The metals were Connected to form a cell as shown in Figure 4.
(a) Name the part labelled z.
(b) State one functi0 of the part labelled Z.
(c) Calculate the e.m.f. of the cell.
(b) State one functi0 of the part labelled Z.
(c) Calculate the e.m.f. of the cell.
You are provided with solid potassium hydrogen carbonate. Describe how a solid sample of potassium nitrate can be prepared.
ANSWERS
Measure a certain volume of dilute nitric(V) acid and place it in a beaker;
Add potassium hydrogen carbonate little by little as the mixture is stirred until effervescence stops;
Evaporate the solution to saturation and allow to cool for crystals to form;
Dry the crystals in between filter papers.
Add potassium hydrogen carbonate little by little as the mixture is stirred until effervescence stops;
Evaporate the solution to saturation and allow to cool for crystals to form;
Dry the crystals in between filter papers.
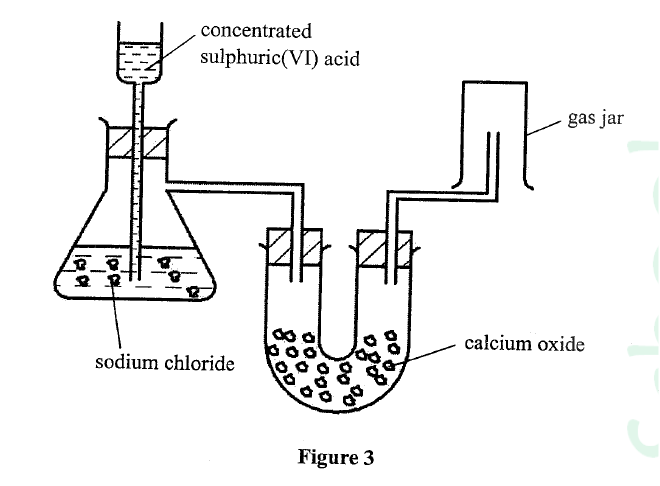
Figure 3 shows a set-up used by a student to prepare dry chlorine gas in the laboratory.
Identify three mistakes in the set-up, and give a reason for each.
ANSWERS
- One reagent is missing, hence reagents provided cannot produce chlorine;
- Wrong drying agent — Calcium oxide will react with the chlorine gas;
- Incorrect method of gas collection
- No gas will be collected / chlorine is denser than air.
In the Haber process, nitrogen reacts with hydrogen according to the following equation.
(a)What would be the effect of adding a catalyst on the position of the equilibrium?
(b) Explain why it is not advisable to use temperatures higher than 773 K in the Haber process.
(b) Explain why it is not advisable to use temperatures higher than 773 K in the Haber process.
ANSWERS
(a)No effect/does not affect the position of the equilibrium.
(b)Forward reaction is exothermic, excessive temperatures would favour the backward reaction therefore lowering the yield of ammonia.
(b)Forward reaction is exothermic, excessive temperatures would favour the backward reaction therefore lowering the yield of ammonia.
When ethene gas is compressed at a high temperature, a solid is formed.
(a) Give the name of the solid.
(b) Explain why it is not advisable to allow the solid to accumulate in the environment.
(a) Give the name of the solid.
(b) Explain why it is not advisable to allow the solid to accumulate in the environment.
ANSWERS
(a)Polythene / Polyethene
(b)It is non-biodegradable, hence pollutes the environment.
(b)It is non-biodegradable, hence pollutes the environment.
(a) Element U has atomic number 12 while element V has atomic number 16. How do the melting points of their oxides compare? Explain.
ANSWERS
- The oxide of U has higher melting point than the oxide of V;
- -This is because the oxide of U is a solid with ionic bonds hence requires a lot of energy to melt;
- -The oxide of V is a gas hence requires little energy to melt/break molecular forces of attraction.
Using iron filings, describe an experiment that can be conducted to show that oxygen is present in air.
ANSWERS
- Wet a measuring cylinder/gas jar and sprinkle some iron filings on the wet surface. Remove the excess iron filings;
- Invert the measuring cylinder in a trough of water;
- Take the reading of the air column in the measuring cylinder. Leave the set-up for 2 days;
- Read and record the volume of the air column;
- The volume of the air reduced and the grey filings changed to a brown substance;
- The brown substance is a compound of iron and oxygen;
- The oxygen in the air reacted with the iron fillings to form rust.
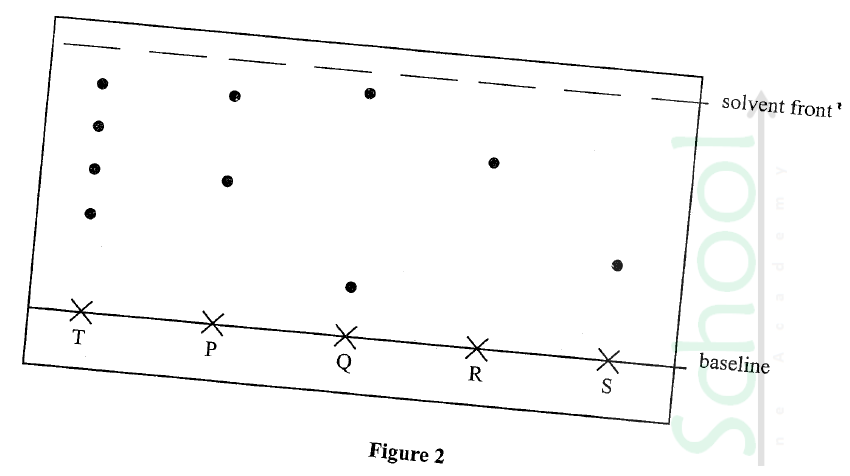
An experiment was Carried out to determine the presence of Substances P, Q, R and S in mixture T. The results obtained are shown in Figure 2.
(a) Ne the method of separation illustrated in Figure 2.
(b) Select:
(i) one Substance Which Contains a Component not present in T.
(ii) a substance which is least Soluble in the solvent used.
(b) Select:
(i) one Substance Which Contains a Component not present in T.
(ii) a substance which is least Soluble in the solvent used.
ANSWERS
(a)Chromatography/paper chromatography
(b)(i)Q
(ii)S
(b)(i)Q
(ii)S
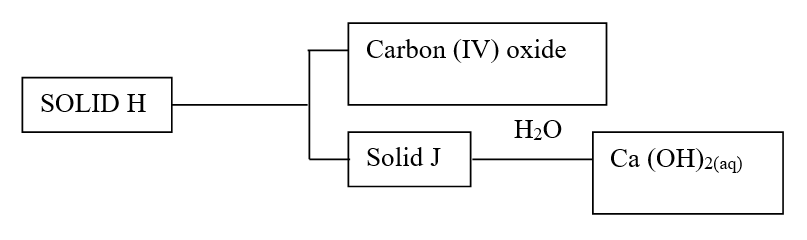
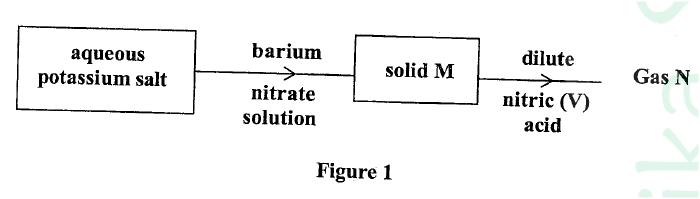
Study the flow chart in Figure 1 and answer the questions that follow.
Gas N forms a while suspension with aqueous calcium hydroxide.
(a) Name the anion present in the potassium salt.
(b) Write an ionic equation for the formation of solid M.
(c) Give one use of gas N.
(a) Name the anion present in the potassium salt.
(b) Write an ionic equation for the formation of solid M.
(c) Give one use of gas N.
30.0 cm3 of aqueous sodium hydroxide containing 8.0 g per litre of sodium hydroxide were completely neutralized by 0.294 g of a dibasic acid. Determine the relative formula mass of the dibasic acid. (Na = 23.0 ; 0 = 16.0; H 1.0)
(a) State Graham’s law of diffusion
(b) Explain why a balloon filled with helium gas deflates faster than a balloon of the same size filled with argon gas.
(b) Explain why a balloon filled with helium gas deflates faster than a balloon of the same size filled with argon gas.
ANSWERS
(a) Graham’s Law of diffusion
The rate of diffusion of a gas is inversely proportional to the square root of its density at constant temperature and pressure.
(b)Helium is less dense than argon hence it diffuses out of the balloon faster than argon.
The rate of diffusion of a gas is inversely proportional to the square root of its density at constant temperature and pressure.
(b)Helium is less dense than argon hence it diffuses out of the balloon faster than argon.
Chemistry Topics
All
Acetylene Gas
Acid Bases And Indicators
Acids Bases And Salts
Air And Combustion
Alkaline
Ammonia
Calcium Carbonate
Carbon And Its Compounds
Charles's Law
Chemical Families
Chemical Reactions
Chemistry Practical Assignments
Chlorine And Its Compounds
Cleansing Agent
DashBoard
Definition Of Chemistry Terms
Differences In Chemistry
Diffusion
Down's Cell
Electric Current On Substance
Electrochemistry I And II
Empirical Formula
Energy Changes In Chemical And Physical Processes
Ethanol
Ethene Gas
Form 1 Level
Form 2
Form 3
Form 4
Gas Laws
General Chemistry
Haber Process
Hard Water
Hydrochloric Acid
Introduction To Chemistry
Kcse-1995
Kcse-1996
Kcse-1997
Kcse-1998
Kcse 1999
Kcse-2000
Kcse-2001
Kcse-2002
Kcse-2003
Kcse-2004
Kcse2005
Kcse-2005
Kcse-2006
Kcse2007
Kcse-2007
Kcse-2008
Kcse-2009
Kcse-2010
Kcse-2011
Kcse-2012
Kcse-2013
Kcse-2014
KCSE 2015
KCSE 2016
KCSE 2017
KCSE 2018
Kcse 2019
Metals
Molecular Formula
Nitrogen And Its Compounds
Notes And Tutorials
Nuclear Reactions
Organic Chemistry I
Organic Chemistry II
Oxidation
Oxygen
Paper 1
Paper 2
Paraffin
Periodic Table
Phenolphthalein Indicator
Phosphorous
Potassium
Propane
Properties-and-trends-across-the-period
Radioactivity
Reaction Rates And Reversible Reactions
Salts
Simple Classification Of Substances
Sodium
Structure Of The Atom And The Periodic Table
Structures And Bonding
Sulphur And Its Compounds
Sulphuric Acid
The Mole
Water And Hydrogen
Zinc
Archives
December 2024
January 2024
December 2023
November 2023
October 2021
November 2020
October 2020
September 2020
July 2020
May 2020
August 2019
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |



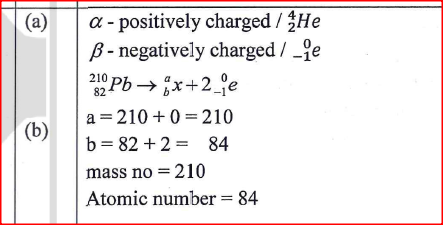
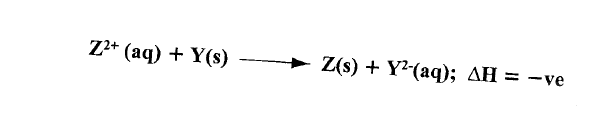

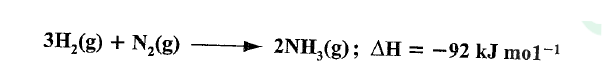



 RSS Feed
RSS Feed

