|
Describe an experiment to show that group one elements react with cold water to form alkaline Solutions
ANSWERS
0 Comments
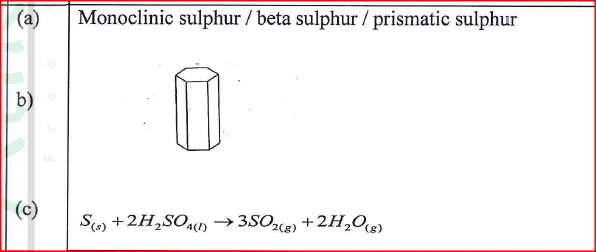
One of the allotrope s of sulphur is rhombic sulphur.
(a) Name the other allotrope of sulphur. (b) Draw a diagram to show the shape of the allotrope named in (a) above. (c) Write an equation for the reaction between concentrated sulphur(VI) acid and sulphur
The following are formulae of organic compounds. Use the formulae to answer the questions that follow:
(a) Select:
(i) two compounds which when reacted together produce a sweet smelling compound. (ii) an unsaturated hydrocarbon. (b) Name the compound selected in (a) (ii).
(a) Explain why it is not advisable to prepare a sample of carbon(IV) oxide using barium carbonate and dilute sulphuric(VI) acid.
(b) State a method that can be used to collect dry carbon(IV) oxide gas. Give a reason.
ANSWERS
(a)The reaction starts but soon stops.
This is because the insoluble barium sulphate produced forms a coating on the surface of the barium carbonate preventing further reaction and evolution of carbon(IV) oxide gas. (b) Downward delivery. Carbon(TV) oxide is denser than air.
(a) Define a soluble base.
(b) Aqueous solutions of 2M ethanoic acid and 2M nitric(V) acid were tested for electrical conductivity. Which solution is a better conductor of electricity? Explain.
ANSWERS
(a)A soluble base is a substance that dissociates in water to produce hydroxide ions as the only negative ions.
(b)Nitric(V) acid. This is because nitric(V) acid is a strong acid and dissociates completely in solution producing many H+ ions.
When an aqueous solution of compound X was mixed with a few drops of bromine water, the colour of the mixture remained yellow.
When another portion of solution X was reacted with acidified potassium dichromate(VI), the colour of the mixture changed from orange to green. (a) What conclusion can be made from the use of: (i) bromine water? (ii) acidified potassium dichromate(VI)? (b) Solution X was reacted with a piece of a metal and a colourless gas was produced. Describe a simple experiment to identify the gas.
(a) Name two ores in which sodium occurs.
(b) During extraction of sodium using the down's process, calcium chloride is added to the ore. Give a reason for the addition of calcium chloride. (c) State two uses of sodium.
(a) What is meant by the term bleaching?
(b) Write the formula of the bleaching nent formed when chlorine gas reacts with aqueous sodium hydroxide. (c) State the role of chlorine in water treatment.
In terms of structure and bonding, explain why graphite is used as a lubricant in machines.
ANSWERS
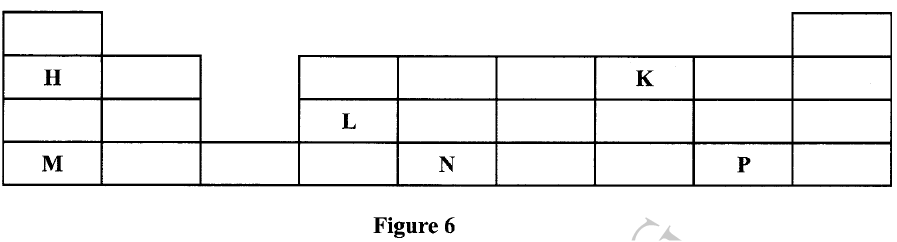
Figure 6 shows part of the periodic table. The letters are not the actual symbols of the elements. Stud it and answer the questions that follow.
(a) Write an equation for the reaction between M and K.
(b) Select the element which can form an ion with a charge of +3. (c) An element J has atomic number 15. Indicate with a tick (✓), on the part of the periodic table the position of J.
Explain how a student can establish whether a liquid sample extracted from a plant is pure.
ANSWERS
(a) What is an inert electrode?
(b) State the products formed when brine is electrolysed using inert electrodes. Anode: Cathode:
ANSWERS
(a)Inert electrode is one which does not participate in the reaction / does not affect the products of electrolysis / does not react;
(b)Anode - chlorine; Cathode - Hydrogen;
The atomic numbers of some elements P, Q, R and S are 6, 8, 12 and 17 respectively.
(a) Draw the dot (•) and cross (X) diagrams for the compounds formed when: (i) R and Q react (ii) P and S react. (b) Explain why the melting point of the compound formed by P and S is lower than that formed by R and Q.
Study the flow chart in Figure 5 and answer the questions that follow.
(a) Identify substances K and L. K:
(b) Name one reagent that can be used to carry out process J.
The following procedure was used to investigate the temperature changes that occur when sodium hydroxide solution is added to dilute hydrochloric acid.
(i) Place the acid in a glass beaker and record its temperature. (ii) Add a known volume of sodium hydroxide solution. (iii) Stir the mixture and record the highest temperature reached. (iv) Repeat steps (ii) and (iii) with different volumes of sodium hydroxide solution. (a) State two factors that must be kept constant in this experiment (b) Explain how the use of a polystyrene cup will affect the results.
ANSWERS
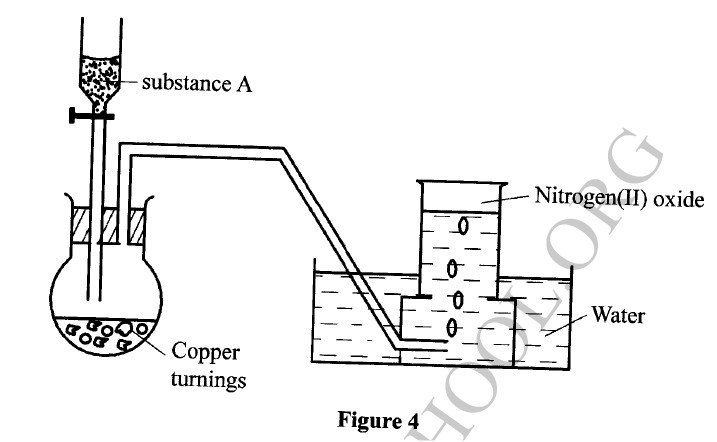
The set-up in Figure 4 can be used to prepare nitrogen (II) oxide. Use it to answer the questions that follow.
(a) Name substance A
(b) When the gas jar containing nitrogen (II) oxide is exposed to air, a brown color is observed. Explain. (c) Write an equation for the reaction which occurred in the flask.
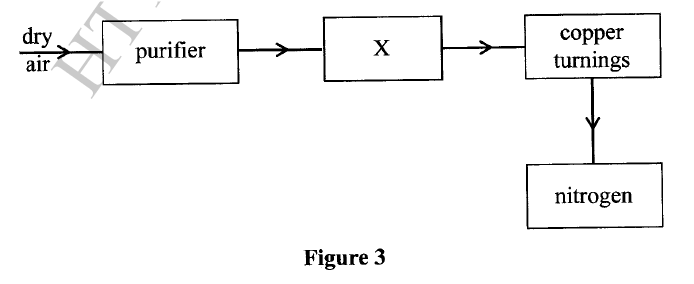
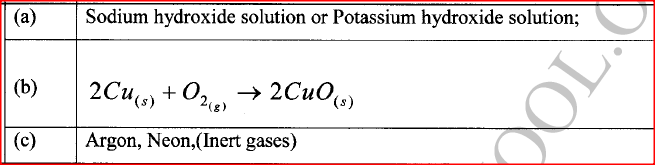
The flow chart in Figure 3 shows the process of obtaining a sample of nitrogen gas. Study it and answer the questions that follow.
(a) Identify X
(b) Write an equation for the reaction with heated copper turnings. (c) Name an impurity in the sample of nitrogen gas.
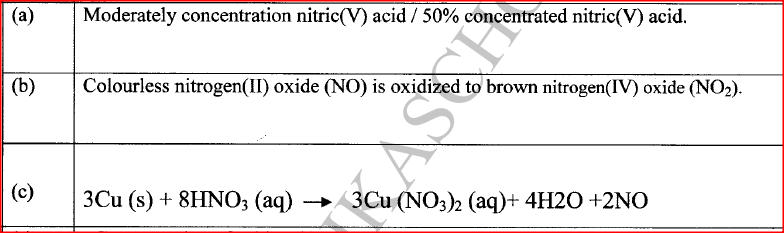
In an experiment, concentrated nitric(V) acid was reacted with iron(II) sulphate. State and explain the observations made.
ANSWERS
The mixture changed from green to yellow / formation of a brown gas;
Iron(II) ions is oxidized by nitric(V) acid to Iron(III) ions / nitric(V) acid is reduced to nitrogen(1I) oxide which is oxidized by oxygen to nitrogen(IV ) oxide.
Starting with copper, describe how a pure sample of copper(II) carbonate can be prepared.
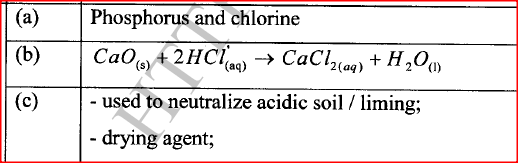
Using the elements chlorine, calcium and phosphorus:
(a) Select elements that will form an oxide whose aqueous solution has a pH less than 7. (b) Write an equation for the reaction between calcium oxide and dilute hydrochloric acid. (c) Give one use of calcium oxide.
Explain the observation made when chlorine gas is passed through a solution of potassium iodide.
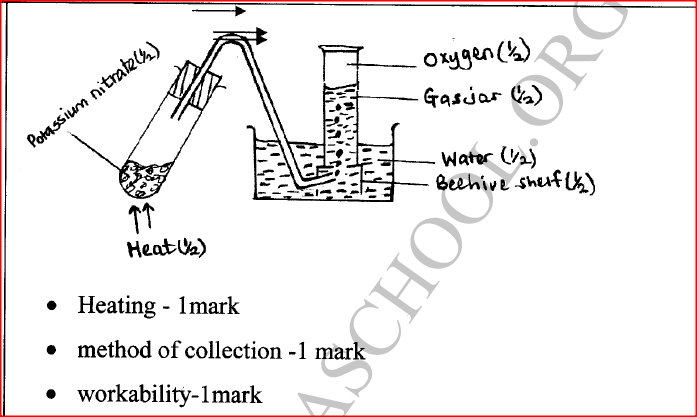
Potassium nitrate liberates oxygen gas when heated. Draw a diagram of a set-up that shows heating of potassium nitrate and collection of oxygen gas.
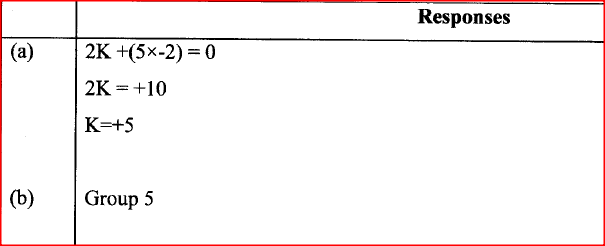
An oxide of element K has the formula K2O5.
(a) Determine the oxidation number of K. (b) To which group of the periodic table does K belong?
20 cm3 of ethanoic acid was diluted to 400 cm3 of solution. Calculate the concentration of the solution in moles per litre. (C = 12.0 ; H = 1.0 ; 0 =16.0) (Density of ethanoic acid = 1.05 g/cm3)
Copper(II) ions react with excess aqueous ammonia to form a complex ion.
(a) (i) Write an equation for the reaction that forms the complex ion. (ii) Name the complex ion. (b) Explain why CH4 is not acidic while HCl is acidic yet both compounds contain hydrogen. |
Chemistry Topics
All
Archives
December 2024
|
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |
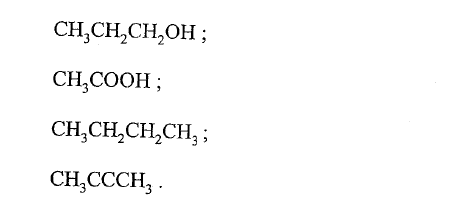













 RSS Feed
RSS Feed

