|
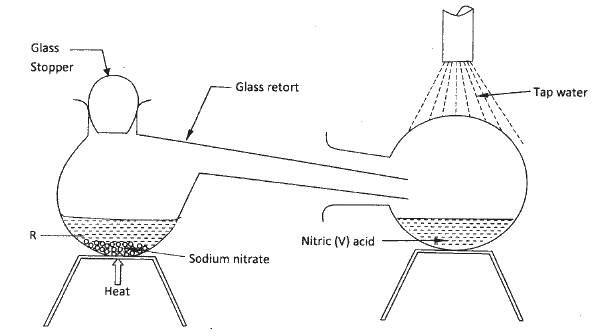
In the laboratory, small quantities of nitric (V) acid can be generated using the following set up. Study it and answer the questions that follow.
a) i) Give the name of substance R.
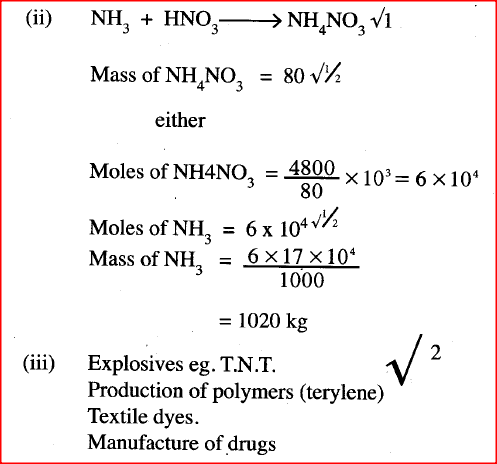
ii) Name one other substance that can be used in place of sodium nitrate. iii) What is the purpose of using tap water in the set up above? b) Explain the following; i) It is not advisable to use a stopper made of rubber in the set-up ii) the reaction between copper metal with 50% nitric (V) acid in an open test-tube produces brown fumes. c) i) Nitrogen is one of the reactants used in the production of ammonia, name two sources of the other reactant. ii) A factory uses nitric (V) acid and ammonia gas in the preparation of a fertilizer. If the daily production of the fertilizer is 4800kg; calculate the mass of ammonia gas used in kg. (N = 14.0; O = 16.0; H = 1.0) iii) State two other uses of nitric (V) acid other than the production of fertilizers.
ANSWERS
(a) (I) Concentrated /sulphuric (VI) acid.
(ii) Potassium nitrate (iii) To condense the fumes or vapour of nitric (V) acid into liquid (b) (i) Nitric acid (V) will corrode the rubber (ii) The reaction produces nitrogen monoxide (colourless) which is oxidised by oxygen from the air to form nitrogen(IV) oxide. (c)(i) Water Alkanes Biogas Water gas Related Chemistry Questions and Answers on Nitrogen and It's Compound Form 3 Level
0 Comments
Leave a Reply. |
Chemistry Topics
All
Archives
December 2024
|
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |

 RSS Feed
RSS Feed

