KCSE CHEMISTRY QUESTIONS AND ANSWERS PER TOPIC
|
(a) describes one method that can be used to distinguish between sodium sulphate and sodium hydrogen sulphate.
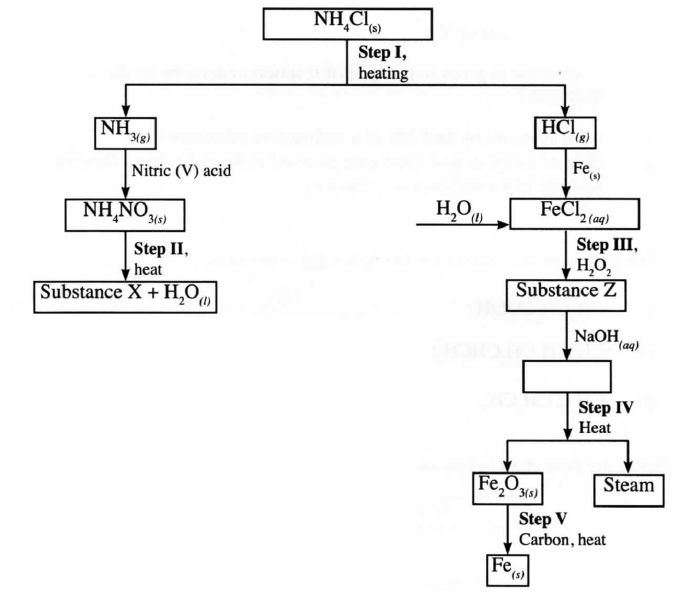
b) Describe how a pure sample of lead (II) sulphate can be prepared in the laboratory starting with lead metal c) Study the flow chart below and answer the questions that follow:
i) Write an equation for the reaction in:
I step II; II step IV ii) State the observation made in step III. Explain. iii) Name another substance that can be used in step V.
ANSWERS
(a)Test the acidity using a litmus pager. There will be no change on litmus when dipped into a solution of sodium sulphate . The litmus paper turns to red when dipped into a solution of sodium hydrogen sulphate .
OR Add a solid carbonate to each solution. No effervescence observed when the carbonate is added to a solution of sodium sulphate. Effervescence is observed when the carbonate is added to a solution of sodium hydrogen sulphate. (b)Add dilute nitric acid to lead to form a soluble salt, Pb(NO3)2, add a soluble salt, Na2SO4. Then dry the salt between filter papers . (c)(i) NH4 NO3 → N2O + 2H2O II 2Fe(OH)3(S) → Fe2O3(s) + 3H2O(l) (ii) The colour changes from pale green to brown . The iron (II) is oxidised to iron (III) chloride by hydrogen peroxide (iii) Carbon monoxide Related Chemistry Questions and Answers on Nitrogen and It's Compound Form 3 Level
1 Comment
|
Chemistry Topics
All
Archives
December 2024
|
We Would Love to Have You Visit Soon! |
Hours24 HR Service
|
Telephone0728 450425
|
|
8-4-4 materialsLevels
Subjects
|
cbc materialsE.C.D.E
Lower Primary
Upper Primary
Lower Secondary
Upper Secondary
|
teacher support
Other Blogs
|

 RSS Feed
RSS Feed