KCSE CHEMISTRY QUESTIONS AND ANSWERS PER TOPIC
|
Sodium thiosulphate solution reacts with dilute hydrochloric acid according to the following equation.
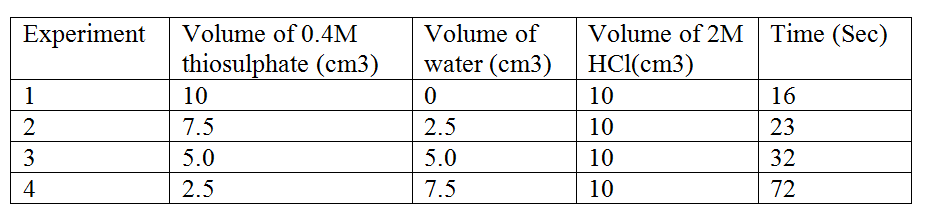
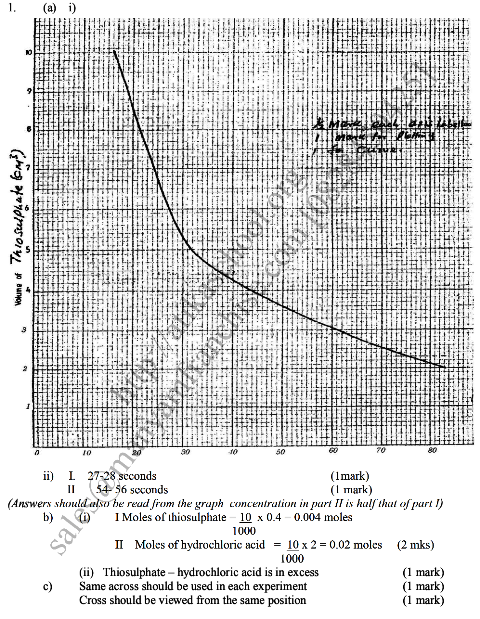
In an experiment to study how the rate of reaction varies withconcentration, 10cm3 of 0.4M sodium thiosulphate was mixed with 10cm3. Of 2M hydrochloric acid in a flask. The flask was placed in a white paper marked with across X.The time taken for the cross X become invisible when viewed from above was noted and recorded in the table below. The experiment was repeated three times as the temperature using the volumes in the table and the results recorded as shown in the table below. a) i) On the grid below, plot a graph of the volume of thiosulphate (Vertical axis) against time taken for the cross (X) to become invisible) ii) From the graph determine how long it would take for the cross to become invisible if the experiment was done. (3mks) i) Using 6cm3 of the 0.4M thiosulphate (1mk) ii) Using 6cm3 of 0.2M thiosulphate solution (1mk) b) i) Using values for experiment I.Calculate i) Moles of thiosulphate used (1mk) ii) Moles of hydrochloric acid used (1mk) ii) Explain which of the two reactants in experiment I controlled the rate of the reaction? Explain (1mk) c) Give two precautions which should be taken in experiment I controlled the rate of the reaction? Explain (2mk) Related Chemistry Questions and Answers on Reaction Rates and Reversible Reactions Form 4 Level
0 Comments
Leave a Reply. |
Chemistry Topics
All
Archives
December 2024
|
We Would Love to Have You Visit Soon! |
Hours24 HR Service
|
Telephone0728 450425
|
|
8-4-4 materialsLevels
Subjects
|
cbc materialsE.C.D.E
Lower Primary
Upper Primary
Lower Secondary
Upper Secondary
|
teacher support
Other Blogs
|


 RSS Feed
RSS Feed