|
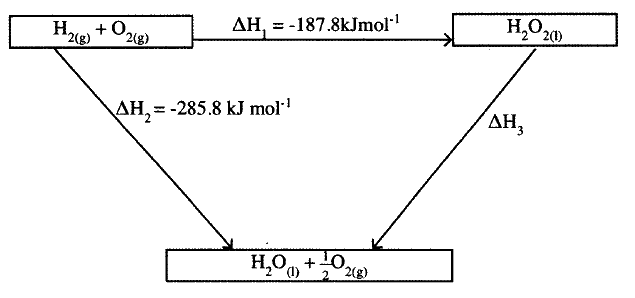
The figure below shows an energy cycle.
a) Give the name of the enthalpy change ΔH1.
b) Determine the value of ΔH3.
0 Comments
The curve below represents the changes in the concentration of substance E and F with time
In the reaction; E (g) ⇄ F(g)
(i) Which curve represents the changes in concentration of substance F? Give a reason
(ii) Give a reason for shapes of curves after time ‘t’ minutes
ANSWERS
a) (i) State the Le chatelier‟s principle.
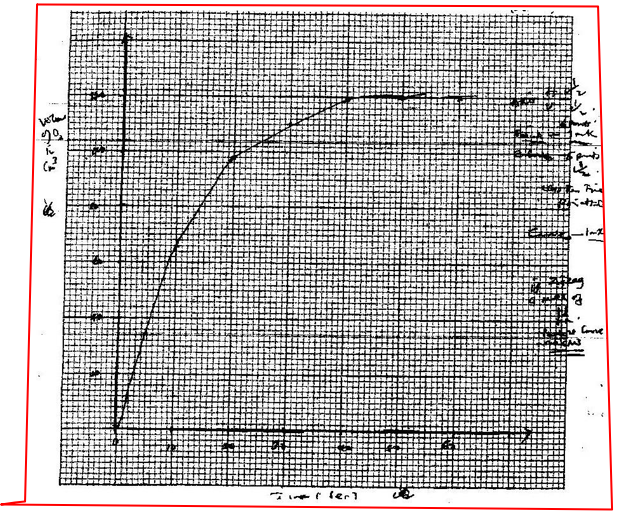
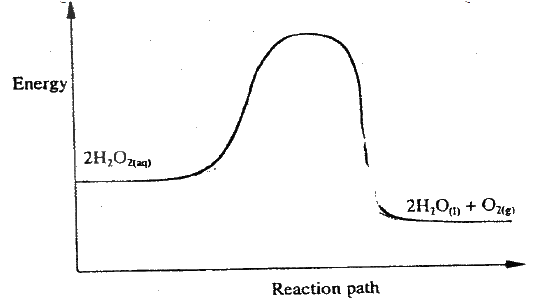
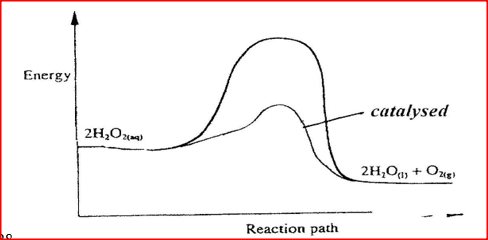
(ii) Carbon (II) oxide gas reacts with steam according to the equation; CO(g) + H2O(g)→ H2(g)+ CO2(g) What would be the effect of increasing the pressure of the system at equilibrium? Explain. b) The table below gives the volumes of oxygen gas produced at different times when hydrogen peroxide decomposed in the presence of a catalyst.
(i) Name the catalyst used for this reaction
(ii) On the grid provided, draw the graph of volume of oxygen gas produced (vertical axis) against time. (iii) Using the graph, determine the rate of decomposition of hydrogen peroxide after 24 seconds. (iv) Give a reason why the total volume of oxygen gas produced after 50 seconds remains constant.
ANSWERS
(a)(i) When change is made to a system in equilibrium the System moves so as to oppose the change.
(ii)Pressure has no effect to equilibrium The moles/Volume/ molecules of gases is reactants and product are equal (iii) DH –ve ( negative) Since lowering of temperature moves to equilibrium to direction which heat is produced. Decrease in temperature favours exothermic reaction (b) (i) Manganese IV oxide
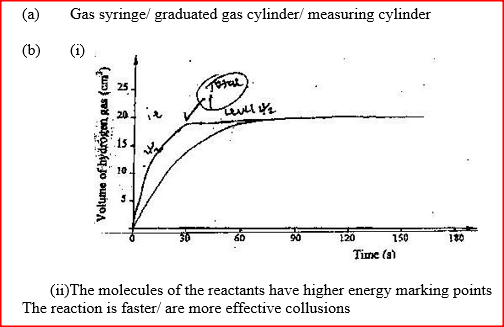
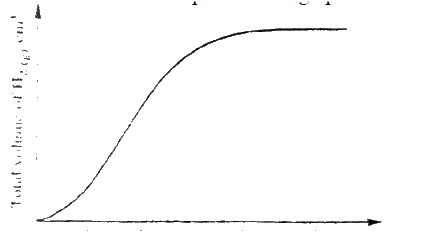
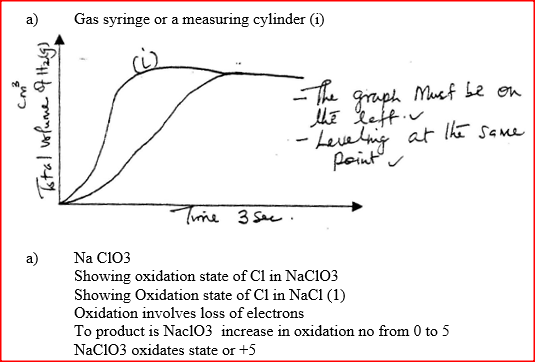
A certain mass of a metal E1 reacted with excess dilute hydrochloric acid at 25°C. The volume of hydrogen gas liberated was measured after every 30 seconds. The results were presented as shown in the graph below.
a) Name one piece of apparatus that may have been used to measure the volume of gas liberated. b) (i) On the same axis, sketch the curve that would be obtained if the experiment was repeated at 35°C. (ii) Explain the shape of your curve in b(i) above.
In a closed system, aqueous iron (III) chloride reacts with sulphide gas as shown in the equation below.
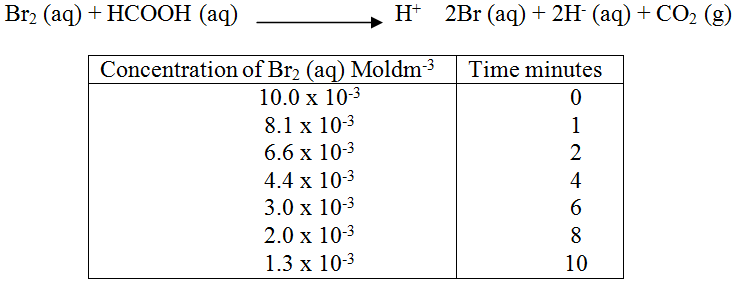
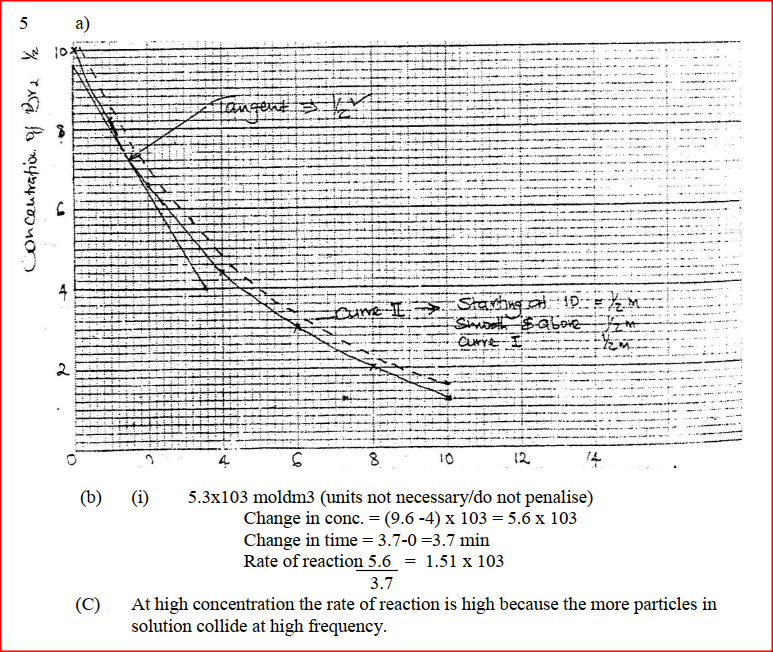
2FeCl3(aq) + H2S(g) → 2FeCl2(aq) 2HCl(aq) + S(s) State and explain the observation that would be made if dilute hydrochloric acid is added to the system at equilibrium. The reaction between and methanoic acid at 300C proceeds according to the information given below (a) On the grid below, plot a graph of concentration of Bromine (Vertical axis against time) (b) From the graph determine: (i) The concentration of bromine at the end of 3 minutes (ii) The rate of reaction at time ‘t’ where t = 1 ½ minutes (c) Explain how the concentration of bromine affects the rate of reaction (d) On the same axis sketch the curve that would be obtained if the reaction was carried out at 200C and label the curve as curve II. Give a reason for your answer.
(a) Methanol is manufactured from carbon (IV) oxide and hydrogen gas according to the equation:
The reaction is carried out in the presence of a chromium catalyst at 700K and 30kPa. Under these conditions, equilibrium is reacted when 2% of the carbon (IV) oxide is converted to methanol
(i) How does the rate of the forward reaction compare with that of the reverse reaction when 2% of the carbon (IV) oxide is converted to methanol? (ii) Explain how each of the following would affect the yield of methanol: I Reduction II Using a more efficient catalyst (iii) If the reaction is carried out at 500K and 30kPa, the percentage of carbon (IV) oxide converted to methanol is higher than 2% I what is the sign of ΔH for the reaction? Give a reason II Explain why in practice the reaction is carried out at 700K but NOT at 500K (b) Hydrogen peroxide decomposes according to the following equation : 2H2O2(aq) →2H2O(l) + O2 (g) In an experiment, the rate of decomposition of hydrogen peroxide was found to be 6.0 x 10-8 mol dm-3 S-1. (i) Calculate the number of moles per dm3 of hydrogen peroxide that had decomposed within the first 2 minutes (ii) In another experiment, the rate of decomposition was found to be 1.8 x 10-7 mol dm-3S-1. The difference in two rates could have been caused by addition of a catalyst. State, giving reasons, one other factor that may have caused the difference in two rates of decomposition
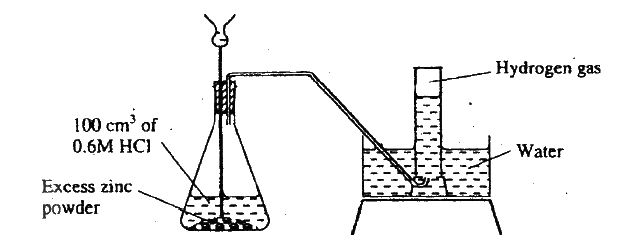
The diagram below shows a student's set-up for the preparation and collection of hydrogen gas.
(a) How would the final volume of hydrogen gas produced be affected if 80cm3 of 0,75M hydrochloric acid was used?
(b) Give a reason why helium is increasingly being preferred to hydrogen in weather balloons.
ANSWERS
(a) No change in volume since the number of moles of acid is equal in both cases.
(b) It is less dense and does not burn like hydrogen
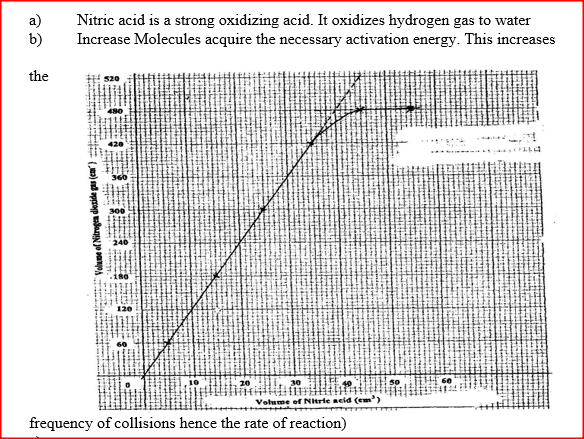
The table below shows the volumes of nitrogen dioxide gas produced when different volume of 1M nitric acid were each reacted with 2.07 g of lead at room temperature.
a) Give a reason why nitric acid is not used to prepare hydrogen gas. b) Explain how the rate of the reaction between lead and nitric acid would be affected if the temperature of the reaction mixture was raised. c) On the grid provided below, plot a graph of the volume of the gas produced (Vertical axis) against volume of acid.
d) Using the graph, determine the volume of
:i) Nitrogen dioxide produced when 30cm3 of 1 M nitric acid were reacted with 2.07 g of lead ii) 1M nitric acid which would react completely with 2.07g of lead. e) Using the answer in d(i) above, determine: i) The volume of 1M nitric acid that would react completely with one mole of lead (Pb=207) ii) The volume of nitrogen dioxide gas produced when one mole of lead reacts with excess 1 M nitric room temperature. f) Calculate the number of moles of: i) 1M nitric acid that reacted with one mole of lead ii) nitrogen dioxide produced when one mole of lead were reacted with excess nitric acid. (Molar gas volume of 2400cm3) g) Using the answers obtained in f (i) and (ii) above, write the equation for the reaction between lead and nitric acid given that one mole of lead nitrate and two moles of water were also produced.
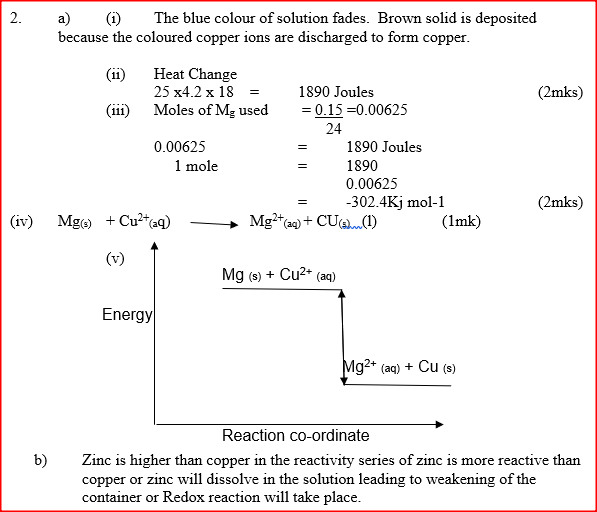
a) In an experiment to determine the molar heat of reaction when magnesium displaces copper ,0.15g of magnesium powder were added to 25.0cm3 of 2.0M copper (II) chloride solution. The temperature of copper (II) chloride solution was 25°C.While that of the mixture was 43°C.
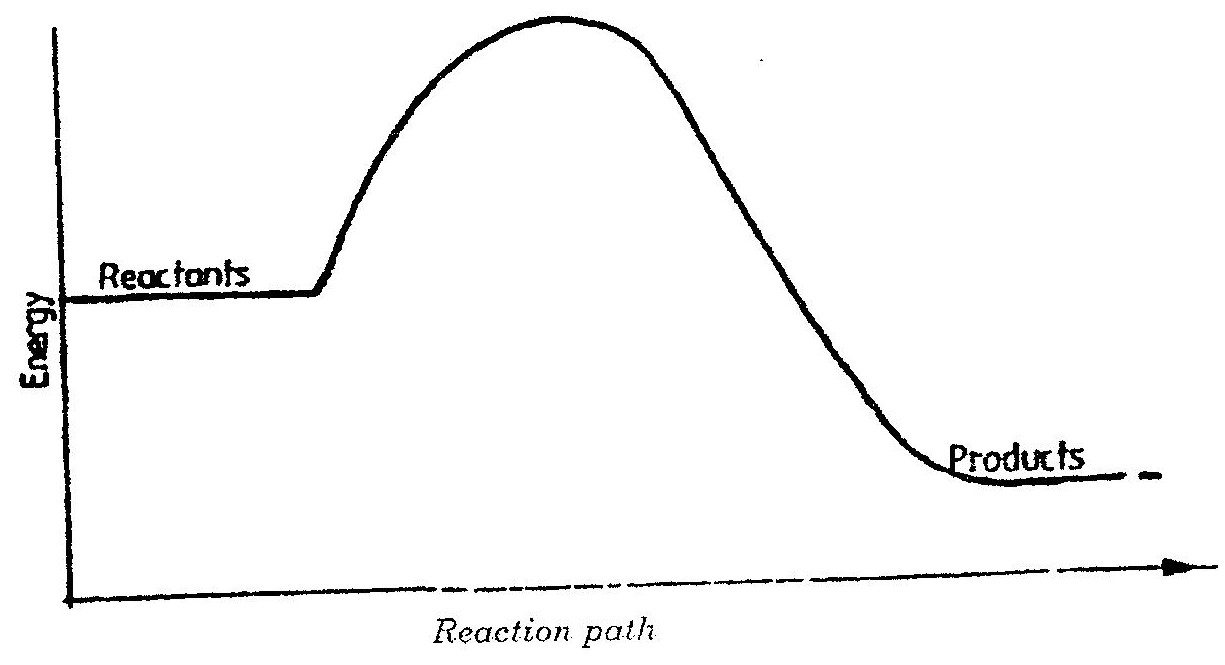
i) Other than increase in temperature, state and explain the observations which were made during the reaction. ii) Calculate the heat change during the reaction (specific heat capacity of the solution = 4.2jg-1k-1and the density of the solution = 1g/cm3 iii) Determine the molar heat of displacement of copper by magnesium.(Mg=24.0). iv) Write the ionic equation for the reaction. v) Sketch an energy level diagram for the reaction. b) Use the reduction potentials given below to explain why a solution containing copper ions should not be stored in a container made of zinc.
ANSWERS
(a) what condition is necessary for an equilibrium to be established?
(b) When calcium carbonate is heated, the equilibrium shown below is established CaCO3 (s) → CaO(s) + CO2(g) How would the position of equilibrium be affected if a small amount of dilute potassium hydroxide is added to the equilibrium mixture? Explain
ANSWERS
ANSWERS
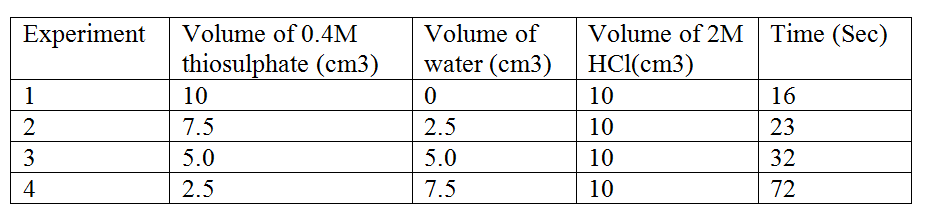
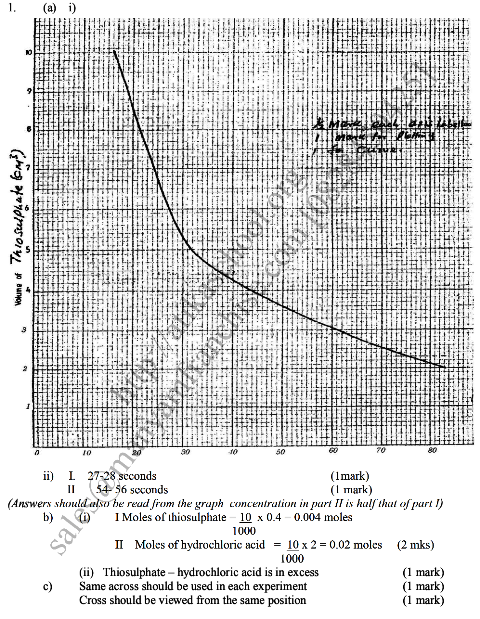
Sodium thiosulphate solution reacts with dilute hydrochloric acid according to the following equation.
In an experiment to study how the rate of reaction varies withconcentration, 10cm3 of 0.4M sodium thiosulphate was mixed with 10cm3. Of 2M hydrochloric acid in a flask. The flask was placed in a white paper marked with across X.The time taken for the cross X become invisible when viewed from above was noted and recorded in the table below. The experiment was repeated three times as the temperature using the volumes in the table and the results recorded as shown in the table below. a) i) On the grid below, plot a graph of the volume of thiosulphate (Vertical axis) against time taken for the cross (X) to become invisible) ii) From the graph determine how long it would take for the cross to become invisible if the experiment was done. (3mks) i) Using 6cm3 of the 0.4M thiosulphate (1mk) ii) Using 6cm3 of 0.2M thiosulphate solution (1mk) b) i) Using values for experiment I.Calculate i) Moles of thiosulphate used (1mk) ii) Moles of hydrochloric acid used (1mk) ii) Explain which of the two reactants in experiment I controlled the rate of the reaction? Explain (1mk) c) Give two precautions which should be taken in experiment I controlled the rate of the reaction? Explain (2mk)
The react between a piece of magnesium ribbon with excess 2m hydrochloric acid was investigated at 25°C by measuring the volume of hydrogen gas produced as the reaction progressed. The sketch below represents the graph that was obtained.
a) Name one piece of apparatus that may be used to measure the volume of hydrogen gas produced.
b) On the same diagram, sketch the curve that would be obtained if the experiment when excess chlorine gas was bubbled into hot concentrated sodium hydroxide, the following reaction occurred.
In which product did chorine undergo oxidation? Explain
Excess marble chips (calcium carbonate ) was put in a beaker containing 100cm3 of dilute hydrochloric acid. The beaker was then placed on a balance and the total loss in mass recorded after every two minutes as shown in the table below.
a) Why was there a loss in mass?
b) Calculate the average rate of loss in mass between: i) 0 and 2 minutes ii) 6 and 8 minutes iii) Explain the difference in the average rates of reaction in (b) (i) and (ii) above c) Write the equation for the reaction which takes place in the beaker d) State three ways in which the rate of the reaction above could be increased e) The solution in the beaker was evaporated to dryness what would happen if the open beaker and its contents were left in the laboratory overnight. f) Finally some water was added to the contents of the beaker. When aqueous sodium sulphate was added to the contents of the beaker, a white precipitate was formed. i) Identify the white precipitate ii) State one use of the substance identified in (f) (i) above
expected response
Increase in pressure would shift the equilibrium to the left; since in pressure favors thereaction will produce less volume of gas Ammonia can be converted to nitrogen monoxide as shown in the equation below (a) Explain how an increase in temperature would affect the yield of nitrogen monoxide (2mks) (b) On the energy level diagram above sketch, the energy level diagram that would be obtained if the reaction is carried out in the presence of platinum catalyst. (1 mk)
answer
(a) Solid dissolves
(b) addition of hydrochloric acid favour backward reaction State and explain how the rate of reaction between zinc granules and steam can be increased
answer
|
Chemistry Topics
All
Archives
December 2024
|
Can't find what you are looking for? Don't worry, Use the Search Box Below.
|
Primary Resources
College Resources
|
Secondary Resources
|
Contact Us
Manyam Franchise
P.O Box 1189 - 40200 Kisii Tel: 0728 450 424 Tel: 0738 619 279 E-mail - sales@manyamfranchise.com |




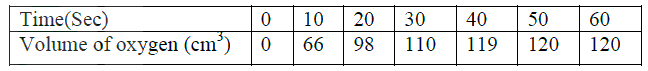




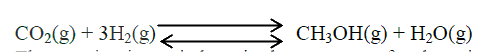

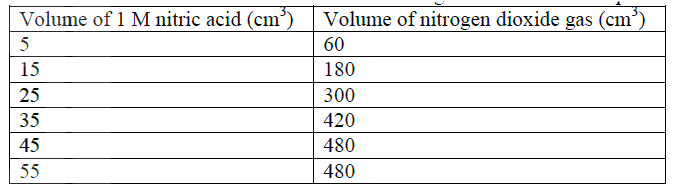

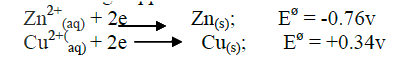



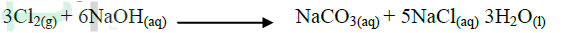
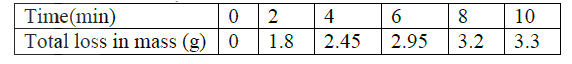
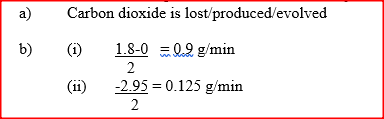
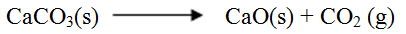


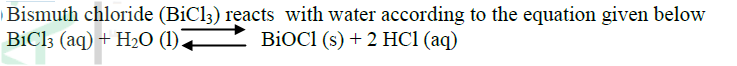





 RSS Feed
RSS Feed

