|
1. 1995 Q1 P1
Name the instrument that would be most suitable for measuring the thickest of one sheet of this question paper. (1 mark) 2. 1995 Q7 P1 State the assumption made when calculating the size of a molecule in the thin oil film experiment (1 mark) 3. 1996 Q1 P1
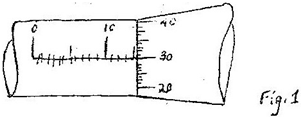
The micrometer screw gauge represented by figure 1 has thimble scale of 50 divisions
What is the reading shown (1 mark) 4. 1996 Q3 P1 State two factors that should be controlled in manufacturing a cylindrical container of uniform thickness, which should normally be in a standing position? 5. 1997 Q1 P1 Figure 1 shows a measuring cylinder, which contains water initially at level A. A solid of mass 11 g is immersed in the water, the level rises to B
Determine the density of the solid. (Give your answer to 1 decimal place)
6. 1997 Q4 P1 A butcher has a beam balance and masses 0.5 kg and 2kg. How would he measure 1.5 kg of meat on the balance at once? 7. 1998 Q1 P1 Figure 1 shows a fencing post whose length is being measured using a strip of a measuring tape.
Use this information to answer questions 7 and 8.
State the accuracy of the tape: 8. 1998 Q2 P1 What is the length of the post? 9. 2000 Q1 P1 Fig 1 shows part of a measuring cylinder calibrated in cm3 containing water whose level is indicated. Some 3.0cm of is added into the cylinder. Indicate on the diagram the new level of water.
10. 2001 Q1 P1
Fig. 1 shows a burette partly filed with a liquid. The burette was initially full to the mark O. If the quantity of the liquid removed has a mass of 22g, determine the density of the liquid.
11. 2001 Q3 P1
Fig 3 shows two identical hollow spheres. Spheres A is completely filled with the liquid while B is partly filled with identical liquid. When the two spheres are rolled gently on a horizontal surface. It is observed that the sphere B stops earlier that the sphere A. Explain this observation.
12. 2001 Q3a P2
You are provided with the following: A large shallow tray Water Lycopodium powder Some olive oil. Either a piece of thin wire with a kink and a millimetre scale or a Burette, a metre rule. Describe an experiment to estimate the diameter of a molecule of the Olive oil. 13. 2003 Q1 P1 Figure 1 shows a measuring cylinder containing some liquid Another 5cm3 of the liquid is added into the cylinder. Indicate on the diagram the new level of the liquid.
14. 2005 Q1 P1
Figure 1 shows the reading on a burette after 55 drops of a liquid have been used. If the initial reading was at Zero mark, determine the volume of one drop. (2 marks)
15. 2006 Q1 P1
Figure 1 shows the change in volume of water in a measuring cylinder when an irregular solid is immersed in it. Given that the mass of the solid is 567 g, determine the density of the solid in gcm-3 ( Give your answer correct to 2 decimal places) (3 marks)
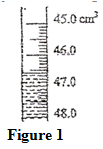
16. 2006 Q14b,c P1
(b) In an experiment to estimate the diameter of an oil molecule, an oil drop of diameter 0.05 cm spreads over a circular patch whose diameter is 20 cm Determine (i) The volume of the oil drop (2 marks) (ii) The area of the patch covered by the oil (2 marks) (iii) The diameter of the oil molecule (3 marks) (c) State (i) Any assumption made in (b) (iii) above (1 mark) (ii) Two possible sources of errors in this experiment (2 marks) 17. 2008 Q1 P1 A drug manufacturer gives the mass of the active ingredient in a tablet as 5 mg. Express this quantity in kilogramme and in standard form. (1 mark) 18. 2008 Q2 P1 The masses of equal volumes of a certain liquid and of water were found to be mv and mw respectively. Given that the density of water is 1gcm-3, express the density, p, of the liquid in terms of mv mw (show your work) (2 marks) 19. 2009 Q1 P1 In an experiment to measure the density of a liquid, a student filled a burette with a liquid to the 0cm3 mark . Figure 1 shows a section of the burette showing the level of the liquid after 54.5g of the liquid had been run out.
Determine the density of the liquid. (3 marks)
20. 2010 Q9 P1 When a drop of oleic acid of known volume is dropped on the surface of water in a large trough, it spreads out to form a large circular patch. State one assumption made when the size of the molecule of oleic acid is estimated by determining the area of the patch. 21. 2010 Q16 P1 In an experiment to determine the density of sand using a density bottle, the following measurements were recorded: Mass of empty density bottle - 43.2g Mass of density bottle full of water = 66.4g Mass of density bottle with some sand = 67.5g Filled up with water = 82.3g Use the above data to determine the: a) Mass of the water that completely filled the bottle: (2 marks) b) Volume of water that completely filled the bottle: (1 mark) c) Volume of the density bottle: (1 marks) d) Mass of sand e) Mass of water that filled the space above the sand. (1marks) f) Volume of the sand: g) Density of the sand (2 marks) 22. 2012 Q1 P1 Figure 1 shows a measuring cylinder containing some water.
Determine the reading on the measuring cylinder, after three drops of water each of volume 0.6cm3 are added (2 marks)
23. 2012 Q18a P1 (a) Figure 12 shows a set up used to determine the mass of a solid S. The rod is pivoted at its centre of gravity C. (i) State two measurements that need to be made to determine the mass of solid S. (1 mark) (ii) Write an expression to show how the measurements in (i) above are used to obtain the mass of S. (2 marks)
1 Comment
|
CATEGORIES
Categories
All
Topics
FORM I - PHYSICS SYLLABUSFORM II - PHYSICS SYLLABUSTOPICS
FORM III - PHYSICS SYLLABUSFORM IV - PHYSICS SYLLABUSARCHIVES
RSS FEEDS
AUTHOR
M.A NyamotiMy passion is to see students pass using right methods and locally available resources. My emphasis is STEM courses
|
We Would Love to Have You Visit Soon! |
Hours24 HR Service
|
Telephone0728 450425
|
|
8-4-4 materialsLevels
Subjects
|
cbc materialsE.C.D.E
Lower Primary
Upper Primary
Lower Secondary
Upper Secondary
|
teacher support
Other Blogs
|










 RSS Feed
RSS Feed